MCAM (Melanoma cell adhesion molecule; also CD146 and MUC18) is a 110‑120 kDa member of a small group of Ig‑superfamily molecules that includes CD239 and CD166. MCAM has also been reported at a molecular weight of approximately 150 kDa. In rodent, MCAM is reportedly expressed on neurons, endothelial cells, NK cells, neutrophils, mesenchymal stem cells and melanoma cells. MCAM appears to contribute to intercellular endothelial cell junctions, and possibly contributes to the migration of select cell types. Mature mouse MCAM is a 625 amino acid (aa) type I transmembrane glycoprotein. Its extracellular region is 540 aa in length (aa 24‑563). It contains two V‑type Ig‑like domains (aa 24‑244) followed by three C2‑type Ig‑like domains (aa 246‑512). One cytoplasmic region splice form shows a seven aa substitution for aa 600‑648. Unlike human, rodent MCAM does not undergo a splicing event that will generate a soluble isoform. Over aa 24‑563, mouse MCAM shares 90% and 74% aa identity with rat and human MCAM, respectively.


Key Product Details
Species Reactivity
Validated:
Mouse
Cited:
Mouse, Transgenic Mouse
Applications
Validated:
Western Blot, Immunocytochemistry, Simple Western
Cited:
Immunohistochemistry
Label
Unconjugated
Antibody Source
Polyclonal Sheep IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant mouse MCAM/CD146
Val24-Tyr563
Accession # Q8R2Y2
Val24-Tyr563
Accession # Q8R2Y2
Specificity
Detects mouse MCAM/CD146 in direct ELISAs and Western blots. In direct ELISAs, approximately 40% cross‑reactivity with recombinant rat MCAM is observed and approximately 10% cross-reactivity with recombinant human MCAM is observed.
Clonality
Polyclonal
Host
Sheep
Isotype
IgG
Scientific Data Images for Mouse MCAM/CD146 Antibody
Detection of Human and Mouse MCAM/CD146 by Western Blot.
Western blot shows lysates of Bowes human melanoma cell line and mouse embryo tissue. PVDF Membrane was probed with 1 µg/mL of Sheep Anti-Mouse MCAM/CD146 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF6106) followed by HRP-conjugated Anti-Sheep IgG Secondary Antibody (Catalog # HAF016). A specific band was detected for MCAM/CD146 at approximately 117 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 8.MCAM/CD146 in B16‑F1 Mouse Cell Line.
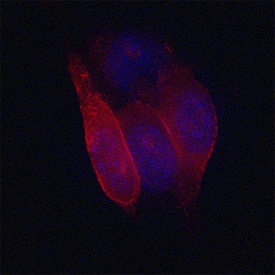
MCAM/CD146 was detected in immersion fixed B16-F1 mouse melanoma cell line using Sheep Anti-Mouse MCAM/CD146 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF6106) at 1.7 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Sheep IgG Secondary Antibody (red; Catalog # NL010) and counterstained with DAPI (blue). Specific staining was localized to plasma membranes. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Detection of Mouse MCAM/CD146 by Simple WesternTM.
Simple Western lane view shows lysates of mouse embryo tissue (15 d.p.c.), loaded at 0.2 mg/mL. A specific band was detected for MCAM/CD146 at approximately 138 kDa (as indicated) using 10 µg/mL of Sheep Anti-Mouse MCAM/CD146 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF6106) followed by 1:50 dilution of HRP-conjugated Anti-Sheep IgG Secondary Antibody (Catalog # HAF016). This experiment was conducted under reducing conditions and using the 12-230 kDa separation system.Detection of Mouse MCAM/CD146 by Immunocytochemistry/Immunofluorescence
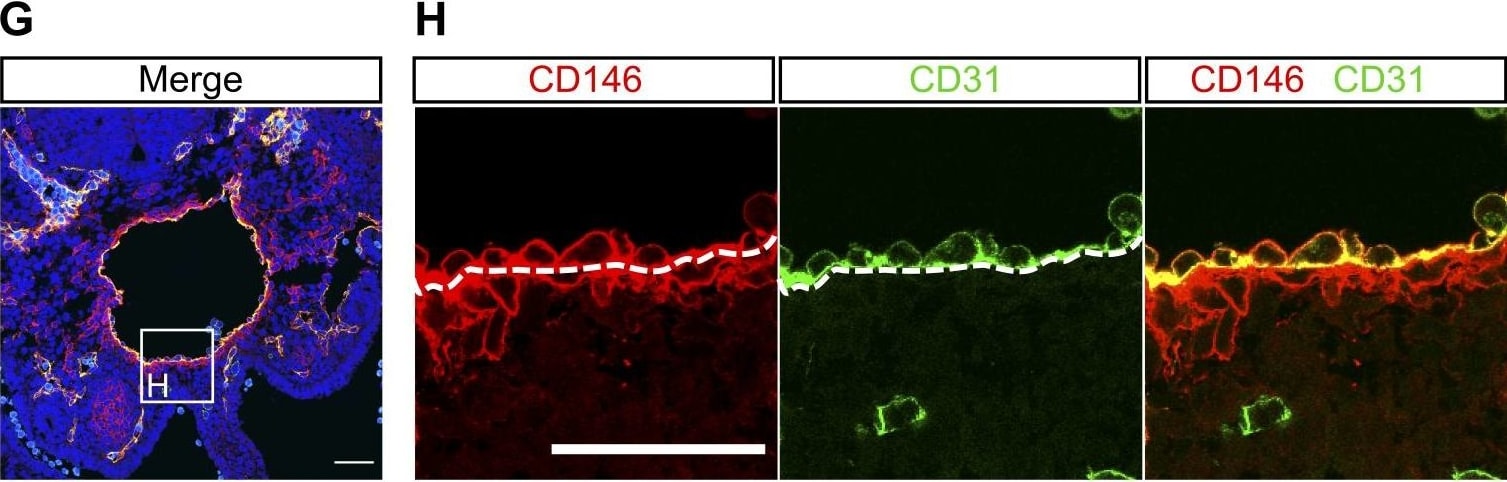
Perivascular cells and subaortic mesenchyme are the main source of BMPER within the AGM region. (A) Distribution of BMPER protein in a transverse section of E10.5 AGM measured by immunostaining. Green, CD31; magenta, BMPER; cyan, RUNX1; blue, DAPI. gut, hindgut; nc, notochord. Bar, 50 µm. (B) Bmper mRNA in transverse section of the E10.5 AGM region by in situ hybridization. Bar, 50 µm. (C) Quantification of the mean immunostaining signal intensity (mean gray values) of DAPI (blue) and BMPER (magenta) along a box (not depicted) drawn over the dorsal-ventral axis of the AGM region from A. Distance is from the notochord (dorsal), position 0, to the intersection with the gut and the AGM region (ventral), position 500. (D) Higher magnification of the highlighted region from A showing the aortic endothelium and perivascular population. Green, CD31; magenta, BMPER; cyan, RUNX1; blue, DAPI. Bar, 50 µm. (E) Higher magnification of the region highlighted in B showing Bmper mRNA around the lining of the aorta. Bar, 50 µm. (F) Expression level of Bmper relative to Tbp in each sorted population: Lin−VC−CD45+, representing hematopoietic cells; Lin−VC+CD45−, endothelial cells; Lin−VC−CD45−CD146+, putative perivascular cells; and Lin−VC−CD45−CD146−, remaining stroma. Expression was normalized to the Lin−VC−CD45−CD146+ population. Each population as percentage of Lin− cells indicated below. Sorting was performed twice, first from one pool of embryos from four to five litters and second from two pools of embryos from four to five litters. Error bars represent SD from the mean (n = 3). Significance calculated by t test: **, P = 0.0016; ***, perivascular versus stroma, P = 0.0006; perivascular versus hematopoietic, P = 0.0002. (G) The distribution of nonendothelial, CD146-positive cells and endothelial CD146-positive cells in transverse section of the E10.5 AGM region. Green, CD31; red, CD146; blue, DAPI. Bar, 50 µm. (H) Higher magnification view of the region highlighted in G showing CD31- and CD146-positive endothelial layer and the CD146-positive CD31-negative perivascular layer around the dorsal aorta. Green, CD31; red, CD146; blue, DAPI. Bar, 50 µm. (I) Expression level of Bmper relative to Tbp in each sorted population: Lin−VC+CD45−CD43−CD41lo proHSC, Lin−VC+CD43+CD41+ type I preHSC, and Lin−VC+CD45+ type II preHSC and Lin−VC−CD45−CD146+ putative perivascular cells. Each population as percentage of Lin− cells indicated below. Sorting was performed twice, from pools of two and six litters, respectively. Expression was normalized to the Lin−VC−CD45−CD146+ population. Error bars represent SD from the mean (n = 2). Significance calculated by t test: *, perivascular versus preHSCI, P = 0.04; perivascular versus preHSCII, P = 0.03. (J) Higher magnification view of highlighted region from A showing the localization of BMPER protein in the hematopoietic clusters of the E10.5 dorsal aorta in sections measured by immunostaining. Magenta, BMPER; green, CD31; cyan, RUNX1; blue, DAPI. Bar, 50 µm. (K and L) Higher magnification view of highlighted region from B (K) and from Fig. S3 C (L) showing Bmper mRNA in some but not all cells of the intra-aortic cluster by in situ hybridization. Bars, 50 µm. Image collected and cropped by CiteAb from the following publication (https://rupress.org/jem/article/214/12/3731/42286/A-molecular-roadmap-o…), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Mouse MCAM/CD146 Antibody
Application
Recommended Usage
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed B16‑F1 mouse melanoma cell line
Sample: Immersion fixed B16‑F1 mouse melanoma cell line
Simple Western
10 µg/mL
Sample: Mouse embryo tissue (15 d.p.c.)
Sample: Mouse embryo tissue (15 d.p.c.)
Western Blot
1 µg/mL
Sample: Bowes human melanoma cell line and mouse embryo tissue
Sample: Bowes human melanoma cell line and mouse embryo tissue
Reviewed Applications
Read 1 review rated 4 using AF6106 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: MCAM/CD146
Long Name
Melanoma Cell Adhesion Molecule
Alternate Names
CD146, L-Gicerin, MUC18
Gene Symbol
MCAM
UniProt
Additional MCAM/CD146 Products
Product Documents for Mouse MCAM/CD146 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse MCAM/CD146 Antibody
For research use only
Citations for Mouse MCAM/CD146 Antibody
Customer Reviews for Mouse MCAM/CD146 Antibody (1)
4 out of 5
1 Customer Rating
Have you used Mouse MCAM/CD146 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
Application: Immunohistochemistry-FrozenSample Tested: See PMID 22699974Species: MouseVerified Customer | Posted 01/08/2015
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways