Mouse Periostin, also known as OSF-2 (osteoblast-specific factor 2) is a 170 kDa, secreted, homodimeric protein that belongs to the periostin family of the FAS1 superfamily of molecules (1‑4). It is a TGF-beta inducible molecule that serves as both an adhesion molecule and tumor suppressor (2, 5, 6, 7). It is synthesized as a 838 amino acid (aa) precursor that contains a 23 aa signal sequence and an 815 aa mature region (2, 8). It is unknown if the molecule has any significant glycosylation (2). Based on human OSF-2, the homodimer is not disulfide-linked (3). The molecule consists of two distinct regions. The N-terminus contains an 55 aa EMI domain, while the C-terminus contains four, 130 aa fasciculin type 1 (or FAS1) domains. The EMI domain is cysteine-rich and shows a highly basic alpha -helix (9). Each FAS1 repeat exhibits a novel 7-stranded beta -wedge with a multiple alpha -helix fold (1, 8). Multiple alternate splice forms are known to exist C-terminal (aa 672‑812) to the four-fold FAS1 repeats. These mature molecules are 760, 761, 787 and 788 aa in length and show block deletions of 54 aa, 27 aa and/or 28 aa (10). The significance of the alternate splice forms is not clear. They do, however, appear to be temporally regulated (6). OSF-2 is known to bind to alpha v beta 3 and alpha v beta 5 integrins (3). It is synthesized by smooth muscle cells, fibroblasts and osteoblasts (2, 5, 7). Mature mouse OSF-2 shares 98%, 92% and 91% aa identity with rat, canine and human OSF‑2, respectively.
Mouse Periostin/OSF‑2 Isoform 2 Antibody
R&D Systems | Catalog # AF2955

Key Product Details
Species Reactivity
Validated:
Mouse
Cited:
Human, Mouse, Rat, Transgenic Mouse
Applications
Validated:
Western Blot, Neutralization, Immunocytochemistry
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Western Blot, Neutralization, Simple Western
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
S. frugiperda insect ovarian cell line Sf 21-derived recombinant mouse Periostin/OSF‑2
Asn24-Gln811
Accession # Q62009
Asn24-Gln811
Accession # Q62009
Specificity
Detects mouse Periostin/OSF‑2 in direct ELISAs and Western blots.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Mouse Periostin/OSF‑2 Isoform 2 Antibody
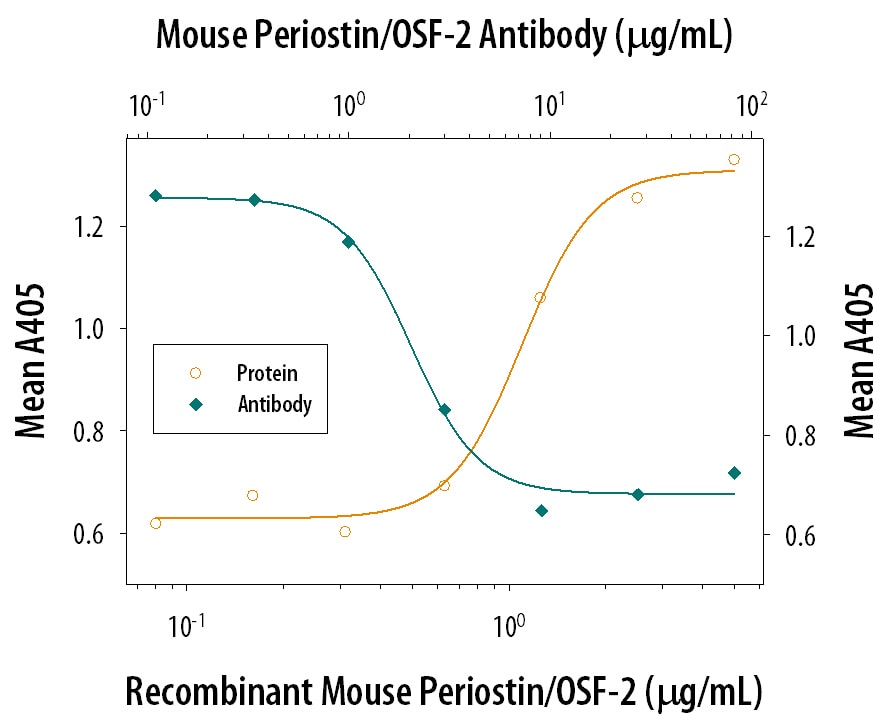
Cell Adhesion Mediated by Periostin/OSF‑2 and Neutral-ization by Mouse Periostin/ OSF‑2 Antibody.
