VEGFR1 (vascular endothelial growth factor receptor 1), also called Flt-1 (Fms-like tyrosine kinase), is a 180 kDa type I transmembrane glycoprotein in the class III subfamily of receptor tyrosine kinases (RTKs) (1, 2). While family members VEGFR1, VEGFR2/KDR/Flk-1 and VEGFR3/Flt-4 are all mainly expressed on endothelial cells and play central roles in vasculogenesis, angiogenesis, and lymphangiogenesis, only VEGFR1 is expressed on macrophages, and mainly plays inhibitory roles (1-3). VEGFR1 expression is also reported on osteoblasts, placental trophoblasts, renal mesangial cells, and some hematopoietic stem cells (1, 2). Like other class III RTKs, human VEGFR1 contains a signal peptide (aa 1-22), an extracellular domain (ECD aa 27-758) with seven Ig-like repeats, a transmembrane domain (aa 759-780) and a cytoplasmic region (aa 781-1338) with a tyrosine kinase domain and several autocatalytic phosphotyrosine sites. Human VEGFR1 ECD shares 78%, 78%, 84%, 87%, and 90% aa sequence identity with mouse, rat, porcine, canine and equine VEGFR1, respectively. Soluble forms of the VEGFR1 ECD are produced by alternative splicing, and may also be shed during regulated intracellular proteolysis (4-10). Both soluble and transmembrane forms can inhibit angiogenesis by binding and sequestering its ligands, VEGF (VEGF-A), VEGF-B or PlGF (6‑11). VEGFR1 dimerizes upon ligand binding, which can include heterodimerization with VEGFR2 that modifies VEGFR2-mediated endothelial proliferation and vessel branching (8, 11, 12). VEGFR1 binds VEGF with higher affinity than does VEGFR2, but shows weaker kinase activity (9, 13). Both PlGF and VEGF induce autophosphorylation of transmembrane VEGFR1 (5, 9, 13). While deletion of mouse VEGFR1 is lethal due to overgrowth and disorganization of the vasculature, kinase-inactive mutants are viable (13, 14). VEGFR1 is up‑regulated during hypoxia, and participates in neovascularization and wound healing (1, 2, 15). VEGFR1 engagement on monocyte/macrophage lineage cells enhances their migration, and release of growth factors and cytokines (1, 3, 13, 16). Lymphangiogenesis, angiogenesis, and growth-promoting effects of VEGFR1 are thought to result from enhanced migration of macrophages from the bone marrow to tumors and tissues where they recruit endothelial progenitors (3, 16). Circulating levels of VEGFR1 increase during pregnancy and are further elevated in preeclampsia (4, 6, 17).
Recombinant Human VEGFR1/Flt-1 Fc Chimera Protein
R&D Systems | Catalog # 321-FL


Key Product Details
- R&D Systems Sf 21 (baculovirus)-derived Recombinant Human VEGFR1/Flt-1 Fc Chimera Protein (321-FL)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
Accession Number
Structure / Form
Applications
Product Specifications
Source
| Human VEGFR1 (Ser27-His687) Accession # AAC50060 |
IEGRMD | Human IgG1 (Pro100-Lys330) |
6-His tag |
| N-terminus | C-terminus | ||
Purity
Endotoxin Level
N-terminal Sequence Analysis
Predicted Molecular Mass
SDS-PAGE
Activity
The ED50 for this effect is 5-30 ng/mL.
Reviewed Applications
Read 12 reviews rated 4.8 using 321-FL in the following applications:
Formulation, Preparation, and Storage
Carrier Free
What does CF mean?CF stands for Carrier Free (CF). We typically add Bovine Serum Albumin (BSA) as a carrier protein to our recombinant proteins. Adding a carrier protein enhances protein stability, increases shelf-life, and allows the recombinant protein to be stored at a more dilute concentration. The carrier free version does not contain BSA.
What formulation is right for me?In general, we advise purchasing the recombinant protein with BSA for use in cell or tissue culture, or as an ELISA standard. In contrast, the carrier free protein is recommended for applications, in which the presence of BSA could interfere.
Carrier: 321-FL
| Formulation | Lyophilized from a 0.2 μm filtered solution in MOPS, NaCl and CHAPS with BSA as a carrier protein. |
| Reconstitution | Reconstitute at 100 μg/mL in sterile PBS containing at least 0.1% human or bovine serum albumin. |
| Shipping | The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Carrier Free: 321-FL/CF
| Formulation | Lyophilized from a 0.2 μm filtered solution in MOPS, NaCl and CHAPS. |
| Reconstitution | Reconstitute at 100 μg/mL in sterile PBS. |
| Shipping | The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Calculators
Background: VEGFR1/Flt-1
References
- Otrock, Z.K. et al. (2007) Blood Cells Mol. Dis. 38:258.
- Peters, K.G. et al. (1993) Proc. Natl. Acad. Sci. USA 90:8915.
- Murakami, M. et al. (2008) Arterioscler. Thromb. Vasc. Biol. 28:658.
- Al-Ani, B. et al. (2010) Hypertension 55:689.
- Rahimi, N. et al. (2009) Cancer Res. 69:2607.
- He, Y. et al. (1999) Molecular Endocrinology 13:537.
- Cai, J. et al. (2012) EMBO Mol. Med. 4:980.
- Kendall, R.L. and K.A. Thomas (1993) Proc. Natl. Acad. Sci. USA 90:10705.
- Sawano, A. et al. (1996) Cell Growth Differ. 7:213.
- Barleon, B. et al. (1997) J. Biol. Chem. 272:10382.
- Kappas, N.C. et al. (2008) J. Cell Biol. 181:847.
- Mac Gabhann, F. and A.S. Popel (2007) Biophys. Chem. 128:125.
- Hiratsuka, S. et al. (1998) Proc. Natl. Acad. Sci. USA 95:9349.
- Fong, G.H. et al. (1995) Nature 376:66.
- Nishi, J. et al. (2008) Circ. Res. 103:261.
- Muramatsu, M. et al. (2010) Cancer Res. 70:8211.
- Levine, R.J. et al. (2004) N. Engl. J. Med. 350:672.
Long Name
Alternate Names
Gene Symbol
UniProt
Additional VEGFR1/Flt-1 Products
Product Documents for Recombinant Human VEGFR1/Flt-1 Fc Chimera Protein
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Human VEGFR1/Flt-1 Fc Chimera Protein
For research use only
Citations for Recombinant Human VEGFR1/Flt-1 Fc Chimera Protein
Customer Reviews for Recombinant Human VEGFR1/Flt-1 Fc Chimera Protein (12)
Have you used Recombinant Human VEGFR1/Flt-1 Fc Chimera Protein?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
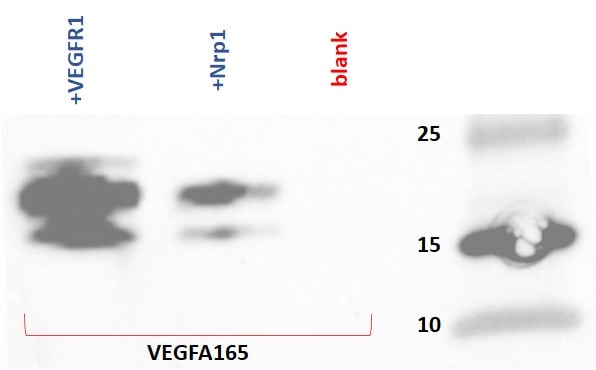
Application: Binding assay/Protein-protein interactionVerified Customer | Posted 07/30/2025
-
Application: Co-immunoprecipitation studyVerified Customer | Posted 08/05/2020
-
Application: SDS-PAGE ControlVerified Customer | Posted 04/12/2018
-
Application: Binding assay/Protein-protein interactionVerified Customer | Posted 03/30/2018
-
Application: SDS-PAGE ControlVerified Customer | Posted 03/15/2018
-
Application: SDS-PAGE ControlVerified Customer | Posted 03/11/2018
-
Application: Binding assay/Protein-protein interactionVerified Customer | Posted 11/27/2017
-
Application: Binding assay/Protein-protein interactionVerified Customer | Posted 11/18/2017
-
