LC3A Antibody - BSA Free
Novus Biologicals | Catalog # NBP1-19167

![Western Blot: LC3A Antibody [NBP1-19167] Western Blot: LC3A Antibody [NBP1-19167]](https://resources.rndsystems.com/images/products/LC3A-Antibody-Western-Blot-NBP1-19167-img0012.jpg)
Key Product Details
Validated by
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Format
Product Specifications
Immunogen
Localization
Marker
Specificity
Clonality
Host
Isotype
Theoretical MW
Disclaimer note: The observed molecular weight of the protein may vary from the listed predicted molecular weight due to post translational modifications, post translation cleavages, relative charges, and other experimental factors.
Scientific Data Images for LC3A Antibody - BSA Free
Western Blot: LC3A Antibody [NBP1-19167]
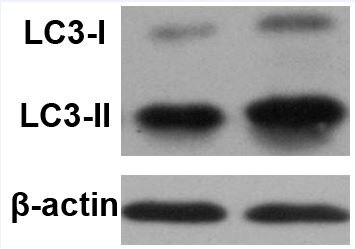
Western Blot: LC3A Antibody [NBP1-19167] - Total protein from HeLa and Neuro2A cells treated with or without 50 uM chloroquine for 24 hours was separated on a 4-15% gel by SDS-PAGE, transferred to 0.2 um PVDF membrane and blocked in 5% non-fat milk in TBST. The membrane was probed with 2.0 ug/mL anti-LC3A in 1% non-fat milk in TBST and detected with an anti-rabbit HRP secondary antibody using chemiluminescence. Note the detection LC3 II upon chloroquine treatment.Immunocytochemistry/ Immunofluorescence: LC3A Antibody [NBP1-19167]
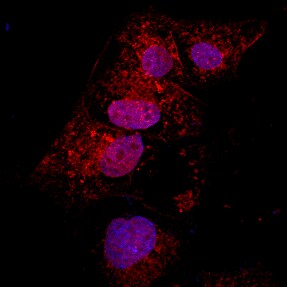
Immunocytochemistry/Immunofluorescence: LC3A Antibody [NBP1-19167] - LC3/MAP1 [NBP1-19167] - LC3 antibody was tested in HeLa cells with Dylight 488 (green). Cells were treated overnight with 50 uM chloroquine to induce autophagosome formation. Nuclei and alpha-tubulin were counterstained with DAPI (blue) and Dylight 550 (red).Immunohistochemistry-Paraffin: LC3A Antibody [NBP1-19167]
Immunohistochemistry-Paraffin: LC3A Antibody [NBP1-19167] - Analysis of a FFPE tissue section of mouse brain using LC3 antibody at 1:300 dilution. The signal was developed using HRP-labelled secondary antibody and DAB reagent, and the sections/nuclei were further counterstained with hematoxylin. Note the diffused cytoplasmic staining of LC3 in all of the cells with highest positivity in various neurons.Western Blot: LC3A Antibody [NBP1-19167]
Western Blot: LC3/MAP1LC3A Antibody [NBP1-19167] - Human brain lysate.Immunohistochemistry-Paraffin: LC3A Antibody [NBP1-19167]
Immunohistochemistry-Paraffin: LC3A Antibody [NBP1-19167] - Analysis of a FFPE tissue section of mouse liver using LC3 antibody at 1:300 dilution. The signal was developed using HRP-labelled secondary antibody and DAB reagent, and the sections/nuclei were further counterstained with hematoxylin. Note the diffused cytoplasmic staining of LC3 in all of the hepatocytes and other liver cells.Simple Western: LC3A Antibody [NBP1-19167]
Simple Western: LC3A Antibody [NBP1-19167] - Image shows a specific band for LC3 in 0.5 mg/mL of Neuro2A lysate. This experiment was performed under reducing conditions using the 12-230 kDa separation system.Applications for LC3A Antibody - BSA Free
Flow Cytometry
Immunocytochemistry/ Immunofluorescence
Immunohistochemistry
Immunohistochemistry-Frozen
Immunohistochemistry-Paraffin
Simple Western
Western Blot
In Simple Western only 10 - 15 uL of the recommended dilution is used per data point.
