TACS MTT Cell Proliferation Assay
R&D Systems | Catalog # 4890-050-K
A sensitive kit for the measurement of cell proliferation using MTT

Loading...
Key Product Details
Features
A sensitive kit for the measurement of cell proliferation based upon the reduction of the tetrazolium salt, 3,[4,5-dimethylthiazol-2- yl]-2,5-diphenyl-tetrazolium bromide (MTT).
Key Benefits
- Allows measurement of proliferation rate, and conversely, the reduction in cell viability.
- Minimizes the number of steps necessary to complete the assay and interpret the data.
- Yields low background absorbance values in the absence of cells.
- Accurate quantification of changes in cell proliferation due to the linear relationship between cell number and signal produced for each cell type.
- Stable when stored at 4°C.
Species
Multi-Species
Product Summary for TACS MTT Cell Proliferation Assay
Why Use the TACS MTT Cell Proliferation Assay?
Changes in cell proliferative activity caused by trophic factors, growth inhibitors, or inducers and inhibitors of apoptosis, may be quantified using the TACS MTT Cell Proliferation Assay. MTT is reduced to an insoluble formazan dye by mitochondrial enzymes associated with metabolic activity. The reduction of MTT is primarily due to glycolytic activity within the cell and is dependent upon the presence of NADH and NADPH. The resultant intracellular purple formazan can be solubilized and quantitated by spectroscopic means.