Recombinant Mouse Periostin/OSF-2 (2955-F2), immobilized onto a microplate, supports the adhesion of the ATDC5 mouse chondrogenic cell line in a dose-dependent manner (orange line). Adhesion elicited by Recombinant Mouse Periostin/OSF-2 (5 µg/mL) is neutralized (green line) by increasing concentrations of Goat Anti-Mouse Periostin/ OSF-2 Isoform 2 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2955). The ND50 is typically 1-5 µg/mL.Periostin/OSF‑2 in Rat Mesenchymal Stem Cells.
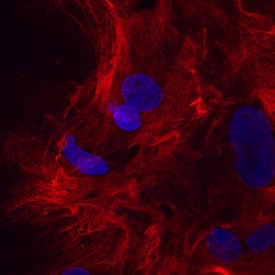
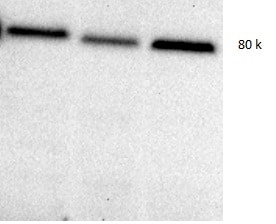
Periostin/OSF-2 was detected in immersion fixed rat mesenchymal stem cells differentiated to osteoblasts using Goat Anti-Mouse Periostin/OSF-2 Isoform 2 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2955) at 10 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; NL001) and counterstained with DAPI (blue). Specific staining was localized to cytoplasm. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Detection of Periostin/OSF-2 Isoform 2 by Western Blot
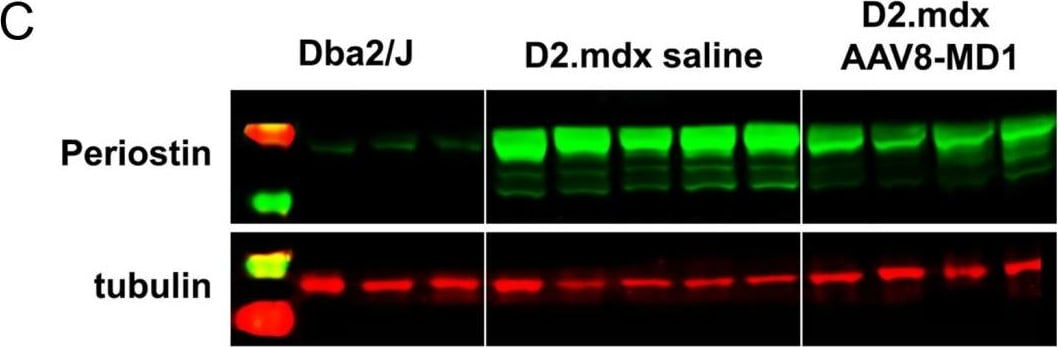
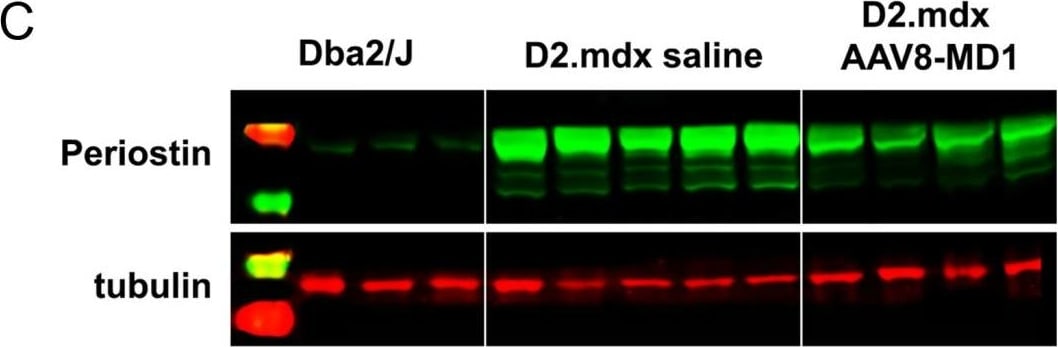
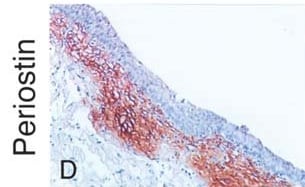
AAV8-MD1 delivery significantly reduces collagen VI and periostin expression in the diaphragm of D2.mdx mice. Male Dba2/J control and D2.mdx mice were treated with a single IV injection of either saline or AAV8-MD1 at 4 x 1012 vg/mouse at 6 weeks of age. (A) Representative images of collagen VI deposition in mouse diaphragms for each treatment group are shown as whole diaphragm sections (top row; scale bar: 500 µm) as well as a random close-up of each section (bottom row; scale bar: 100 µm). (B) Collagen VI-positive