(+)-JQ1
Tocris Bioscience | Catalog # 4499

Key Product Details
Description
Product Description
(+)-JQ1 is a potent, high affinity, selective BET bromodomain inhibitor (IC50 values are 17.7, 32.6, 76.9 and 12942 nM for BRD2 (N-terminal (N)), BRD4 (C-terminal (C)), BRD4 (N) and CREBBP respectively; Kd values are 49, 59.5, 82, 90.1, 128 and 190 nM for BRD4 (N), BRD3 (N), BRD3 (C), BRD4 (C), BRD2 (N) and BRDT (N) respectively). (+)-JQ1 induces squamous differentiation in NUT midline carcinoma (NMC) cell lines and inhibits tumor growth in NMC xenograft models in vivo. (+)-JQ1 inhibits proliferation and induces autophagy in bladder cancer cells in vitro and in vivo. It also suppresses MYC gene expression and inhibits proliferation of lymphoma and leukemia cell lines. In human pulmonary microvasular endothelial cells (HPMEC), NF-κB activation, IL-6 and IL-8 expression and proliferation are inhibited by (+)-JQ1. The compound also inhibits transcription of ACE2 and TMPRSS2 genes in mouse lung tissue and prevents infection by SARS-CoV-2. In germ cells from male mice, (+)-JQ1 exhibits reversible contraceptive effects. (+)-JQ1 inhibits the BRD4-JUN-CCL2-TNF-α axis in pancreatic cancer cells and improves survival by reducing macrophage recruitment.Inactive Analog also available.
Carboxylic acid-functionalized (Cat. No. 6588) and click-activated (alkyne) (Cat. No. 6589) versions for PROTAC development also available.
Licensing Information
This probe is supplied in conjunction with the Structural Genomics Consortium. For further characterization details, please visit the (+)-JQ1 probe summary on the SGC website.External Portal Information
Chemicalprobes.org is a portal that offers independent guidance on the selection and/or application of small molecules for research. The use of (+)-JQ1 is reviewed on the chemical probes website.
Product Specifications for (+)-JQ1
Molecular Weight
Formula
Storage
Purity
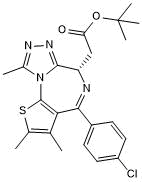
Chemical Name
CAS Number
PubChem ID
InChI Key
SMILES
The technical data provided above is for guidance only. For batch specific data refer to the Certificate of Analysis.
Solubility
| Solvent | Max Conc. mg/mL | Max Conc. mM | |
|---|---|---|---|
| Solubility | |||
| DMSO | 45.7 | 100 | |
| Ethanol | 45.7 | 100 |
Preparing Stock Solutions for (+)-JQ1
The following data is based on the product molecular weight 456.99.
Batch specific molecular weights may vary from batch to batch due to the degree of hydration, which all affect the solvent volumes required to prepare stock solutions.
| Concentration / Solvent Volume / Mass | 1 mg | 5 mg | 10 mg |
|---|---|---|---|
| 1 mM | 2.19 mL | 10.94 mL | 21.88 mL |
| 5 mM | 0.44 mL | 2.19 mL | 4.38 mL |
| 10 mM | 0.22 mL | 1.09 mL | 2.19 mL |
| 50 mM | 0.04 mL | 0.22 mL | 0.44 mL |
Calculators
Background References
References are publications that support the biological activity of the product. See our Citations tab to view 44 publications citing the usage of this product.
- Tu TNF-a-producing macrophages determine subtype identity and prognosis via AP1 enhancer reprogramming in pancreatic cancer. Nat.Cancer 2021 PMID: 35122059
- Qiao Targeting transcriptional regulation of SARS-CoV-2 entry factors ACE2 and TMPRSS2. Proc.Natl.Acad.Sci.U.S.A. 2020 PMID: 33310900
- Mertz Targeting MYC dependence in cancer by inhibiting BET bromodomains. Proc.Natl.Acad.Sci.U.S.A. 2011 PMID: 21949397
- Mumby Bromodomain and extra-terminal protein mimic JQ1 decreases inflammation in human vascular endothelial cells: Implications for pulmonary arterial hypertension. Respirology 2017 PMID: 27539364
- Li BET inhibitor JQ1 suppresses cell proliferation via inducing autophagy and activating LKB1/AMPK in bladder cancer cells. Cancer Med. 2019 PMID: 31250978
- Matzuk Small-molecule inhibition of BRDT for male contraception. Cell 2012 PMID: 22901802
- Herrmann Small-molecule inhibition of BRD4 as a new potent approach to eliminate leukemic stem- and progenitor cells in acute myeloid leukemia AML. Oncotarget 2012 PMID: 23249862
- Filippakopoulos Selective inhibition of BET bromodomains. Nature 2010 PMID: 20871596
Product Documents for (+)-JQ1
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for (+)-JQ1
For research use only
Citations for (+)-JQ1
Customer Reviews for (+)-JQ1 (2)
Have you used (+)-JQ1?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
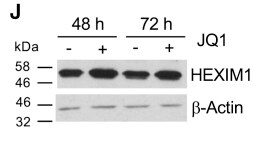
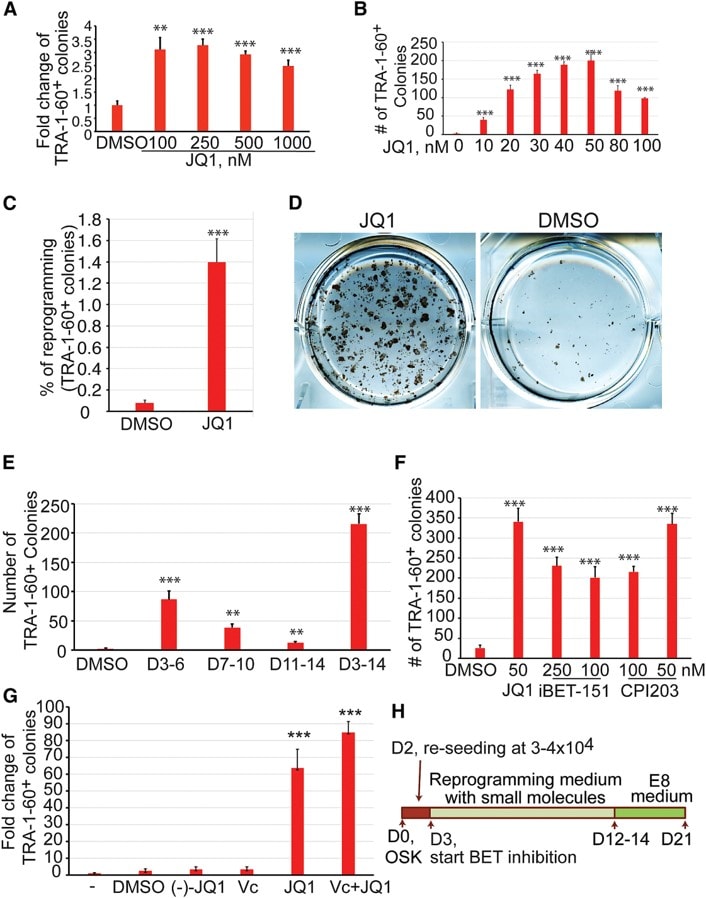
Species: HumanAssay Type: In VitroCell Line/Tissue: U2OSVerified Customer | Posted 06/22/2020JQ1 (1 μM)
-
Species: MouseAssay Type: In VitroCell Line/Tissue: ipscVerified Customer | Posted 02/23/2020500 nM
There are no reviews that match your criteria.