Delta-Like protein 4 (DLL4) is a type I membrane protein belonging to the Delta/Serrate/Lag2 (DSL) family of Notch ligands (1). Notch signaling is an evolutionarily conserved pathway that controls cell fate and is required in multiple developmental processes including vascular development, hematopoiesis, somatogenesis, myogenesis, and neurogenesis (2-4). Dysregulation in the Notch pathway is associated with various human diseases. In mammals, four Notch homologs (Notch 1 to 4) and five ligands (DLL 1, 3 and 4, Jagged 1 and 2) have been identified. Notch ligands are transmembrane proteins with a DSL motif necessary for Notch binding, tandem EGF repeats, a transmembrane region and a short intracellular domain (ICD). Notch ligands are categorized into two subfamilies based on the presence of an extracellular cysteine-rich domain and insertions that interrupt some EGF repeats in the Jagged but not the Delta ligand family. Interactions of Notch receptors with their ligands results in reciprocal Regulated Intramembrane Proteolysis (RIP) (4). RIP is a mechanism for transmembrane signal transduction that involves the sequential processing by A Disintegrin Metalloprotease (ADAM) and then by Presenilin/ gamma -Ssecretase, resulting in shedding of the extracellular domains and the generation of the soluble ICD signaling fragments, respectively. The Notch ICD translocates to the nucleus and interacts with transcriptional coactivators, resulting in the transcription of target genes. The ICDs of the Notch ligands have also been shown to translocate to the nucleus where they may have a signaling function (5, 6). DLL4 is expressed highly and selectively within the arterial endothelium and has been shown to function as a ligand for Notch 1 and Notch 4. Human and mouse DLL4 share 86% amino acid sequence identity (1).

Key Product Details
Species Reactivity
Validated:
Mouse
Cited:
Human, Mouse, Rat, Chicken, Transgenic Mouse
Applications
Validated:
Immunohistochemistry, Western Blot, Immunocytochemistry
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Immunohistochemistry-Frozen, Western Blot, Immunocytochemistry, IHC-F
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant mouse DLL4
Ser28-Pro525
Accession # BAB18580
Ser28-Pro525
Accession # BAB18580
Specificity
Detects mouse DLL4 in direct ELISAs and Western blots. In direct ELISAs, approximately 50% cross‑reactivity with recombinant human DLL4 is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Mouse DLL4 Antibody
Detection of Mouse DLL4 by Western Blot.
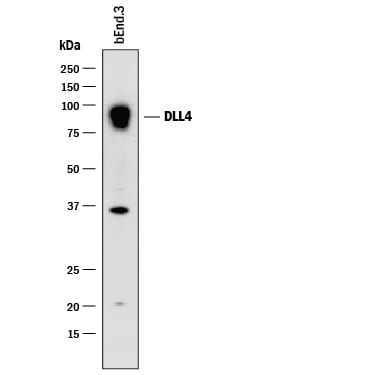
Western blot shows lysates of bEnd.3 mouse endothelioma cell line. PVDF membrane was probed with 2 µg/mL of Goat Anti-Mouse DLL4 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1389) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF017). A specific band was detected for DLL4 at approximately 90 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.DLL4 in bEnd.3 Mouse Cell Line.
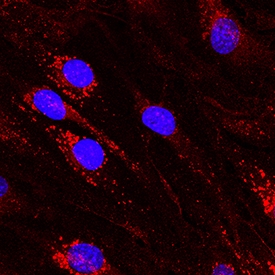
DLL4 was detected in immersion fixed bEnd.3 mouse endothelioma cell line using Goat Anti-Mouse DLL4 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1389) at 10 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; Catalog # NL001) and counterstained with DAPI (blue). Specific staining was localized to cytoplasm. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.DLL4 in Mouse Embryo.