MTT can be used to safely assess cell proliferation, cell viability, and/or cytotoxicity. MTT is added directly to the culture medium and is reduced by metabolically active cells to insoluble purple formazan dye crystals. The absorbance of the sample is read directly in the wells at an optimal wavelength of 570 nm, but any filter that absorbs between 550 and 600 nm may be used.
MTT can be used to safely assess cell proliferation, cell viability, and/or cytotoxicity. MTT is added directly to the culture medium and is reduced by metabolically active cells to insoluble purple formazan dye crystals. The absorbance of the sample is read directly in the wells at an optimal wavelength of 570 nm, but any filter that absorbs between 550 and 600 nm may be used.
Loading...
Scientific Data Images for TACS MTT Cell Proliferation Assay
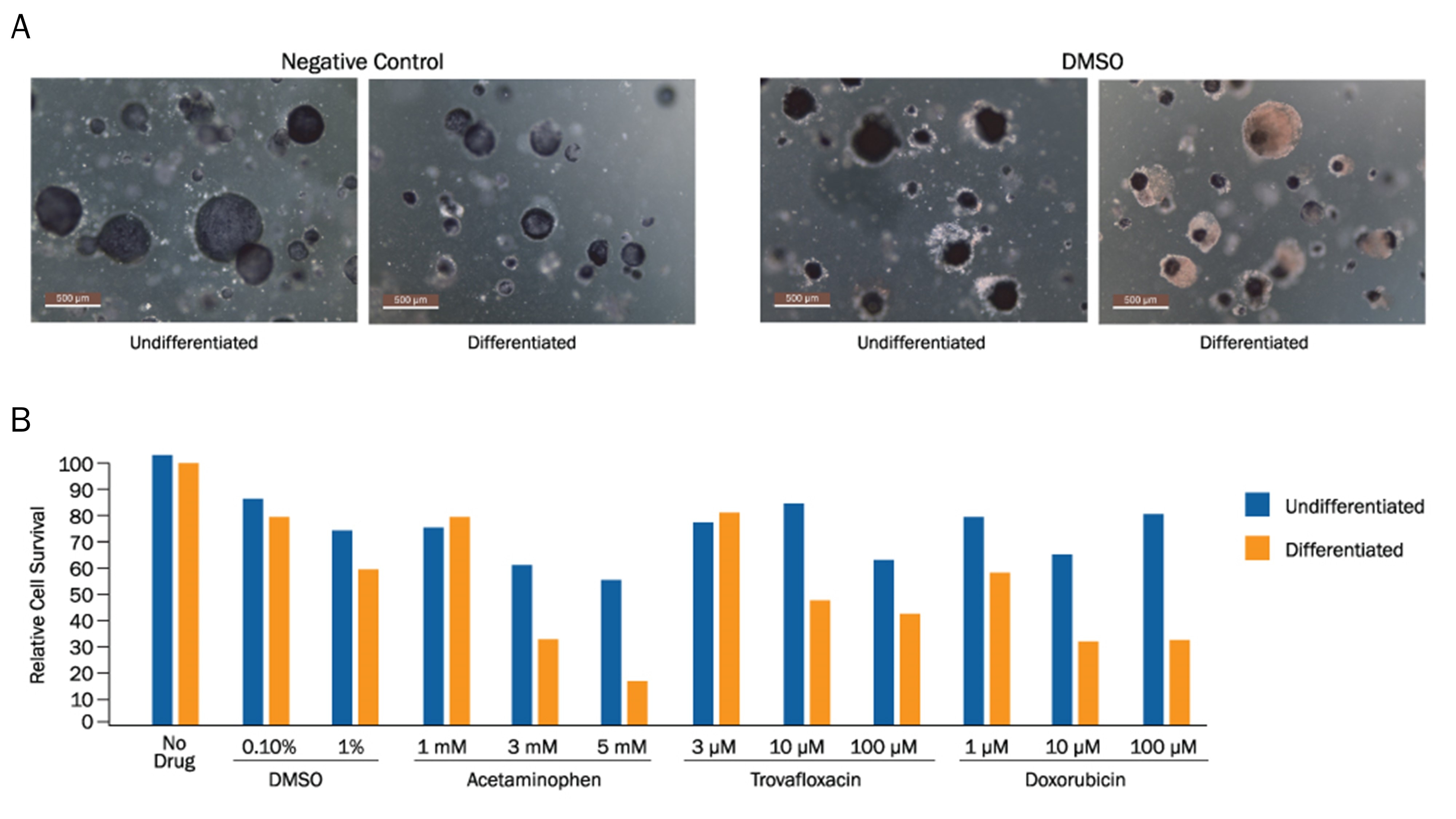
MTT Assay of Human Tissue-derived Liver Organoids.
Human Liver organoids were derived from human biopsy tissue. Undifferentiated liver organoids were formed by embedding dissociated tissue in Cultrex RGF BME, Type 2 (Catalog # 3533-005-02) and culturing in specialized media. Liver organoids were differentiated using media containing