area within an entire diaphragm section was quantified and expressed as percentage of the total diaphragm section area. (C) Western blot analysis displaying periostin protein expression and (D) subsequent quantification of the level of periostin, normalized to alpha -tubulin, n = 4–5. In (B,D), data shown as ± SEM, n = 4–5. Statistical analysis was performed by one-way ANOVA followed by Tukey’s multiple comparisons test: p < 0.0001 (****), p < 0.001 (***), p < 0.001 (**), ns: not significant. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37175881), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Periostin/OSF-2 Isoform 2 by Immunohistochemistry
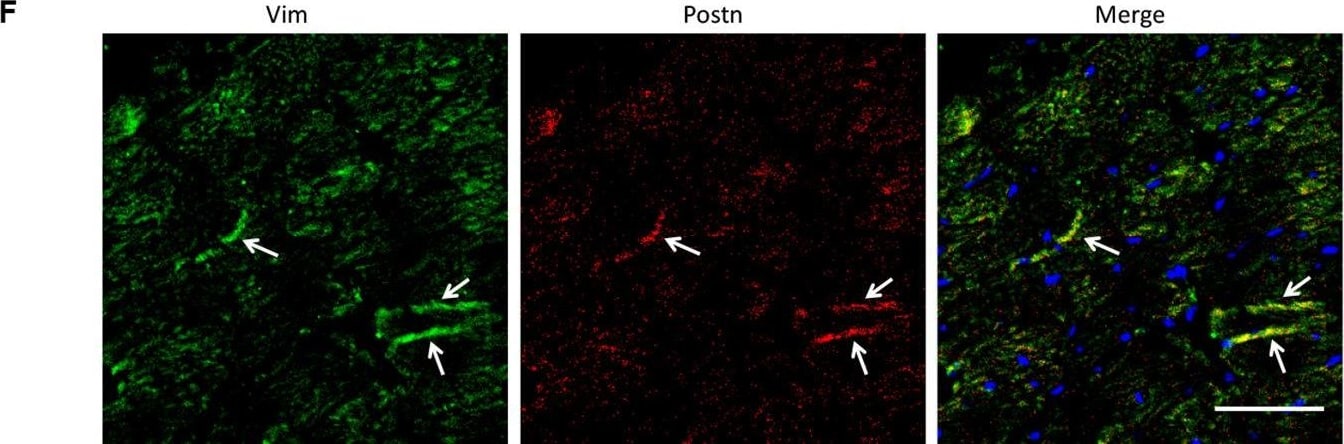
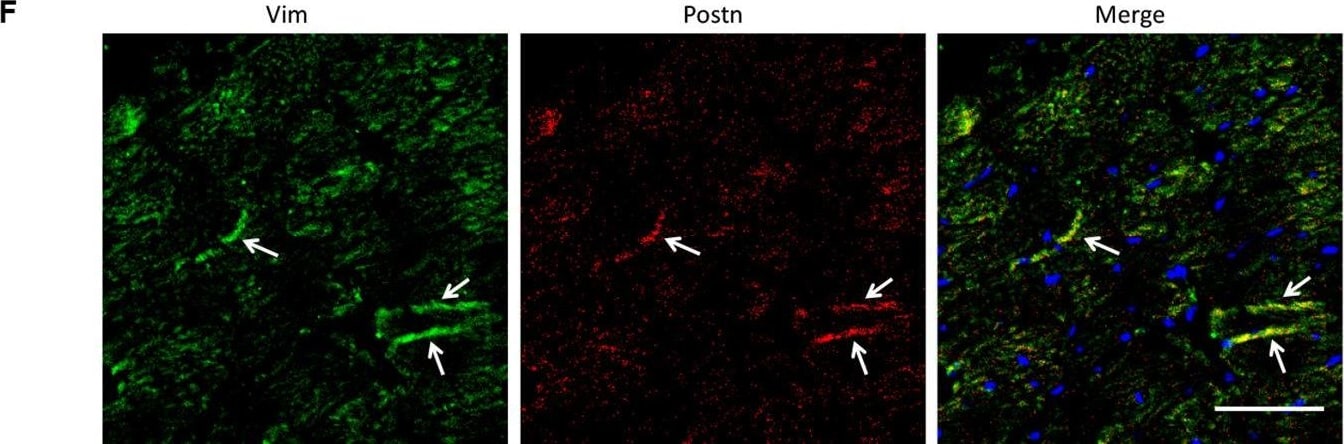
Analysis of the heterogeneity of the fibroblast subpopulations.(A) t-SNE plot of the 10 cardiac fibroblast subpopulations in the high-fat diet (HFD)/streptozotocin (STZ)-treated and control mice (n = 3428 fibroblasts from healthy control and n = 2988 fibroblasts from 6 diabetic mice). (B) Correlation heatmap of the gene-expression signatures of each fibroblast subpopulation. Color differences indicate subpopulations that were resolved through multiscale bootstrapping. (C) Heatmap showing the top 5 marker genes in each fibroblast subpopulation. The red color indicates high expression, while green color indicates low expression. (D) Heatmap showing the enriched Gene Ontology (GO) terms associated with cardiac fibrosis in each fibroblast population. (E, F) Representative immunofluorescence images of Hrc (E) and Postn (F) in mouse heart (n = 6 mice per group), scale bar = 100 µm. The details of the 10 transcriptionally distinct fibroblast subpopulations are listed in Supplementary file 15. The details of distinct signatures of each fibroblast subpopulation in the heart are listed in Supplementary file 16. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37010266), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Periostin/OSF-2 Isoform 2 by Western Blot
AAV8-MD1 delivery significantly reduces collagen VI and periostin expression in the diaphragm of D2.mdx mice. Male Dba2/J control and D2.mdx mice were treated with a single IV injection of either saline or AAV8-MD1 at 4 x 1012 vg/mouse at 6 weeks of age. (A) Representative images of collagen VI deposition in mouse diaphragms for each treatment group are shown as whole diaphragm sections (top row; scale bar: 500 µm) as well as a random close-up of each section (bottom row; scale bar: 100 µm). (B) Collagen VI-positive area within an entire diaphragm section was quantified and expressed as percentage of the total diaphragm section area. (C) Western blot analysis displaying periostin protein expression and (D) subsequent quantification of the level of periostin, normalized to alpha -tubulin, n = 4–5. In (B,D), data shown as ± SEM, n = 4–5. Statistical analysis was performed by one-way ANOVA followed by Tukey’s multiple comparisons test: p < 0.0001 (****), p < 0.001 (***), p < 0.001 (**), ns: not significant. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37175881), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Periostin/OSF-2 Isoform 2 by Immunohistochemistry
Analysis of the heterogeneity of the fibroblast subpopulations.(A) t-SNE plot of the 10 cardiac fibroblast subpopulations in the high-fat diet (HFD)/streptozotocin (STZ)-treated and control mice (n = 3428 fibroblasts from healthy control and n = 2988 fibroblasts from 6 diabetic mice). (B) Correlation heatmap of the gene-expression signatures of each fibroblast subpopulation. Color differences indicate subpopulations that were resolved through multiscale bootstrapping. (C) Heatmap showing the top 5 marker genes in each fibroblast subpopulation. The red color indicates high expression, while green color indicates low expression. (D) Heatmap showing the enriched Gene Ontology (GO) terms associated with cardiac fibrosis in each fibroblast population. (E, F) Representative immunofluorescence images of Hrc (E) and Postn (F) in mouse heart (n = 6 mice per group), scale bar = 100 µm. The details of the 10 transcriptionally distinct fibroblast subpopulations are listed in Supplementary file 15. The details of distinct signatures of each fibroblast subpopulation in the heart are listed in Supplementary file 16. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37010266), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Periostin/OSF-2 Isoform 2 by Western Blot
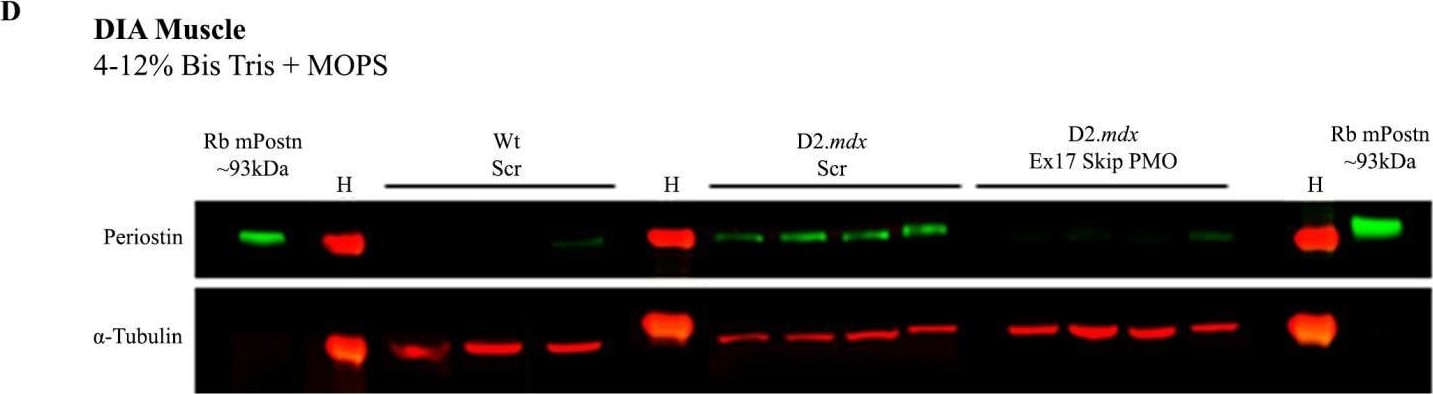
vivoPMO significantly reduced mPostn exon 17 expression and induced beneficial effect on muscles. RT-qPCR analysis of e17+ (A) and total (B) Postn mRNA expression represented as fold change compared to wild type (N = 4–6). (C) Full-length protein expression assessment using Western blot (D) analysis. (E) DIA hydroxyproline analysis (indicative of collagen level). (F) Forelimb grip strength was measured as the average of 5 discrete measurements of maximum force normalised to body weight. Outliers were identified