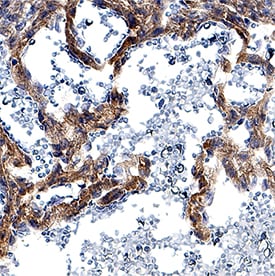
DLL4 was detected in immersion fixed paraffin-embedded sections of mouse embryo (13 d.p.c.) using Goat Anti-Mouse DLL4 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1389) at 5 µg/mL for 1 hour at room temperature followed by incubation with the Anti-Goat IgG VisUCyte™ HRP Polymer Antibody (Catalog # VC004). Tissue was stained using DAB (brown) and counterstained with hematoxylin (blue). Specific staining was localized to developing vasculature. View our protocol for IHC Staining with VisUCyte HRP Polymer Detection Reagents.Detection of Mouse DLL4 by Western Blot
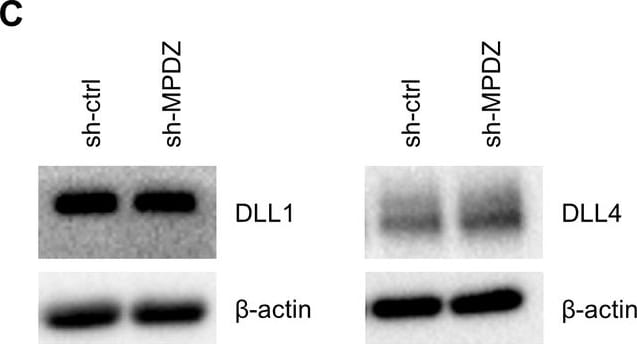
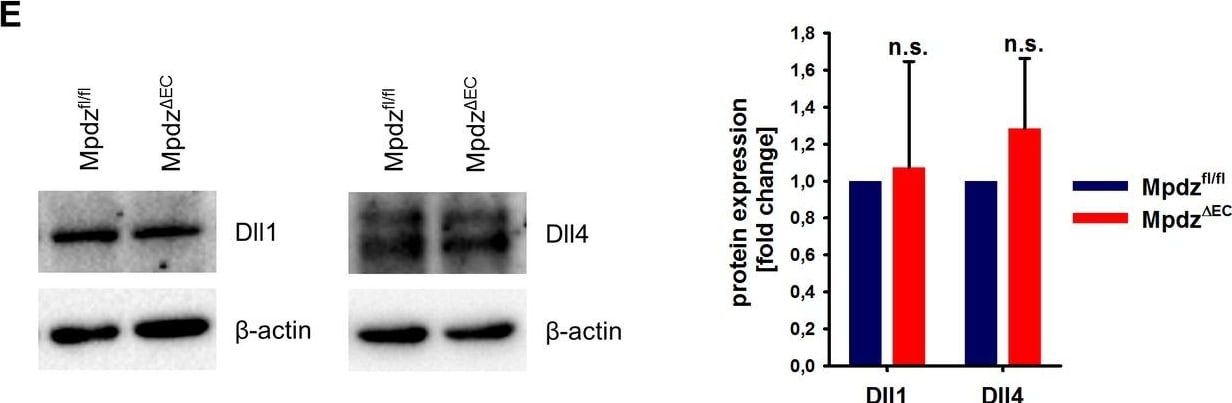
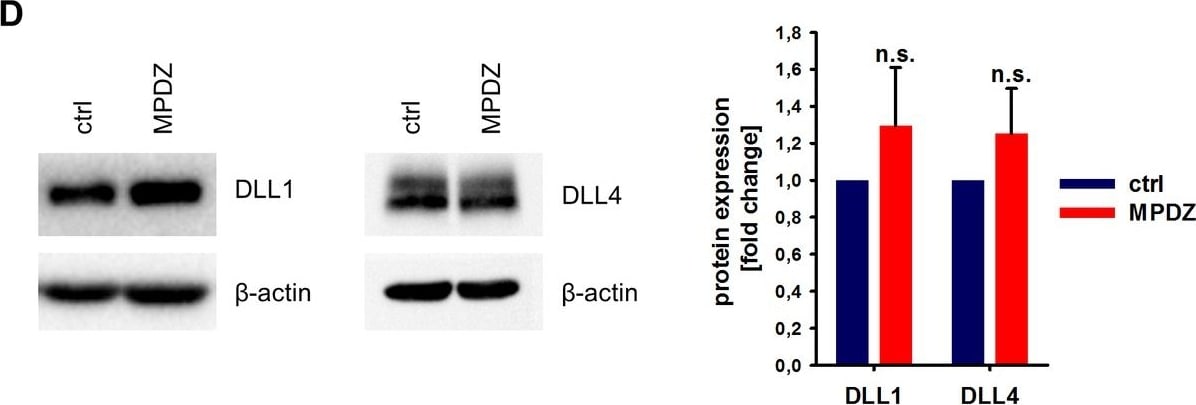
MPDZ promotes Notch signaling activity.(A) HUVECs were either transduced with lentivirus expressing GFP (sh-ctrl) or with lentivirus expressing shRNA against MPDZ (sh-MPDZ). Expression level of Notch target genes HEY1, HEY2 and HES1 were analyzed by qPCR 48 hr after transduction. Data are presented as mean ±SD. n ≥ 3; *, p<0.05; **, p<0.01; ***, p<0.001 unpaired Student’s t-test. (B) Cardiac endothelial cells were isolated from Mpdzfl/fl and Mpdz delta EC mice by magnetic beads bound with CD31 antibodies. Expression levels of Notch target genes Hey1 and Hey2 were analyzed by qPCR. Data are presented as mean ±SD. n = 3; *, p<0.05; ***, p<0.001 unpaired Student’s t-test. (C) HUVECs were either transduced with lentivirus expressing GFP (sh-ctrl) or with lentivirus expressing shRNA against MPDZ (sh-MPDZ). Expression levels of DLL1 and DLL4 were analyzed by immunoblotting 48 hr after transduction. beta -actin served as loading control. Data are presented as mean ±SD. n ≥ 3; n.s., not significant. (D) HUVECs were either transduced with adenovirus expressing GFP (ctrl) or with adenovirus expressing MPDZ. Expression levels of DLL1 and DLL4 were analyzed by immunoblotting 48 hr after transduction. beta -actin served as loading control. Data are presented as mean ±SD. n ≥ 3; n.s., not significant. (E) Lung endothelial cells were isolated from Mpdzfl/fl and Mpdz delta EC mice by CD31 magnetic beads. Protein amounts of Dll1 and Dll4 were analyzed by immunoblotting. beta -actin served as loading control. Data are presented as mean ±SD. n = 3; n.s., not significant.10.7554/eLife.32860.007Figure 2—source data 1.Source data of qantitative PCR analysis related to Figure 2A and B.Source data of qantitative PCR analysis related to Figure 2A and B. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/29620522), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse DLL4 by Immunocytochemistry/ Immunofluorescence