See Simple Western Antibody Database for Simple Western validation: Tested in Neuro2A lysate 0.5 mg/mL, separated by Size, antibody dilution of 1:40, apparent MW was 17 kDa. Separated by Size-Wes, Sally Sue/Peggy Sue.
The observed molecular weight of the protein may vary from the listed predicted molecular weight due to post translational modifications, post translation cleavages, relative charges, and other experimental factors.
Reviewed Applications
Read 2 reviews rated 4.5 using NBP1-19167 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Formulation
Format
Preservative
Concentration
Shipping
Stability & Storage
Background: LC3A
The process of autophagy is associated with a variety of diseases including neurodegenerative diseases, neuromuscular, tumorigenesis, and viral and bacterial infections (4). LC3 is a useful marker of autophagy in both healthy and diseased cells (4). Interestingly, LC3A has two variants (v1 and v2) which differ in N-terminal sequence due to the varying transcriptional start sites (5). One particular study found that LC3Av1, but not v2 or LC3B, was silenced in various cancer cell lines due to aberrant DNA methylation and re-expression of LC3Av1 in LC3Av1-silenced cells inhibited tumor growth, where overall findings suggest a possible tumor-suppressive role (5).
Alternative names for LC3A include Apg8, APG8a, ATG8E, Autophagy-related protein LC3 A, Autophagy-related ubiquitin-like modifier LC3 A, MAP1A/1B light chain 3 A, microtubule-associated proteins 1A/1B light chain 3, and MLP3A.
References
1. Shpilka, T., Weidberg, H., Pietrokovski, S., & Elazar, Z. (2011). Atg8: an autophagy-related ubiquitin-like protein family. Genome biology. https://doi.org/10.1186/gb-2011-12-7-226
2. Koukourakis, M. I., Kalamida, D., Giatromanolaki, A., Zois, C. E., Sivridis, E., Pouliliou, S., Mitrakas, A., Gatter, K. C., & Harris, A. L. (2015). Autophagosome Proteins LC3A, LC3B and LC3C Have Distinct Subcellular Distribution Kinetics and Expression in Cancer Cell Lines. PloS one. https://doi.org/10.1371/journal.pone.0137675
3. Weidberg, H., Shvets, E., & Elazar, Z. (2011). Biogenesis and cargo selectivity of autophagosomes. Annual review of biochemistry. https://doi.org/10.1146/annurev-biochem-052709-094552
4. Tanida, I., Ueno, T., & Kominami, E. (2004). LC3 conjugation system in mammalian autophagy. The international journal of biochemistry & cell biology. https://doi.org/10.1016/j.biocel.2004.05.009
5. Schaaf, M. B., Keulers, T. G., Vooijs, M. A., & Rouschop, K. M. (2016). LC3/GABARAP family proteins: autophagy-(un)related functions. FASEB journal : official publication of the Federation of American Societies for Experimental Biology. https://doi.org/10.1096/fj.201600698R
Long Name
Alternate Names
Gene Symbol
UniProt
Additional LC3A Products
Product Documents for LC3A Antibody - BSA Free
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for LC3A Antibody - BSA Free
This product is for research use only and is not approved for use in humans or in clinical diagnosis. Primary Antibodies are guaranteed for 1 year from date of receipt.
Related Research Areas
Citations for LC3A Antibody - BSA Free
Customer Reviews for LC3A Antibody - BSA Free (2)
Have you used LC3A Antibody - BSA Free?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Application: ImmunocytochemistrySample Tested: MEF cellsSpecies: MouseVerified Customer | Posted 06/12/2015anti-LC3 (NB100-2331) in PFA fixed MEF cells
-
Application: Western BlotSample Tested: Rat bone marrow stromal cellsSpecies: RatVerified Customer | Posted 02/24/2015LC3 expression in rat bone marrow stromal cells with different passage
There are no reviews that match your criteria.
Protocols
View specific protocols for LC3A Antibody - BSA Free (NBP1-19167):
Immunocytochemistry Protocol
Culture cells to appropriate density in 35 mm culture dishes or 6-well plates.