via the ROUT method and omitted from the statistical analysis. The data were analysed by one-way ANOVA with Tukey post hoc tests and significance was recorded as * = p < 0.05; ** = p < 0.01; **** = p < 0.0001. Error bars = +/−SD; qPCR was performed in triplicate. Legend: ‘wt’ = Dba.2J wild type; ‘mdx’ = D2.mdx + Scramble vivoPMO; ‘mdx + pmo’ = D2.mdx + Postn exon 17 skipping vivoPMO. Image collected and cropped by CiteAb from the following open publication (https://www.mdpi.com/1422-0067/25/11/6113), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Periostin/OSF-2 Isoform 2 by Immunohistochemistry
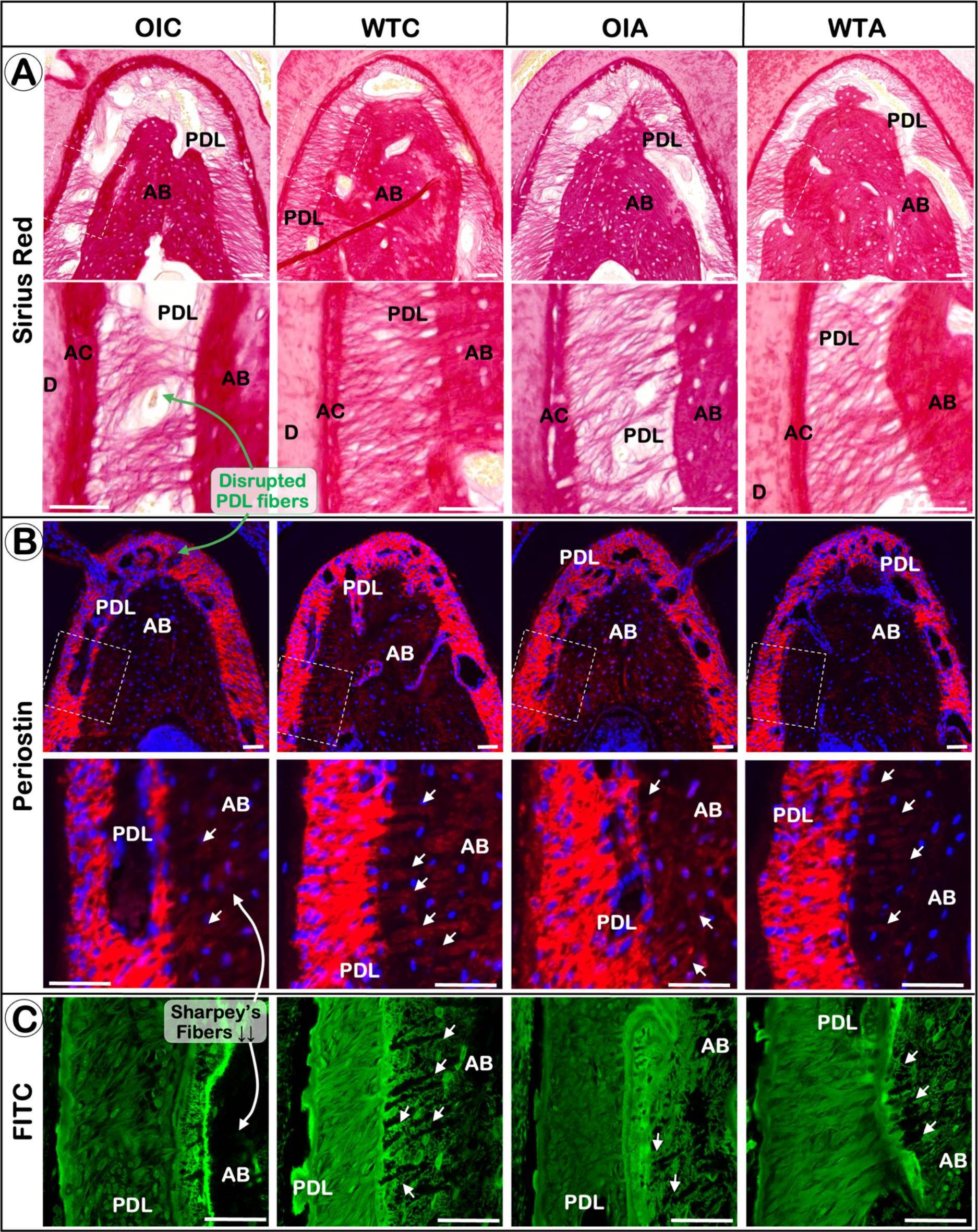
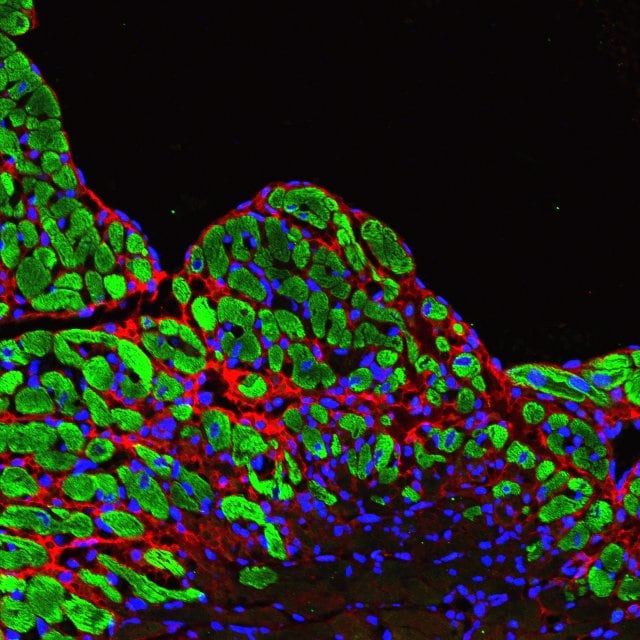
Oim−/− mice display a severe PDL phenotype which can be partially improved by alendronate treatment. Representative images of Sirius red staining (A), immunofluorescent staining of periostin (B) and FITC staining (C). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/39747677), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Mouse Periostin/OSF‑2 Isoform 2 Antibody
Application
Recommended Usage
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed rat mesenchymal stem cells differentiated to osteoblasts
Sample: Immersion fixed rat mesenchymal stem cells differentiated to osteoblasts
Western Blot
0.1 µg/mL
Sample: Recombinant Mouse Periostin/OSF‑2 (Catalog # 2955-F2)
Sample: Recombinant Mouse Periostin/OSF‑2 (Catalog # 2955-F2)
Neutralization
Measured by its ability to neutralize Periostin/OSF‑2-mediated adhesion of the ATDC5 mouse chondrogenic cell line. The Neutralization Dose (ND50) is typically 1-5 µg/mL in the presence of 5 µg/mL Recombinant Mouse Periostin/OSF‑2.