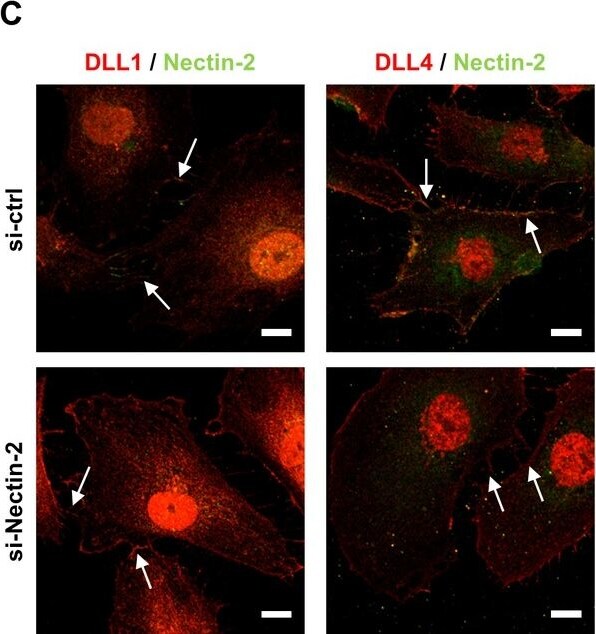
Mpdz does not affect cell cell junction assembly.(A, B) HUVECs were either transduced with lentivirus expressing GFP (sh-ctrl) or with lentivirus expressing shRNA against MPDZ (sh-MPDZ). Cells were cultured under sparse conditions (A) or confluent conditions (B). After PFA fixation cells were stained for DLL1 and Nectin-2 or DLL4 and Nectin-2 and counterstained with DAPI. Images were acquired with the confocal microscope LSM 700. Arrow indicates co-localization of DLL1/4 with Nectin-2 at the cell membrane. Arrow head indicates diminished co-localization at the cell membrane. Scale bar: 10 µm. (C) HUVECs were either transfected with control siRNA (si-ctrl) or with siRNA against Nectin-2 (si-Nectin-2). After PFA fixation cells were stained for DLL1 and Nectin-2 or DLL4 and Nectin-2. Images were acquired with the confocal microscope LSM 700. Arrow indicates localization of DLL1/4 at the cell membrane.Scale bar: 10 µm. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/29620522), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse DLL4 by Western Blot
MPDZ promotes Notch signaling activity.(A) HUVECs were either transduced with lentivirus expressing GFP (sh-ctrl) or with lentivirus expressing shRNA against MPDZ (sh-MPDZ). Expression level of Notch target genes HEY1, HEY2 and HES1 were analyzed by qPCR 48 hr after transduction. Data are presented as mean ±SD. n ≥ 3; *, p<0.05; **, p<0.01; ***, p<0.001 unpaired Student’s t-test. (B) Cardiac endothelial cells were isolated from Mpdzfl/fl and Mpdz delta EC mice by magnetic beads bound with CD31 antibodies. Expression levels of Notch target genes Hey1 and Hey2 were analyzed by qPCR. Data are presented as mean ±SD. n = 3; *, p<0.05; ***, p<0.001 unpaired Student’s t-test. (C) HUVECs were either transduced with lentivirus expressing GFP (sh-ctrl) or with lentivirus expressing shRNA against MPDZ (sh-MPDZ). Expression levels of DLL1 and DLL4 were analyzed by immunoblotting 48 hr after transduction. beta -actin served as loading control. Data are presented as mean ±SD. n ≥ 3; n.s., not significant. (D) HUVECs were either transduced with adenovirus expressing GFP (ctrl) or with adenovirus expressing MPDZ. Expression levels of DLL1 and DLL4 were analyzed by immunoblotting 48 hr after transduction. beta -actin served as loading control. Data are