1. Remove culture medium and add 10% formalin to the dish. Fix at room temperature for 30 minutes.
2. Remove the formalin and add ice cold methanol. Incubate for 5-10 minutes.
3. Remove methanol and add washing solution (i.e. PBS). Be sure to not let the specimen dry out. Wash three times for 10 minutes.
4. To block nonspecific antibody binding incubate in 10% normal goat serum from 1 hour to overnight at room temperature.
5. Add primary antibody at appropriate dilution and incubate at room temperature from 2 hours to overnight at room temperature.
6. Remove primary antibody and replace with washing solution. Wash three times for 10 minutes.
7. Add secondary antibody at appropriate dilution. Incubate for 1 hour at room temperature.
8. Remove antibody and replace with wash solution, then wash for 10 minutes. Add Hoechst 33258 to wash solution at 1:25,0000 and incubate for 10 minutes. Wash a third time for 10 minutes.
9. Cells can be viewed directly after washing. The plates can also be stored in PBS containing Azide covered in Parafilm (TM). Cells can also be cover-slipped using Fluoromount, with appropriate sealing.
*The above information is only intended as a guide. The researcher should determine what protocol best meets their needs. Please follow safe laboratory procedures.
Immunohistochemistry-paraffin embedded sections
Antigen Unmasking
Bring slides to a boil in 10 mM sodium citrate buffer pH 6.0 then maintain at a sub-boiling temperature for 10 minutes. Cool slides on bench top for 30 minutes.
Staining
1. Wash sections in dH2O three times for 5 minutes each.
2. Wash section in wash buffer (1X PBS/0.1% Tween-20 (1X PBST)) for 5 minutes.
3. Block each section with 100-400 ul blocking solution (1X PBST, 5% goat serum) for 1 hour at room temperature.
4. Remove blocking solution and add 100-400 ul primary antibody diluted in 1X PBST, 5% goat serum to each section. Incubate overnight at 4C.
5. Remove antibody solution and wash sections in wash buffer three times for 5 minutes each.
6. Add 100-400 ul biotinylated secondary antibody, diluted in 1X PBST, 5% goat serum. Incubate 30 minutes at room temperature.
7. Remove secondary antibody solution and wash sections three times with wash buffer for 5 minutes each.
8. Add 100-400 ul Striptavidin-HRP reagent to each section and incubate for 30 minutes at room temperature.
9. Wash sections three times in wash buffer for 5 minutes each.
10. Add 100-400 ul DAB substrate to each section and monitor staining closely.
11. As soon as the sections develop, immerse slides in dH2O.
12. Counterstain sections in hematoxylin.
13. Wash sections in dH2O two times for 5 minutes each.
14. Dehydrate sections.
15. Mount coverslips.
Protocol: Inhibition of Autophagy and LC3 Antibody (NBP1-19167) Western Blot
Materials
Chloroquine diphosphate (CQ) (10 mM) in dH2O
1X PBS
Sample buffer, 2X Laemmli buffer: 4% SDS, 5% 2-mercaptoethanol (BME), 20% glycerol, 0.004% bromophenol blue, 0.125 M Tris HCl, pH 6.8
RIPA buffer: 150 mM NaCl, 1% NP-40 or Triton X-100, 0.5% sodium deoxycholate, 0.1% SDS, 50 mM Tris-HCl, pH 8.0, 20 mM Tris-HCl, pH 7.5
1X Running Buffer: 25 mM Tris-base, 192 mM glycine, 0.1% SDS. Adjust to pH 8.3
1X Transfer buffer (wet): 25 mM Tris-base, 192 mM glycine, 20% methanol, Adjust to pH 8.3
TBS
TBST, TBS and 0.1% Tween
Blocking solution: TBST, 5% non-fat dry milk
rabbit anti-LC3 primary antibody (NBP1-19167) in blocking buffer (~2 ug/mL)
Methods
Tip: For more information on Western Blotting, see our Western Blot handbook.
1. Grow cells (e.g. HeLa or Neuro2A) in vitro to semi-confluency (70-75%).
2. Add CQ to culture dishes to a final concentration of 50 uM and incubate overnight (16 hours). Remember to include an untreated sample as a negative control.
