Prolactin (PRL) is a neuroendocrine pituitary hormone. Prolactin is synthesized by the anterior pituitary, placenta, brain, uterus, dermal fibroblasts, decidua, B cells, T cells, NK cells and breast cancer cells. Originally characterized as a lactogenic hormone, further studies have demonstrated broader roles in breast cancer development, regulation of reproductive function, and immunoregulation. In the immune system, Prolactin has been shown to be secreted by human PBMC and to act as a proliferative growth factor. Additionally, Prolactin treatment of human PBMC has been shown to enhance IFN-gamma production. In the breast, Prolactin-induced morphogenesis of the mammary cells is mediated through IGF-2, which in turn upregulates cyclin D1. Prolactin has several molecular forms. The predominant form is a monomer; the non-glycosylated form is 23 kDa and the glycosylated form is 25 kDa. Glycosylated Prolactin is removed from the circulation faster and has been reported to have lower biological potency. Mouse Prolactin cDNA encodes a 228 amino acid (aa) residue protein with a putative 31 aa residue signal peptide. The Prolactin receptor is a transmembrane type I glycoprotein that belongs to the cytokine hematopoietic receptor family. B cells, T cells, macrophages, NK cells, monocytes, CD34+ progenitor cells, neutrophils, mammary gland, liver, kidney, adrenals, ovaries, testis, prostrate, seminal vesicles, and hypothalamus have all been shown to express the Prolactin receptor. Three forms of the receptor, generated by differential splicing, have been identified. These isoforms differ in the length of their cytoplasmic domains. It is believed that the short cytoplasmic form is non-functional. Prolactin signal transduction involves the JAK/STAT families and Src kinase family (1‑9).
Mouse/Rat Prolactin Antibody
R&D Systems | Catalog # AF1445

Key Product Details
Species Reactivity
Validated:
Mouse, Rat
Cited:
Mouse, Rat, Lizard - Gecko
Applications
Validated:
Immunohistochemistry, Western Blot, ELISA Capture (Matched Antibody Pair), Neutralization, Simple Western
Cited:
Immunohistochemistry-Paraffin, Immunohistochemistry-Frozen, Neutralization, Immunofluorescence, Immunocytochemistry, Bioassay
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant mouse Prolactin
Leu32-Cys228
Accession # NP_035294
Leu32-Cys228
Accession # NP_035294
Specificity
Detects mouse and rat Prolactin in Western blots. In sandwich immunoassays, less than 1% cross-reactivity with recombinant human Prolactin is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Mouse/Rat Prolactin Antibody
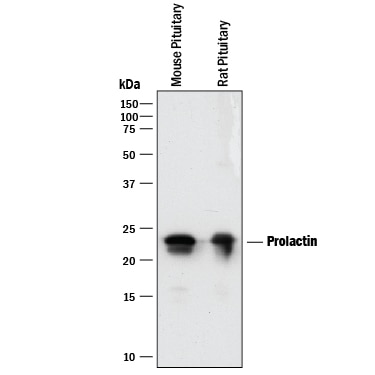
Detection of Mouse and Rat Prolactin by Western Blot.
Western blot shows lysates of mouse pituitary tissue and rat pituitary tissue. PVDF membrane was probed with 0.25 µg/mL of Goat Anti-Mouse/Rat Prolactin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1445) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (HAF017). A specific band was detected for Prolactin at approximately 23 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Cell Proliferation Induced by Prolactin and Neutralization by Mouse Prolactin Antibody.
Recombinant Mouse Prolactin (1445-PL) stimulates proliferation in the Nb2-11 rat lymphoma cell line in a dose-dependent manner (orange line). Proliferation elicited by Recombinant Mouse Prolactin (10 ng/mL) is neutralized (green line) by increasing concentrations of Goat Anti-Mouse/Rat Prolactin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1445). The ND50 is typically 0.25-1.0 µg/mL.Detection of Prolactin in Mouse Pituitary.