presented as mean ±SD. n ≥ 3; n.s., not significant. (E) Lung endothelial cells were isolated from Mpdzfl/fl and Mpdz delta EC mice by CD31 magnetic beads. Protein amounts of Dll1 and Dll4 were analyzed by immunoblotting. beta -actin served as loading control. Data are presented as mean ±SD. n = 3; n.s., not significant.10.7554/eLife.32860.007Figure 2—source data 1.Source data of qantitative PCR analysis related to Figure 2A and B.Source data of qantitative PCR analysis related to Figure 2A and B. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/29620522), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse DLL4 by Immunocytochemistry/ Immunofluorescence
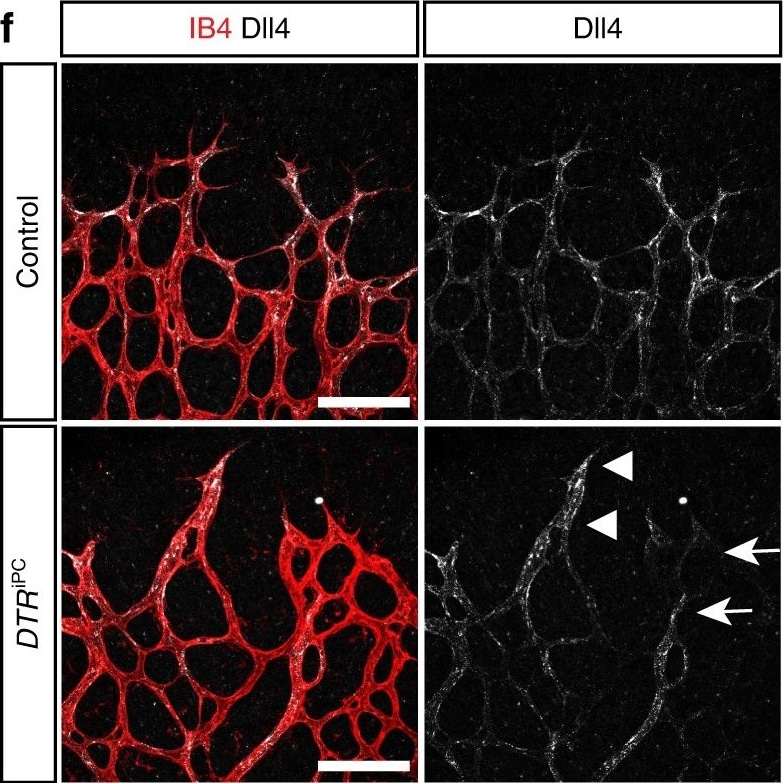
Endothelial changes after pericyte depletion. a–f Maximum intensity projection of confocal images from control and DTRiPC P6 retinas stained for IB4 (red) in combination with VEGF-A a, VEGFR2 b, VEGFR3 c, Tie2 d, Esm1 e, and Dll4 f (all in white), as indicated. Note local increase of VEGFR2, VEGFR3, and Esm1 (arrowheads in b, c, e) but not Tie2 or VEGF-A at the edge of the vessel plexus. Dll4 expression in DTRiPC sprouts is increased in some regions (arrowheads) but absent in others (arrows). Scale bar, 100 µm. g–j Quantitation of VEGF-A immunosignals area and intensity g, signal intensity for VEGFR2 h and VEGFR3 i and proportion of Esm1+ area with respect to vascular area j in the P6 control and DTRiPC angiogenic front. Error bars, s.e.m. p-values, Student’s t-test. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/29146905), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse DLL4 by Immunohistochemistry
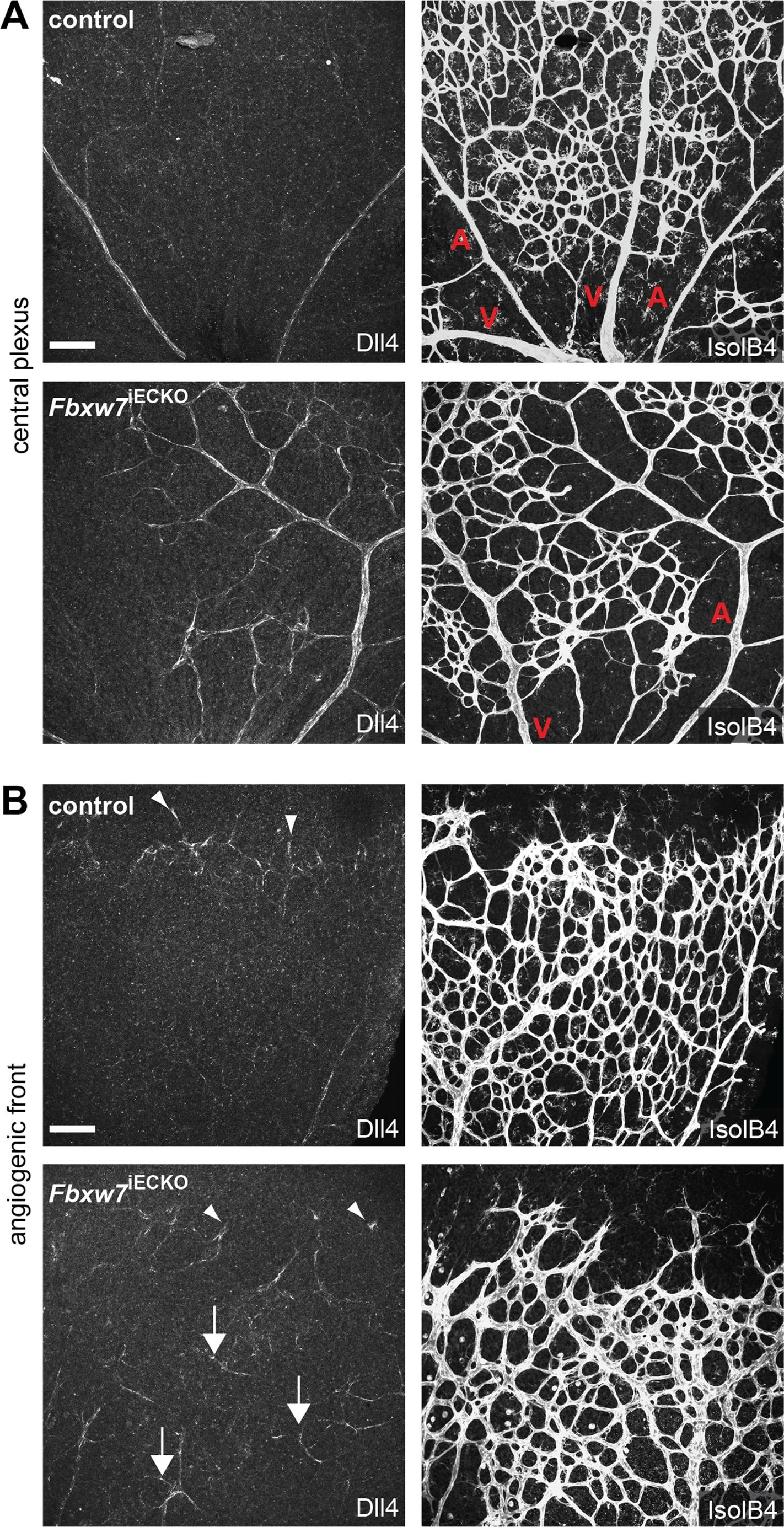
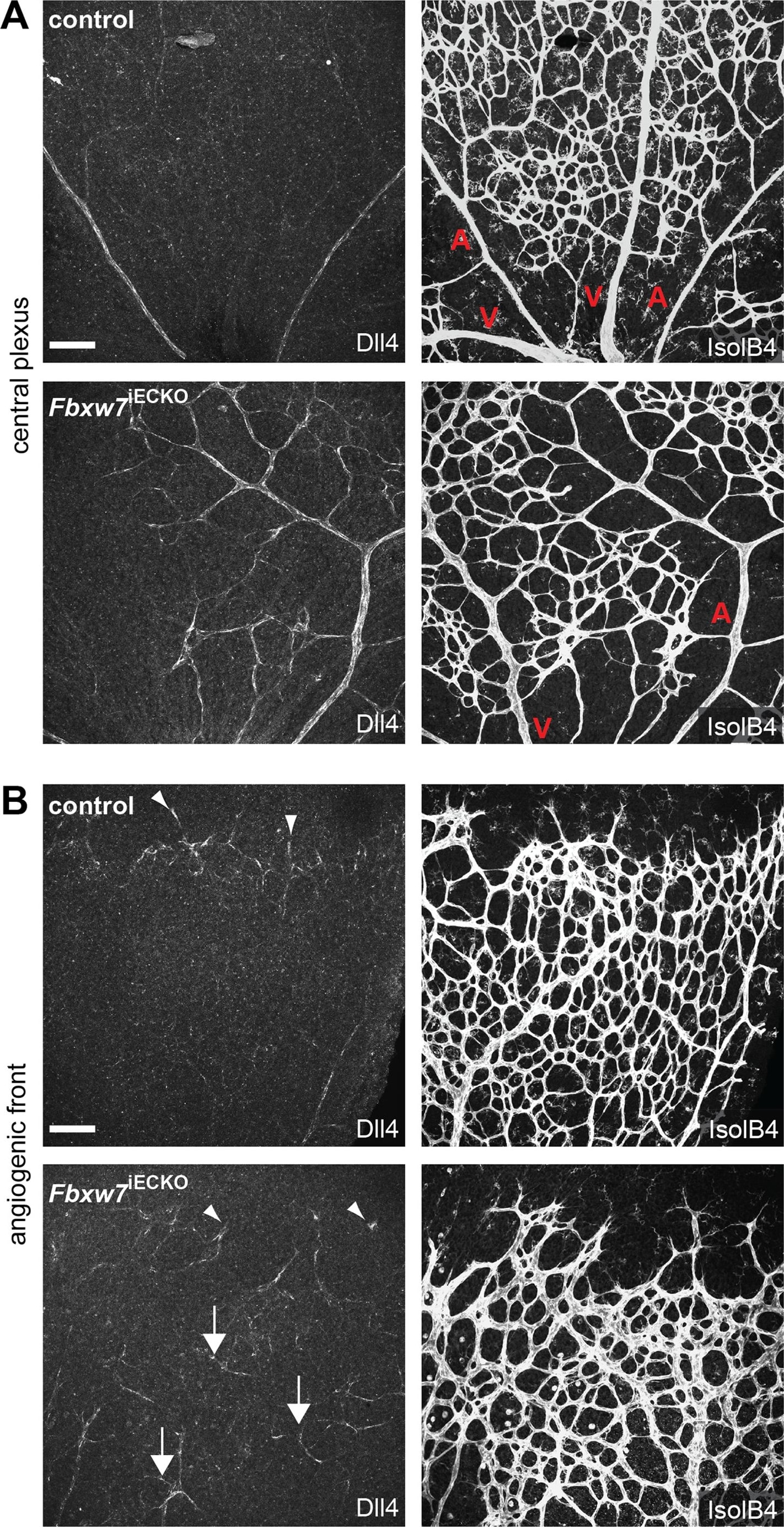
Targeting of Fbxw7 induced the upregulation of Dll4 expression.Anti-Dll4 and Isolectin B4 (IsolB4)-stained retinal whole-mounts of Fbxw7iECKO and littermate control retinas, as indicated (A, B). Arrowheads in (B) mark Dll4+ peripheral sprouts at the edge of the growing plexus, arrows indicate upregulated Dll4 in Fbxw7iECKO retinal capillaries. Quantitative analysis (with Volocity 5; n = 3 for each group) of image data confirmed elevated Dll4 levels (number of pixels) in the mutant endothelium (C). Likewise, quantitative RT-PCR analysis showed reduced Fbxw7 expression but upregulated Dll4 transcript levels in P6 Fbxw7iECKO lungs (D). Expression of the Cdh5 gene was used for normalization. Error bars indicate SEM. P values are indicated as ** (<0.001) and * (p<0.05). Scale bars are 100 µm. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/22848434), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse DLL4 by Immunohistochemistry