Reviewed Applications
Read 7 reviews rated 4.6 using AF2955 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Periostin/OSF-2
References
- Clout, N.J. and D. Tisi (2003) Structure 11:197.
- Horiuchi, K. et al. (1999) J. Bone Miner. Res. 14:1239.
- Gillan, L. et al. (2002) Cancer Res. 62:5358.
- Litvin, J. et al. (2005) Anat. Rec. A Discov. Mol. Cell. Evol. Biol. 287A:1205.
- Lindner, V. et al. (2005) Arterioscler. Thromb. Vasc. Biol. 25:77.
- Kruzynska-Frejtag, A. et al. (2004) Dev. Dyn. 229:857.
- Yoshioka, N. et al. (2002) Exp. Cell Res. 279:91.
- Takeshita, S. et al. (1993) Biochem. J. 294:271.
- Callebaut, I. et al. (2003) Biochem. Biophys. Res. Commun. 300:619.
- Swiss-Prot Accession # Q62009.
Long Name
Osteoblast Specific Factor 2
Alternate Names
Fasciclin I-like, OSF-2, OSF2, POSTN, TRIF52
Gene Symbol
POSTN
UniProt
Additional Periostin/OSF-2 Products
Product Documents for Mouse Periostin/OSF‑2 Isoform 2 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse Periostin/OSF‑2 Isoform 2 Antibody
For research use only
Related Research Areas
Citations for Mouse Periostin/OSF‑2 Isoform 2 Antibody
Customer Reviews for Mouse Periostin/OSF‑2 Isoform 2 Antibody (7)
4.6 out of 5
7 Customer Ratings
Have you used Mouse Periostin/OSF‑2 Isoform 2 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
5 of
7 reviews
Showing All
Filter By:
-
Application: Western BlotSample Tested: Heart tissueSpecies: MouseVerified Customer | Posted 07/19/2021
-
Application: ImmunohistochemistrySample Tested: Bladder tissueSpecies: RatVerified Customer | Posted 06/29/2021
-
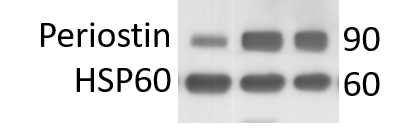
Application: Western BlotSample Tested: Adult heartSpecies: MouseVerified Customer | Posted 05/10/20203 mice hearts extractions expressing periostin after MI. HSP60 used as loading marker.
-
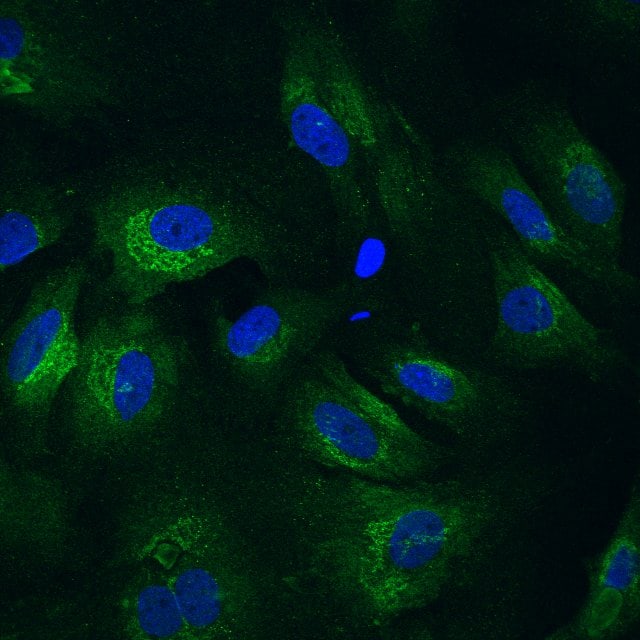
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: primary cardiac fibroblastsSpecies: RatVerified Customer | Posted 05/10/2020primary rat cardiac fiboblasts. periostin in green and DAPI in blue
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: Adult heartSpecies: MouseVerified Customer | Posted 05/10/2020mouse heart post MI fixed in PFA stained to periostin in red, troponin I in green and DAPI in blue
-
Application: Western BlotSample Tested: Tissue LysatesSpecies: MouseVerified Customer | Posted 06/17/2019
-
Application: Immunohistochemistry-ParaffinSample Tested: See PMID 23729679Species: MouseVerified Customer | Posted 01/07/2015
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...