Recombinant Human FGF-19 (Catalog # 969-FG), DAPT (Catalog # 2634), and Dexamethasone (Catalog # 1126). A) Representative images of untreated control and DMSO-treated (positive control) undifferentiated and differentiated liver organoids. B) Quantification of MTT staining of cells treated with varying doses of acetaminophen, Trovafloxin, or Doxorubicin showed a dose-dependent loss of cell viability under each condition. Interestingly, liver organoids showed increased vulnerability to hepatotoxicity as they matured in differentiation media.Toxicity analysis of iPSC-derived Hepatocytes using TACS MTT Proliferation Assay and the CometChip Assay.
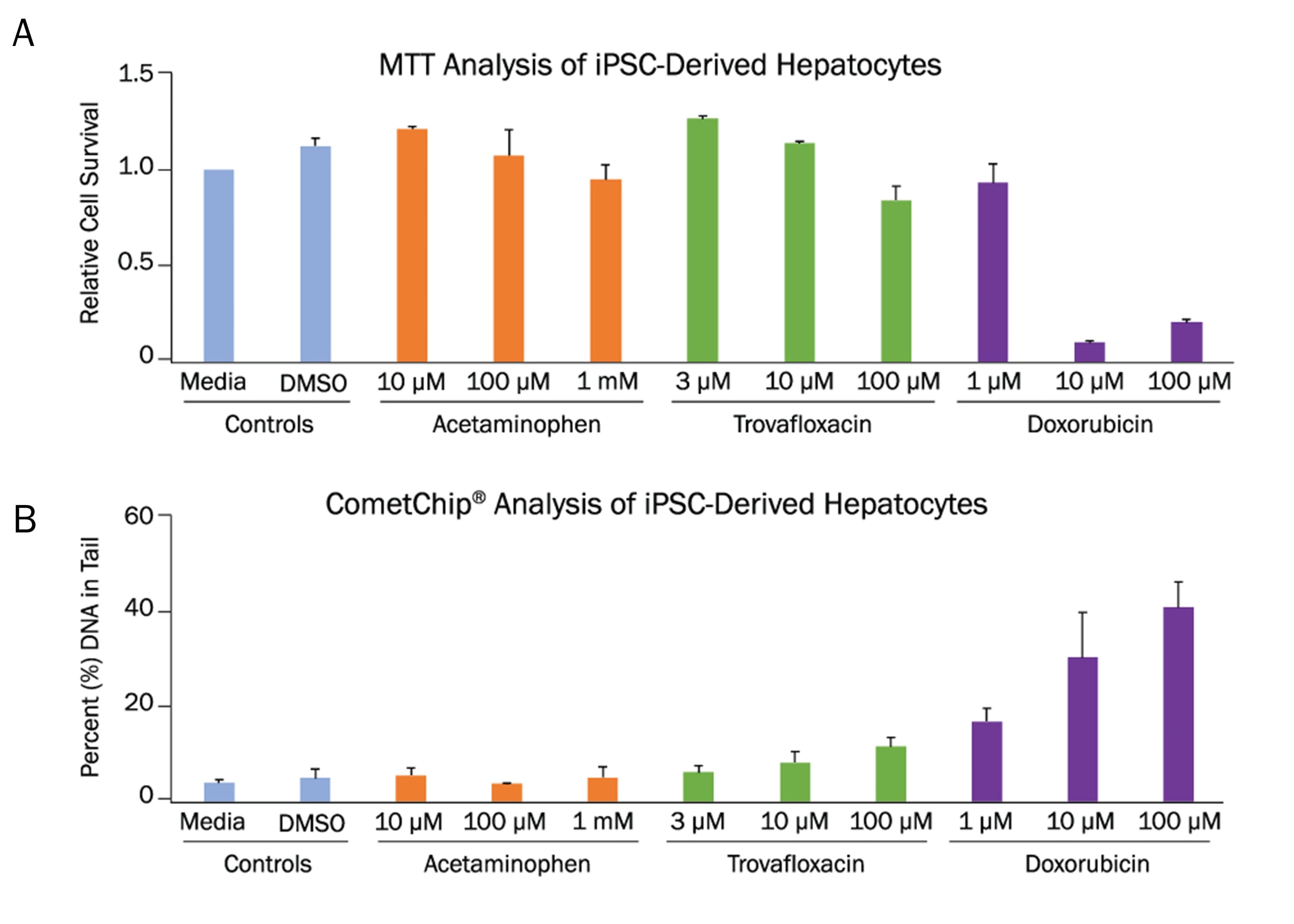
iPSC-derived hepatocytes were generated using the StemXVivo Hepatocyte Differentiation Kit (Catalog # SC033). iPSC-derived hepatocytes were treated with Acetaminophen, Trovafloxacin, and Doxorubicin and dose response toxicity profiles using the (A) TACS MTT Cell Proliferation Assay or (B) CometChip (Catalog # 4260-096-ESK). Using the MTT assay, Trovafloxacin and Doxorubicin showed significantly increased loss of cell metabolic activity at 100 µM and >10 µM, respectively. Increased DNA damage was detected at these two concentrations using the CometChip (n ≥ 3 for each group).HepG2 Spheroid Toxicity Detected using the MTT Assay.