Targeting of Fbxw7 induced the upregulation of Dll4 expression.Anti-Dll4 and Isolectin B4 (IsolB4)-stained retinal whole-mounts of Fbxw7iECKO and littermate control retinas, as indicated (A, B). Arrowheads in (B) mark Dll4+ peripheral sprouts at the edge of the growing plexus, arrows indicate upregulated Dll4 in Fbxw7iECKO retinal capillaries. Quantitative analysis (with Volocity 5; n = 3 for each group) of image data confirmed elevated Dll4 levels (number of pixels) in the mutant endothelium (C). Likewise, quantitative RT-PCR analysis showed reduced Fbxw7 expression but upregulated Dll4 transcript levels in P6 Fbxw7iECKO lungs (D). Expression of the Cdh5 gene was used for normalization. Error bars indicate SEM. P values are indicated as ** (<0.001) and * (p<0.05). Scale bars are 100 µm. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/22848434), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Mouse DLL4 Antibody by Immunohistochemistry-Frozen
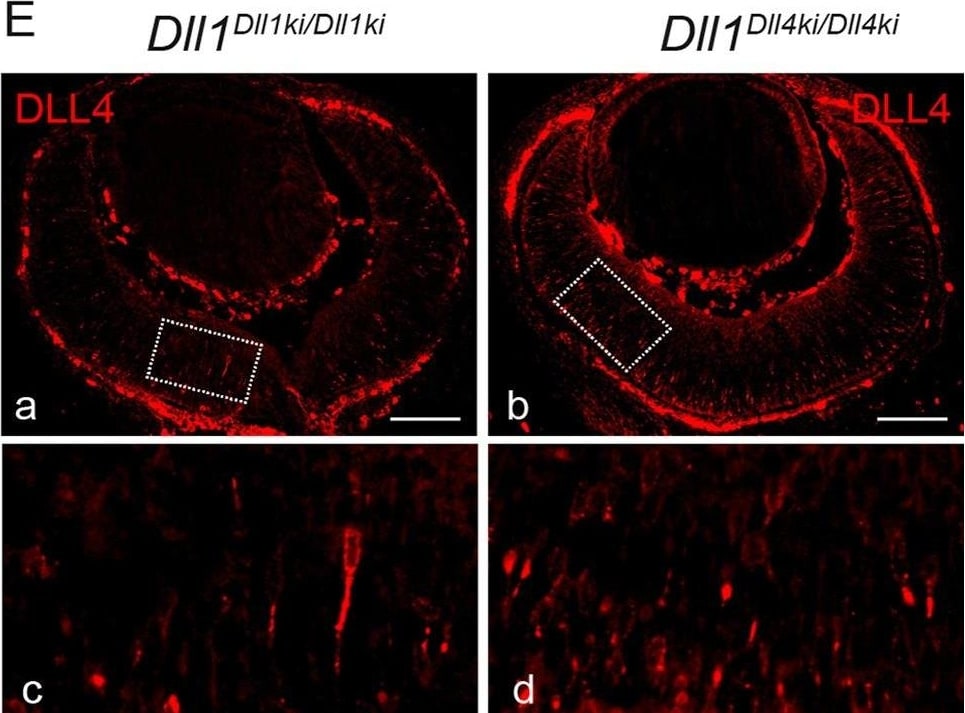
DLL4 expressed from the Dll1 locus rescues DLL1 loss-of-function in the retina.(A)Dll1 null mutant retinas show epithelial disruption with formation of polarised rosettes in which the apical markers N-Cadherin (NCad, a) and ZO-1 (ZO1, b) are abnormally present at the central lumen. Ectopic proliferating progenitors, labelled with PHH3 (b, arrowheads), are located close to the apical lumen of these rosettes. (B) In contrast, the neuroepithelium of homozygous Dll1Dll1ki and Dll1Dll4ki embryos is correctly organised without rosettes, and N-Cadherin shows the normal apical localisation close to the retinal pigmented epithelium (a,b). Mitotic progenitors (PHH3+) are only detected at the apical region of the neuroepithelium (a,b arrowheads). A normal stratification of CHX10+ progenitors and P27+ differentiating neurons is also observed (c,d). (C, D) E13.5 homozygous Dll1Dll1ki and Dll1Dll4ki retinas show no significant difference in the number of ISL1+ RGCs (C) and CRABP+ amacrine cells (D). Cells immunopositive for Islet-1 and Crabp were counted and related to the total number of cells in the retina (DAPI+). Percentages are shown as mean ± SEM; ns, not significant. (E) Expression of DLL4 in homozygous Dll1Dll1ki (a,c) and in homozygous Dll1Dll4ki (b,d) E13.5 retinas as detected by an anti-DLL4 antibody. (c) and (d) are magnifications of (a) and (b), respectively. Endogenous plus transgenic DLL4 is expressed in more cells in Dll1Dll4ki/Dll4ki as compared to endogenous DLL4 expression in Dll1Dll1ki/Dll1ki while signal strength is similar. Scale bars are 50 μm in (A, B) and 100 μm in (E). Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/26114479), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse DLL4 by Immunohistochemistry
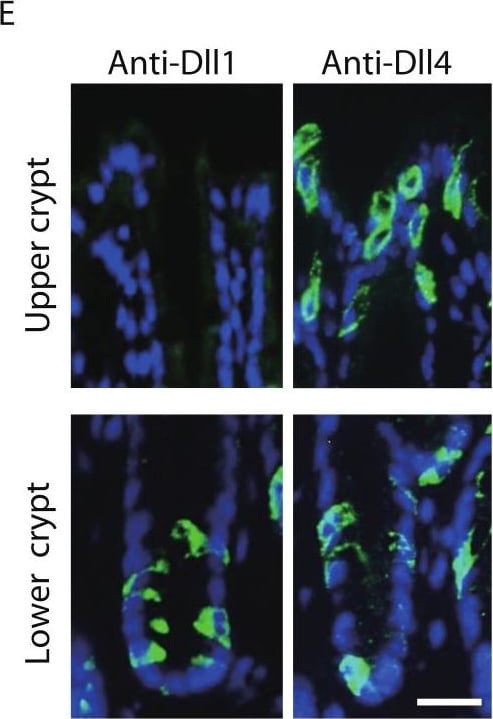
Dll1 and Dll4 are expressed in distinct patterns in the mice gastrointestinal epithelium.Immunohistochemistry of Dll1 and Dll4 were performed using mice small intestinal (A, B) and colonic tissues (D, E). Within the small intestinal epithelium, staining by anti-Dll1 antibody showed positive cells exclusively within the crypt (A, B), whereas staining by anti-Dll4 antibody showed positive cells both in the crypt and in the villi. Quantification of Dll1+ve and Dll4+ve intestinal epithelial cells (IECs) showed that Dll1+ve IECs are predominantly found within the crypt, whereas Dll4+ve IECs are found mostly in the villi (C). In the colonic epithelium, staining by anti-Dll1 antibody showed positive cells exclusively within the lower part of the crypt (D, E), whereas staining by anti-Dll4 antibody showed positive cells both in the lower- and upper part of the crypt, including the surface epithelium. Quantification of Dll1+ve and Dll4+ve IECs showed that Dll1+ve IECs are found exclusively within the lower half of the crypt, whereas Dll4+ve IECs are found both in the lower and upper region of the crypt at a comparable frequency (F). Negative staining of non-immunized isotype antibodies (Control Ab) confirmed the specific staining of Dll1 (A) and Dll4 (D). Scale bar represents 20 µm. Quantitative data are shown as mean ∓ SD of triplicate experiments (n = 3). ∗ indicates P < 0.05 as determined by Welch’s t-test. N.S. indicates not significant. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/24860699), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse DLL4 by Immunohistochemistry
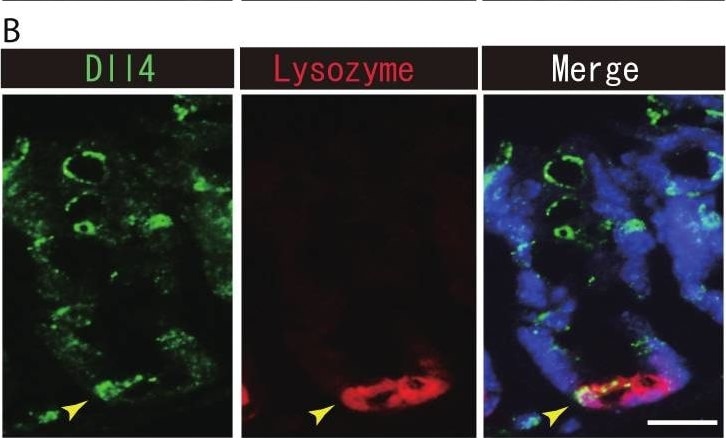
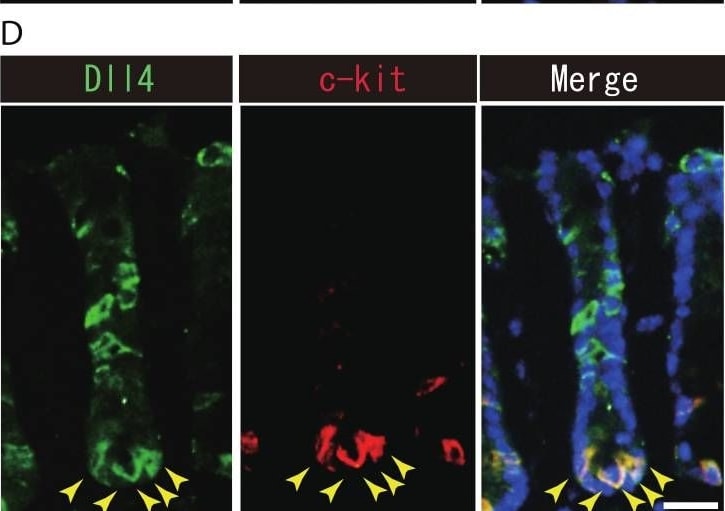
Dll1+ve or Dll4+ve IECs constitute the intestinal stem cell niche.Double immunostaining of Dll1 or Dll4 with a stem-cell niche cell marker, Lysozyme or c-kit, was performed in the mice small intestine and in the colon, respectively. (A) Immunostaining of Dll4 (green) with Lysozyme (red) shows that Dll4 is expressed in Paneth cells of the small intestine (yellow arrowhead). Scale bar represents 20 µm. (B) Double immunostaining of Dll1 (green) and c-kit (red) shows that Dll1+ve colonic IECs mostly co-express c-kit (yellow arrowhead). Also, double immunostaining of Dll4 (green) and c-kit (red) showed that a distinct population of Dll4+ve cells co-express c-kit (yellow arrowhead). Scale bar represents 20 µm. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/24860699), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse DLL4 by Immunohistochemistry
Dll1+ve or Dll4+ve IECs constitute the intestinal stem cell niche.Double immunostaining of Dll1 or Dll4 with a stem-cell niche cell marker, Lysozyme or c-kit, was performed in the mice small intestine and in the colon, respectively. (A) Immunostaining of Dll4 (green) with Lysozyme (red) shows that Dll4 is expressed in Paneth cells of the small intestine (yellow arrowhead). Scale bar represents 20 µm. (B) Double immunostaining of Dll1 (green) and c-kit (red) shows that Dll1+ve colonic IECs mostly co-express c-kit (yellow arrowhead). Also, double immunostaining of Dll4 (green) and c-kit (red) showed that a distinct population of Dll4+ve cells co-express c-kit (yellow arrowhead). Scale bar represents 20 µm. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/24860699), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse DLL4 by Western Blot
MPDZ promotes Notch signaling activity.(A) HUVECs were either transduced with lentivirus expressing GFP (sh-ctrl) or with lentivirus expressing shRNA against MPDZ (sh-MPDZ). Expression level of Notch target genes HEY1, HEY2 and HES1 were analyzed by qPCR 48 hr after transduction. Data are presented as mean ±SD. n ≥ 3; *, p<0.05; **, p<0.01; ***, p<0.001 unpaired Student’s t-test. (B) Cardiac endothelial cells were isolated from Mpdzfl/fl and Mpdz delta EC mice by magnetic beads bound with CD31 antibodies. Expression levels of Notch target genes Hey1 and Hey2 were analyzed by qPCR. Data are presented as mean ±SD. n = 3; *, p<0.05; ***, p<0.001 unpaired Student’s t-test. (C) HUVECs were either transduced with lentivirus expressing GFP (sh-ctrl) or with lentivirus expressing shRNA against MPDZ (sh-MPDZ). Expression levels of DLL1 and DLL4 were analyzed by immunoblotting 48 hr after transduction. beta -actin served as loading control. Data are presented as mean ±SD. n ≥ 3; n.s., not significant. (D) HUVECs were either transduced with adenovirus expressing GFP (ctrl) or with adenovirus expressing MPDZ. Expression levels of DLL1 and DLL4 were analyzed by immunoblotting 48 hr