Application: In vitro bioactivity in cell cultureVerified Customer | Posted 11/10/2017
-
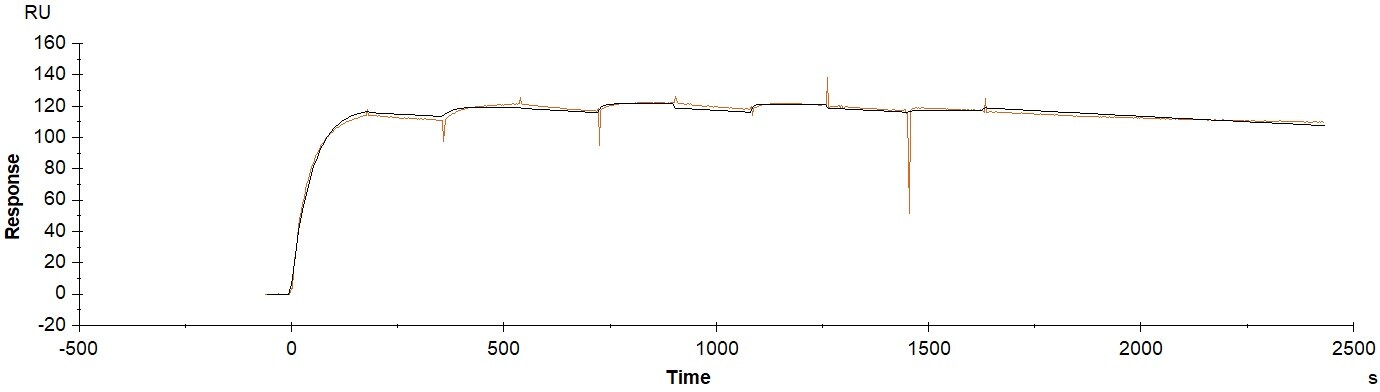
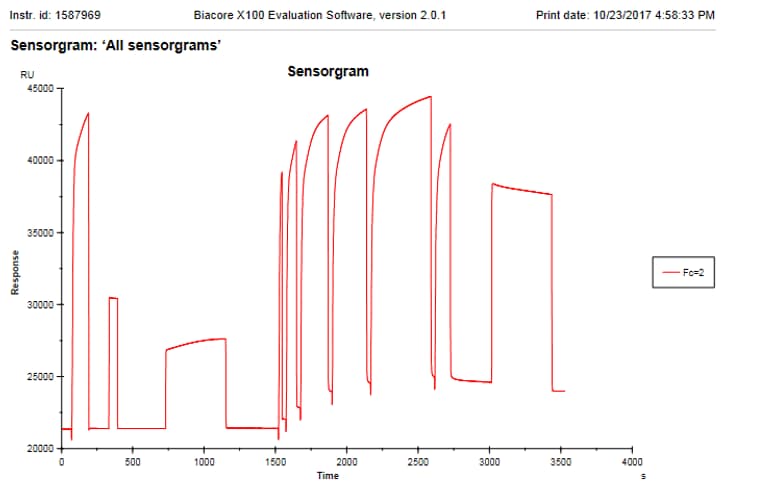
Application: Surface plasma resonanceVerified Customer | Posted 10/23/2017
-
Application: Immunoassay StandardVerified Customer | Posted 07/29/2017
-
Application: Binding assay/Protein-protein interactionVerified Customer | Posted 07/29/2017
There are no reviews that match your criteria.
FAQs for Recombinant Human VEGFR1/Flt-1 Fc Chimera Protein
-
Q: Can Recombinant Human VEGF R1/Flt-1 Fc Chimera (Catalog # 321-FL) block the activity of Mouse VEGF?
A:
Yes. In our testing of HUVECs, catalog # 321-FL blocks recombinant mouse VEGF as well as it blocks recombinant human VEGF.
-
Q: Is Recombinant Human VEGFR1/Flt-1 Fc Chimera Protein (Catalog # 321-FL) the soluble form or membrane-bound?
A: VEGFR1 can be either membrane-bound or soluble. The soluble forms are either one of the isoforms that lack the transmembrane and cytoplasmic domains, or the extracellular domain (ECD) that is proteolytically cleaved off the membrane-bound form. Catalog # 321-FL consists of Ser27-His687 of the canonical isoform (which is in the ECD), plus a linker sequence, an Fc chimera, and a C-terminal 6-his tag.
-
Q: Can Recombinant Human VEGF R1/Flt-1 Fc Chimera (Catalog # 321-FL) block the activity of Mouse VEGF?
A:
Yes. In our testing of HUVECs, catalog # 321-FL blocks recombinant mouse VEGF as well as it blocks recombinant human VEGF.
-
Q: Is Recombinant Human VEGFR1/Flt-1 Fc Chimera Protein (Catalog # 321-FL) the soluble form or membrane-bound?
A: VEGFR1 can be either membrane-bound or soluble. The soluble forms are either one of the isoforms that lack the transmembrane and cytoplasmic domains, or the extracellular domain (ECD) that is proteolytically cleaved off the membrane-bound form. Catalog # 321-FL consists of Ser27-His687 of the canonical isoform (which is in the ECD), plus a linker sequence, an Fc chimera, and a C-terminal 6-his tag.
Associated Pathways
 MAPK Signaling Pathway: Mitogen Stimulation Pathway
MAPK Signaling Pathway: Mitogen Stimulation Pathway
 mTOR Signaling Pathway
mTOR Signaling Pathway