Note: Validated autophagy inducers should be included as positive controls.
3. Rinse cells with ice-cold 1X PBS and lyse cells with sample buffer.
Note: LC3-I and LC3-II are sensitive to degradation, although LC3-I is more labile. These proteins are sensitive to freeze-thaw cycles and SDS sample buffers. Fresh samples should be analyzed quickly to prevent protein degradation.
4. Sonicate and incubate cells for 5 minutes at 95oC.
Tip: Cells are lysed directly in sample buffer or may be lysed in RIPA buffer.
5. Load samples of Chloroquine-treated and -untreated cell lysates 40 ug/lane on a 4-20% polyacrylamide gradient gel (SDS-PAGE).
Tip: For detection of LC3 it is particularly important to monitor the progress of the gel as this protein is relatively small (~14kDa).
Tip: Alternatively, for non-gradient gels, use a 20% polyacrylamide gel.
6. Transfer proteins to a 0.2 um PVDF membrane for 30 minutes at 100V.
7. After transfer, rinse the membrane with dH2O and stain with Ponceau S for 1-2 minutes to confirm efficiency of protein transfer.
8. Rinse the membrane in dH2O to remove excess stain and mark the loaded lanes and molecular weight markers using a pencil.
9. Block the membrane using blocking buffer solution (5% non-fat dry milk in TBST) for 1 hour at room temperature.
10.Rinse the membrane with TBST for 5 minutes.
11.Dilute the rabbit anti-LC3 primary antibody (NBP1-19167) (~2 ug/mL) in blocking buffer and incubate the membrane for 1 hour at room temperature.
12.Rinse the membrane with dH2O.
13.Rinse the membrane with TBST, 3 times for 10 minutes each.
14.Incubate the membrane with diluted secondary antibody, according with product's specifications, (e.g. anti-rabbit-IgG HRP-conjugated) in blocking buffer for 1 hour at room temperature.
Note: Tween-20 may be added to the blocking or antibody dilution buffer at a final concentration of 0.05-0.2%, provided it does not interfere with antibody-antigen binding.
15.Rinse the membrane with TBST, 3 times for 10 minutes each.
16.Apply the detection reagent of choice (e.g. BioFX Super Plus ECL) in accordance with the manufacturer's instructions.
17.Image the blot.
Tip: LC3-I and it's lipidated form LC3-II have different electrophoretic mobility properties, with the lipidated form moving faster in an SDS-PAGE gel, albeit its larger molecular weight. LC3-II runs at 14-16 kDa while LC3-I runs at 16-18kDa.
Note: This assay measures the difference in the LC3-II signal in the presence and absence of inhibitors (e.g., lysosomotropic agents). When autophagic flux is present or induced in a system an increase in the LC3-II signal should be observed with the inhibitor.
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
FAQs for LC3A Antibody - BSA Free
-
Q: We have received an antibody against LC3 (# NBP1-19167) and I need a precision regarding its specificity. According to the datasheet, the antibody was raised against a peptide 100% identical to LC3A and 62% to LC3B. My question is: Is LC3A used as a synonym for LC3-I and LC3B as a synonym of LC3-II?
A: LC3A is not a synonym for LC3-I or LC3-II. When we refer to LC3-I and -II, we actually mean LC3B-I and LC3B-II. I hope this helps, but please let me know if you have any questions.
-
Q: We purchased an antibody against LC3 (NBP1-19167) from your company. According to the information found on your web site, three papers used this antibody to detect both the LC3-I and LC3-II form of the protein. The catalog number of the antibody is not mentioned in the papers. How can I be sure that it is the one used by the authors? It is important since I want to know if this antibody NBP1-19167 recognize both LC3 forms.
A: Our publication evaluation team is highly qualified and we found that these publications were cited under # NBP1-19167 after consulting the archived order information of the customers. As far as the detection of the two forms is concerned, any given LC3 antibody should detect LC3-I as well as LC3-II forms because the only difference between the two forms is that LC3-II carries the phosphatidylethanolamine (PE) conjugate whereas LC3-I does not have a PE conjugate. In nutshell, following translation, the unprocessed form of LC3 (pro-LC3) is proteolytically cleaved by Atg4 protease, resulting in the cytosolic form of LC3 (LC3-I) with a carboxyterminal exposed glycine. Upon induction of autophagy, the exposed glycine of LC3-I is conjugated by Atg7 (an E1-like activity), Atg3 (an E2-like conjugating activity) and by Atg12-Atg5-Atg16L multimers (E3-like ligase activity) to the highly lipophilic PE moiety to generate LC3-II (which is recruited to autophagosomal membranes). Therefore, the presence of both bands (LC3-I & LC3-II) will depend upon the extent of autophagy in your particular samples.
-
Q: We have received an antibody against LC3 (# NBP1-19167) and I need a precision regarding its specificity. According to the datasheet, the antibody was raised against a peptide 100% identical to LC3A and 62% to LC3B. My question is: Is LC3A used as a synonym for LC3-I and LC3B as a synonym of LC3-II?
A: LC3A is not a synonym for LC3-I or LC3-II. When we refer to LC3-I and -II, we actually mean LC3B-I and LC3B-II. I hope this helps, but please let me know if you have any questions.
-
Q: We purchased an antibody against LC3 (NBP1-19167) from your company. According to the information found on your web site, three papers used this antibody to detect both the LC3-I and LC3-II form of the protein. The catalog number of the antibody is not mentioned in the papers. How can I be sure that it is the one used by the authors? It is important since I want to know if this antibody NBP1-19167 recognize both LC3 forms.
A: Our publication evaluation team is highly qualified and we found that these publications were cited under # NBP1-19167 after consulting the archived order information of the customers. As far as the detection of the two forms is concerned, any given LC3 antibody should detect LC3-I as well as LC3-II forms because the only difference between the two forms is that LC3-II carries the phosphatidylethanolamine (PE) conjugate whereas LC3-I does not have a PE conjugate. In nutshell, following translation, the unprocessed form of LC3 (pro-LC3) is proteolytically cleaved by Atg4 protease, resulting in the cytosolic form of LC3 (LC3-I) with a carboxyterminal exposed glycine. Upon induction of autophagy, the exposed glycine of LC3-I is conjugated by Atg7 (an E1-like activity), Atg3 (an E2-like conjugating activity) and by Atg12-Atg5-Atg16L multimers (E3-like ligase activity) to the highly lipophilic PE moiety to generate LC3-II (which is recruited to autophagosomal membranes). Therefore, the presence of both bands (LC3-I & LC3-II) will depend upon the extent of autophagy in your particular samples.

![Immunocytochemistry/ Immunofluorescence: LC3A Antibody [NBP1-19167] Immunocytochemistry/ Immunofluorescence: LC3A Antibody [NBP1-19167]](https://resources.rndsystems.com/images/products/LC3A-Antibody-Immunocytochemistry-Immunofluorescence-NBP1-19167-img0013.jpg)
![Immunohistochemistry-Paraffin: LC3A Antibody [NBP1-19167] Immunohistochemistry-Paraffin: LC3A Antibody [NBP1-19167]](https://resources.rndsystems.com/images/products/LC3A-Antibody-Immunohistochemistry-Paraffin-NBP1-19167-img0014.jpg)
![Western Blot: LC3A Antibody [NBP1-19167] Western Blot: LC3A Antibody [NBP1-19167]](https://resources.rndsystems.com/images/products/LC3-MAP1LC3A-Antibody-Western-Blot-NBP1-19167-img0004.jpg)
![Immunohistochemistry-Paraffin: LC3A Antibody [NBP1-19167] Immunohistochemistry-Paraffin: LC3A Antibody [NBP1-19167]](https://resources.rndsystems.com/images/products/LC3-MAP1LC3A-Antibody-Immunohistochemistry-Paraffin-NBP1-19167-img0010.jpg)
![Simple Western: LC3A Antibody [NBP1-19167] Simple Western: LC3A Antibody [NBP1-19167]](https://resources.rndsystems.com/images/products/LC3-MAP1LC3A-Antibody-Simple-Western-NBP1-19167-img0009.jpg)