after transduction. beta -actin served as loading control. Data are presented as mean ±SD. n ≥ 3; n.s., not significant. (E) Lung endothelial cells were isolated from Mpdzfl/fl and Mpdz delta EC mice by CD31 magnetic beads. Protein amounts of Dll1 and Dll4 were analyzed by immunoblotting. beta -actin served as loading control. Data are presented as mean ±SD. n = 3; n.s., not significant.10.7554/eLife.32860.007Figure 2—source data 1.Source data of qantitative PCR analysis related to Figure 2A and B.Source data of qantitative PCR analysis related to Figure 2A and B. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/29620522), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Mouse DLL4 Antibody
Application
Recommended Usage
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed bEnd.3 mouse endothelioma cell line
Sample: Immersion fixed bEnd.3 mouse endothelioma cell line
Immunohistochemistry
5-15 µg/mL
Sample: Immersion fixed paraffin-embedded sections of mouse embryo (13 d.p.c.)
Sample: Immersion fixed paraffin-embedded sections of mouse embryo (13 d.p.c.)
Western Blot
2 µg/mL
Sample: bEnd.3 mouse endothelioma cell line
Sample: bEnd.3 mouse endothelioma cell line
Reviewed Applications
Read 1 review rated 3 using AF1389 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: DLL4
References
- Shutter, J.R. et al. (2000) Genes Dev. 14:1313.
- Iso, Tatsuya et al. (2002) Arterioscler. Thromb. Vasc. Biol. 23:543.
- Walker, L. et al. (2001) Stem Cells 19:543.
- Baron, M. (2002) Semin. Cell Dev. Biol. 14:113.
- Ikeuchi, T. and S.S. Sisodia (2003) J. Biol. Chem. 278:7751.
- Bland, C.E. et al. (2003) J. Biol. Chem. 278:13607.
Long Name
Delta-like 4
Alternate Names
delta 4, Delta 4 precursor, delta ligand 4, delta4, delta-like 4 (Drosophila), delta-like 4 homolog, delta-like 4 homolog (Drosophila), delta-like 4 protein, delta-like protein 4, Drosophila Delta homolog 4, hdelta2, MGC126344, notch ligand delta-2, notch ligand DLL4
Gene Symbol
DLL4
UniProt
Additional DLL4 Products
Product Documents for Mouse DLL4 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse DLL4 Antibody
For research use only
Related Research Areas
Citations for Mouse DLL4 Antibody
Customer Reviews for Mouse DLL4 Antibody (1)
3 out of 5
1 Customer Rating
Have you used Mouse DLL4 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
Application: ImmunocytochemistrySample Tested: See PMID 23091063Species: MouseVerified Customer | Posted 01/05/2015
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways