Choosing the Right Model System for Hepatotoxicity: A Comparison of 3-D Liver Organoids, HepG2 Spheroids and iPSC-Derived Hepatocytes Using MTT and DNA Comet Assays
by Susan Tousey, David Galitz, Marie Pundt, Xi Yu, Marnelle Anderson, Greg Herr, Sol Degese, Cyrus Muns.
Scientific Meeting PostersABSTRACT
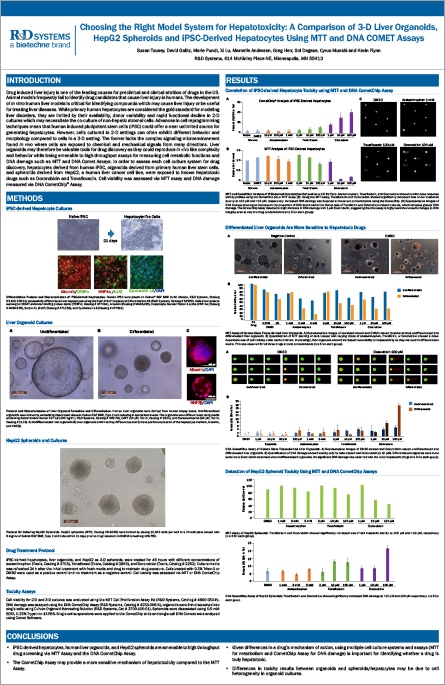
The liver is the primary organ system for drug metabolism and detoxification. In this role, it is also highly susceptible to damage from pharmaceuticals and other chemical toxicants. Animal models and traditional in vitro assays modeling liver metabolism often fail to recapitulate the in vivo toxicity of drugs in human patients. A classic example is the cancer drug Trovafloxacin mesylate, which cleared preclinical animal models for safety and achieved FDA clearance, but was later retracted due to acute liver toxicity resulting in numerous fatalities. Failures, such as this, can have devastating health outcomes as well as result in huge financial costs for pharmaceutical companies. Therefore, it is imperative to use appropriate preclinical models that can accurately predict human toxicity. Although regulatory guidelines still mandate the minimum lethal dose test in rats, cell culture models can reap substantial time- and cost-savings. Hepatocyte cell culture models are increasingly being used for high throughput liver toxicity screening early during the drug development pipeline. In this study, we compare three in vitro hepatotoxicity model systems: induced pluripotent stem cell (iPSC)-derived hepatocytes, 3-D liver organoids, and HepG2 spheroids. Using a panel of known hepatotoxic compounds we evaluated the performance of these models using high throughput toxicity assays, including the MTT Assay, a robust test for cellular toxicity, and the DNA Comet assay and CometChip® technology to assess genotoxicity. We conclude that each of the different hepatocyte model systems tested offer advantages for evaluating toxicity, and that these advantages can be used in a complementary fashion to accurately probe dose responses for liver toxicity.
View All Products for 3-D Cell Culture and Organoid Technology
View All Products for induced Pluripotent Stem Cell Research
View Comet Assays and CometChip Technology
View MTT Cell Proliferation/Viability Assays
Download PDF