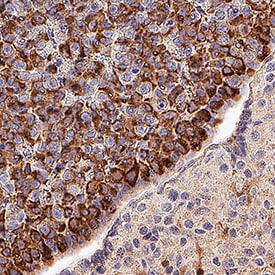
Prolactin was detected in immersion fixed paraffin-embedded sections of Mouse Pituitary using Goat Anti-Mouse/Rat Prolactin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1445) at 1 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Goat IgG VisUCyte™ HRP Polymer Antibody (brown; Catalog # VC004) and counterstained with hematoxylin (blue). Specific staining was localized to cytoplasm in lactotroph cells. View our protocol for IHC Staining with VisUCyte HRP Polymer Detection Reagents.Detection of Mouse and Rat Prolactin by Simple WesternTM.
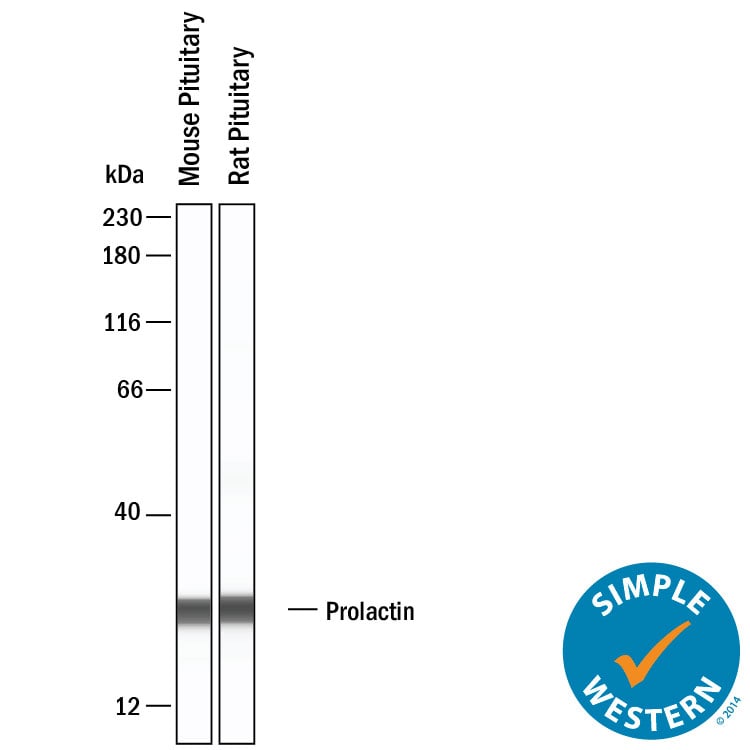
Simple Western shows lysates of mouse pituitary tissue and rat pituitary tissue, loaded at 0.2 mg/ml. A specific band was detected for Prolactin at approximately 26 kDa (as indicated) using 2.5 µg/mL of Goat Anti-Mouse/Rat Prolactin Antigen Affinity-purified Polyclonal Antibody (Catalog # af1445). This experiment was conducted under reducing conditions and using the 12-230kDa separation system.Detection of Prolactin by Western Blot
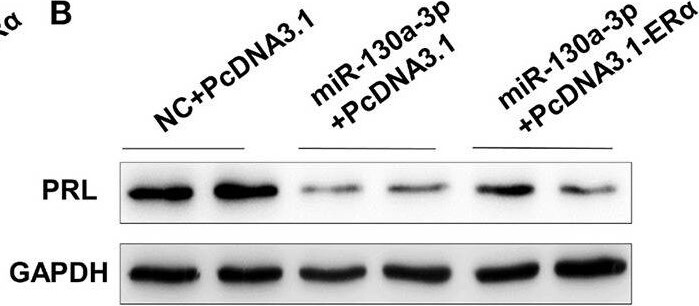
Overexpression of ER alpha rescues miR-130a-3p-inhibited expression of PRL. The GH3 cells were transfected with the miR-130a-3p mimic and the ER alpha overexpressing vector. The rescue efficiency of ER alpha in the GH3 cells was confirmed by western blotting. (A) The protein expression level of ER alpha was detected by western blotting. GAPDH was used as loading control. (B) The protein expression level of PRL was detected by western blotting. GAPDH was used as loading control. (C) Quantitation of ER alpha protein level. Data are presented as mean ± S.E.M of n = 4 samples per group (P < 0.05 by ANOVA). (D) Quantitation of PRL protein level. Data are presented as mean ± S.E.M of n = 4 samples per group (P < 0.05 by ANOVA). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32194503), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Prolactin by Western Blot
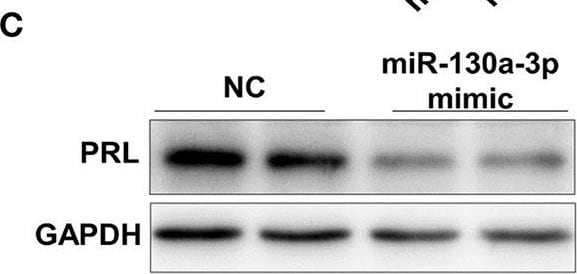
MiR-130a-3p overexpression reduces PRL expression in GH3 cells. GH3 cells were transfected with miR-130a-3p mimic and NC and then the expressions of miR-130a-3p and PRL were analyzed. (A) The expression level of miR-130a-3p was detected by quantitative real-time PCR (qRT-PCR). U6 snRNA was used to normalize the miRNA expression. Data are presented as mean ± S.E.M of n = 6 samples per group (*P < 0.05 by t-test). (B) The expression level of PRL mRNA was detected by qRT-PCR. GAPDH was used to normalize each gene expression. Data are presented as mean ± S.E.M of n = 6 samples per group (*P < 0.05 by t-test). (C) The protein level of PRL in GH3 cells was analyzed by western blotting. GAPDH was used as loading control. (D) Quantitation of the PRL protein level. Data are presented as mean ± S.E.M of n = 4 samples per group (*P < 0.05 by t-test). (E) The protein level of PRL was detected by ICC after GH3 cells transfected with miR-130a-3p mimic and NC. Scale bar, 50 μM. **P < 0.01. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32194503), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Prolactin by Western Blot
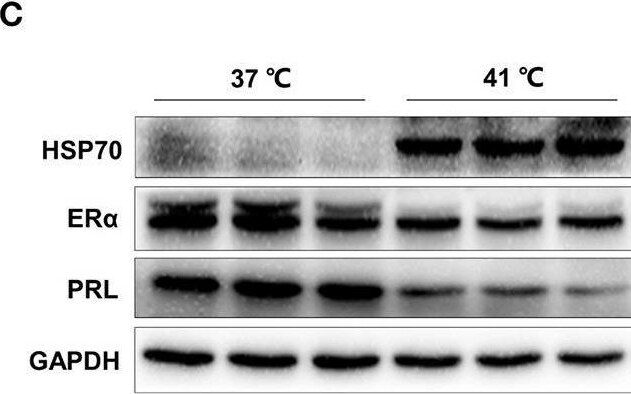
Heat stress increases miR-130a-3p expression, reduces PRL and ER alpha expressions in GH3 cells. GH3 cells were separately cultured in 37 or 41°C for 24 h, after that, miR-130a-3p, PRL, and ER alpha expressions were analyzed. (A) The expression level of miR-130a-3p was detected by quantitative real-time PCR (qRT-PCR). U6 snRNA was used to normalize the miRNA expression. Data are presented as mean ± S.E.M of n = 4 samples per group (*P < 0.05 by t-test). (B) The expression levels of HSP70, ER alpha, and PRL mRNAs in GH3 cells were detected by qRT-PCR. GAPDH was used to normalize each gene expression. Data are presented as mean ± S.E.M of n = 4 samples per group (*P < 0.05, ns, not significant by t-test). (C) The protein levels of HSP70, ER alpha, and PRL in GH3 cells were analyzed by western blotting. GAPDH was used as loading control. (D) Quantitation of HSP70, ER alpha, and PRL protein levels. Data are presented as mean ± S.E.M of n = 4 samples per group (*P < 0.05 by t-test). **P < 0.01. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32194503), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Prolactin by Western Blot
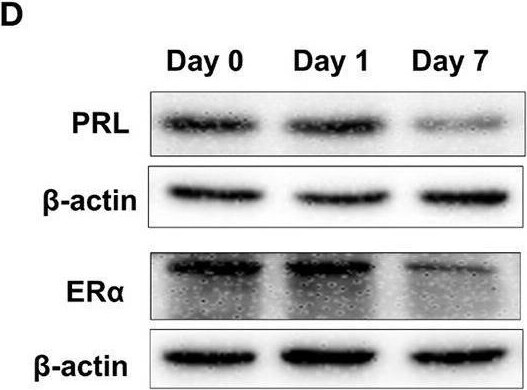
Heat stress reduces PRL and ER alpha expressions as well as increases miR-130a-3p expression in the pituitary gland. The mice of treated groups were placed in 40°C for 2 h each time, and the stimulus, respectively lasted 1 and 7 days. The mice in control group were fed as normal in 25°C. Then the expressions of PRL and ER alpha in the pituitary gland were analyzed. (A–B) The mRNA levels of PRL (A) and ER alpha (B) in the pituitary gland of three groups were analyzed by quantitative real-time PCR (qRT-PCR). GAPDH was used to normalize each gene expression. Data are presented as mean ± S.E.M of n = 5 animals per group. Bars that share different letter are significantly different (P < 0.05 by ANOVA). (C) The serum PRL concentration was detected by Elisa assay. Data are presented as mean ± S.E.M of n = 5 animals per group. Bars that share different letter are significantly different (P < 0.05 by ANOVA). (D) The protein levels of PRL and ER alpha in the pituitary gland were analyzed by western blotting. beta -actin was used as loading control. (E) Quantitation of PRL and ER alpha protein levels. Data are presented as mean ± SD of n = 3 animals per group. Bars that share different letter are significantly different (P < 0.05 by ANOVA). The expression levels of miR-130a-3p (F), miR-130b-3p (G), miR-301a-3p (H), and miR-301b-3p (I) were detected by qRT-PCR. U6 snRNA was used to normalize each miRNA expression. Data are presented as mean ± S.E.M of n = 5 animals per group. Bars that share different letter are significantly different (P < 0.05 by ANOVA). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32194503), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Mouse/Rat Prolactin Antibody
Application
Recommended Usage
Immunohistochemistry
1-15 µg/mL
Sample: Immersion fixed paraffin-embedded sections of Mouse Pituitary
Sample: Immersion fixed paraffin-embedded sections of Mouse Pituitary
Simple Western
2.5 µg/mL
Sample: Mouse pituitary tissue and rat pituitary tissue
Sample: Mouse pituitary tissue and rat pituitary tissue
Western Blot
0.25 µg/mL
Sample: Mouse pituitary tissue and rat pituitary tissue
Sample: Mouse pituitary tissue and rat pituitary tissue
Neutralization
Measured by its ability to neutralize Prolactin-induced proliferation in the Nb2‑11 rat lymphoma cell line. Gout, P. W. et al. (1980) Cancer Research 40:2433. The Neutralization Dose (ND50) is typically 0.25-1.0 µg/mL in the presence of 10 ng/mL Recombinant Mouse Prolactin.
Mouse Prolactin Sandwich Immunoassay
Please Note: Optimal dilutions of this antibody should be experimentally determined.
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. See Certificate of Analysis for details.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Prolactin
References
- Freeman, M. et al. (2000) Physiological Reviews 80:1523.
- Ben-Johnson, N. et al. (1996) Endoc. Rev. 17:639.
- Cesario, T. et al. (1994) Proc. Soc. Exp. Biol. Med. 205:89.
- Price, A.E. et al. (1995) Endoc. 136:4827.
- Hoffmann, T. et al. (1993) J. Endoc. Invest. 16:807.
- Cole, E. et al. (1991) Endoc. 129:2639.
- Lewis, U. et al. (1985) Endoc. 116:359.
- Matalk, K. (2003) Cytokine 21:187.
- Brisken, C. et al. (2002) Dev. Cell 3:877.
Alternate Names
PRL
Gene Symbol
PRL
UniProt
Additional Prolactin Products
Product Documents for Mouse/Rat Prolactin Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse/Rat Prolactin Antibody
For research use only
Related Research Areas
Citations for Mouse/Rat Prolactin Antibody
Customer Reviews for Mouse/Rat Prolactin Antibody
There are currently no reviews for this product. Be the first to review Mouse/Rat Prolactin Antibody and earn rewards!
Have you used Mouse/Rat Prolactin Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...