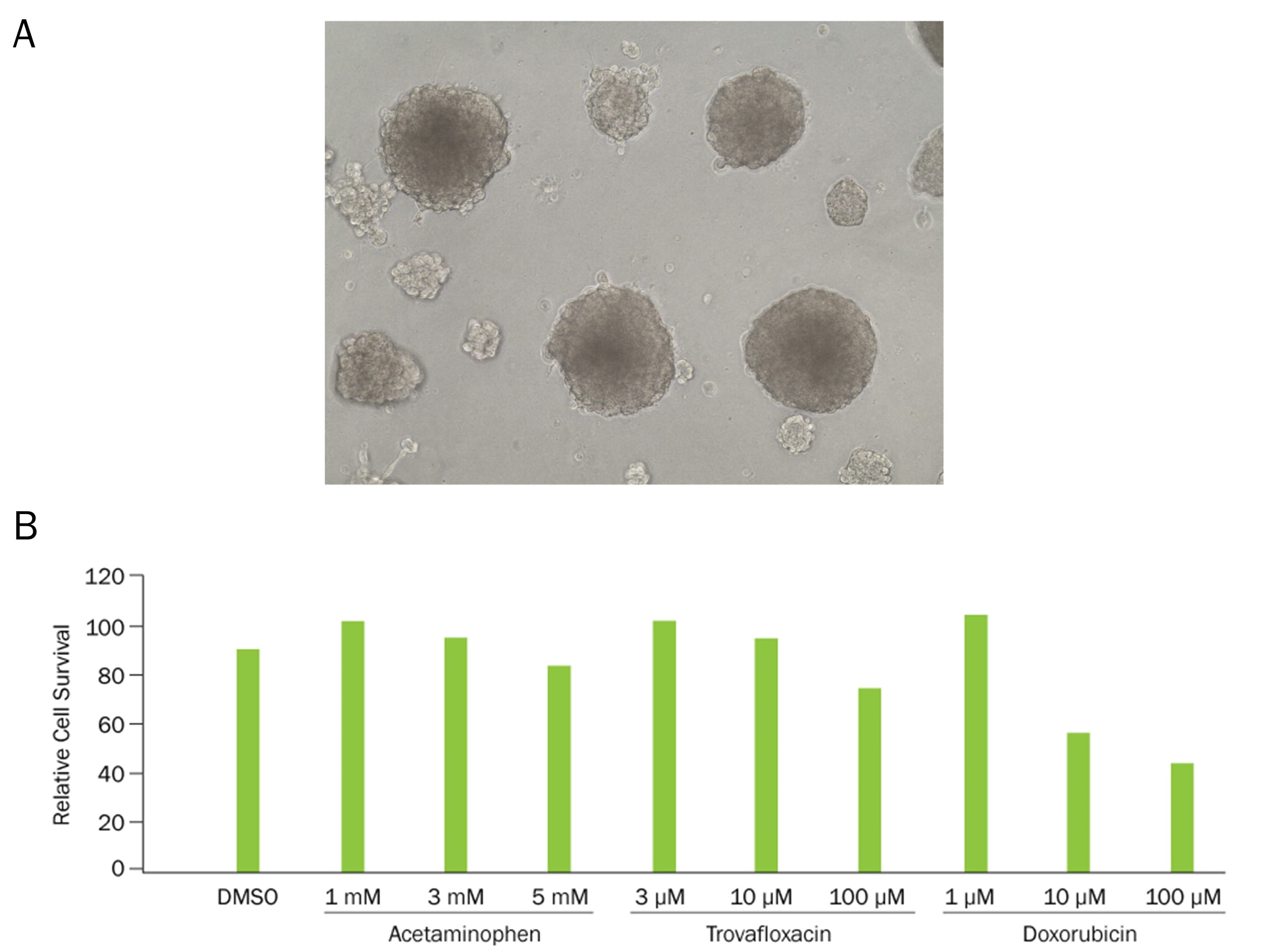
A) Hepatocyte spheroids were formed by plating HepG2 liver hepatocellular carcinoma cells (10,000 cells per well) in a 24-well plate coated with Cultrex RGF BME, Type 2 (Catalog # 3533-005-02). B) HepG2 spheroids were cultured for 21 days prior exposure to acetaminophen, trovafloxacin, or doxorubicin. Spheroids showed significantly increased loss of cell metabolic activity at 100 µM and >10 µM, respectively (n ≥ 3 for each group), as detected by the MTT Assay.Kit Contents for TACS MTT Cell Proliferation Assay
- MTT Reagent
- Detergent Reagent
Formulation, Preparation, and Storage
Shipping
The components for this kit may require different storage/shipping temperatures and may arrive in separate packaging. Upon receipt, store products immediately at the temperature recommended on the product labels.
Storage
Store the unopened product at 2 - 8 °C. Do not use past expiration date.
Product Documents for TACS MTT Cell Proliferation Assay
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Related Research Areas
Citations for TACS MTT Cell Proliferation Assay
Customer Reviews for TACS MTT Cell Proliferation Assay
There are currently no reviews for this product. Be the first to review TACS MTT Cell Proliferation Assay and earn rewards!
Have you used TACS MTT Cell Proliferation Assay?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Loading...