122 results for "Butyrylcholinesterase/BCHE" in Products
Butyrylcholinesterase/BCHE Products
Butyrylcholinesterase (BCHE) is a major acetylcholine hydrolyzing enzyme in the circulation. Although it is present in significant amounts (~3 mg/L) in human plasma, no endogenous physiological substrate has been described for this enzyme. It can degrade a large number of ester-containing compounds in addition to acylcholines. Thus, it is likely to play significant pharmacological and toxicological roles. It is thought to be involved in the pathological process of Alzheimer's disease (AD) by ...
| Reactivity: | Mouse, Rat |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
| Reactivity: | Human |
| Sensitivity: | 0.06 ng/mL |
| Assay Range: | 0.156-10 ng/mL (Cell Culture Supernates, Cell Lysates, Tissue Lysates, Serum, EDTA Plasma, Heparin Plasma, Saliva, Urine, Human Milk) |
| Applications: | ELISA |
Cholinesterase inhibitor
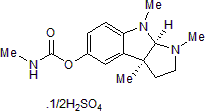
| Chemical Name: | (3aS)-cis-1,2,3,3a,8,8a-Hexahydro-1,3a,8-trimethylpyrrolo[2,3-b]indol-5-ol methylcarbamate hemisulfate |
| Purity: | ≥98% |
Cholinesterase inhibitor
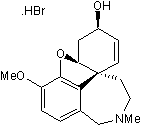
| Chemical Name: | 4a,5,9,10,11,12-Hexahydro-3-methoxy-11-methyl-6H-benzofuro[3a,3,2-ef][2]benzazepin-6-ol hydrobromide |
| Purity: | ≥98% |
| Reactivity: | Human |
| Source: | CHO |
| Accession #: | P06276 |
| Applications: | Enzyme Activity |
Dual AChE and BChE inhibitor
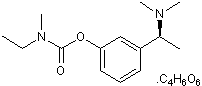
| Alternate Names: | ENA 713 |
| Chemical Name: | N-Ethyl-N-methylcarbamic acid 3-[(1S)-1-(dimethylamino)ethyl]phenyl ester (2R,3R)-2,3-dihydroxybutanedioate |
| Purity: | ≥98% |
| Reactivity: | Human, Mouse |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC |
| Reactivity: | Mouse |
| Source: | NS0 |
| Accession #: | AAH99977 |
| Applications: | Enzyme Activity |
For use with catalog number DBCHE0
| Reactivity: | Human |
| Applications: | ELISA, Ctrl |
| Reactivity: | Human, Canine |
| Details: | Mouse IgG1 Monoclonal Clone #OTI1E1 |
| Applications: | WB, Flow |
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB |
| Reactivity: | Mouse, Rat |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
| Reactivity: | Mouse, Rat |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
| Reactivity: | Mouse, Rat |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
| Reactivity: | Mouse, Rat |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
| Reactivity: | Mouse, Rat |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
| Reactivity: | Mouse, Rat |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
| Reactivity: | Mouse, Rat |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
| Reactivity: | Mouse, Rat |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
| Reactivity: | Mouse |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, ELISA |
| Reactivity: | Human |
| Applications: | WB |
Recombinant Monoclonal Antibody
| Reactivity: | Human |
| Details: | Rabbit IgG Monoclonal Clone #4N2N9 |
| Applications: | WB |
Recombinant Monoclonal Antibody
| Reactivity: | Human |
| Details: | Rabbit IgG Monoclonal Clone #1E9 |
| Applications: | WB, ELISA |
| Reactivity: | Human |
| Details: | Rabbit IgG Monoclonal Clone #24GB3485 |
| Applications: | WB, Flow, ICC/IF |
Recombinant Monoclonal Antibody
| Reactivity: | Human |
| Details: | Rabbit IgG Monoclonal Clone #SR2272 |
| Applications: | WB |






![Immunohistochemistry-Paraffin: Butyrylcholinesterase/BCHE Antibody [NBP1-85633] Immunohistochemistry-Paraffin: Butyrylcholinesterase/BCHE Antibody [NBP1-85633]](https://resources.rndsystems.com/images/products/Butyrylcholinesterase-BCHE-Antibody-Immunohistochemistry-Paraffin-NBP1-85633-img0004.jpg)

![Western Blot: Butyrylcholinesterase/BCHE Antibody (OTI1E1) [NBP2-03523] Western Blot: Butyrylcholinesterase/BCHE Antibody (OTI1E1) [NBP2-03523]](https://resources.rndsystems.com/images/products/Butyrylcholinesterase-Antibody-1E1-Western-Blot-NBP2-03523-img0003.jpg)
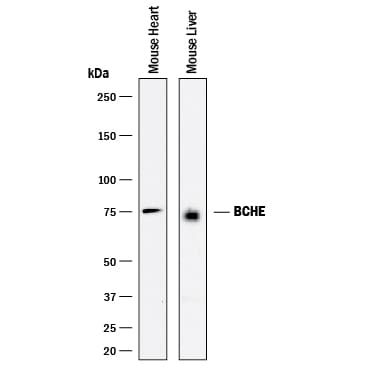
![Western Blot: Butyrylcholinesterase/BCHE Antibody [NBP1-69541] Western Blot: Butyrylcholinesterase/BCHE Antibody [NBP1-69541]](https://resources.rndsystems.com/images/products/Butyrylcholinesterase-BCHE-Antibody-Western-Blot-NBP1-69541-img0003.jpg)
![Immunohistochemistry-Paraffin: Butyrylcholinesterase/BCHE Antibody [NBP2-98275] Immunohistochemistry-Paraffin: Butyrylcholinesterase/BCHE Antibody [NBP2-98275]](https://resources.rndsystems.com/images/products/Butyrylcholinesterase-BCHE-Antibody-Immunohistochemistry-Paraffin-NBP2-98275-img0002.jpg)
![Western Blot: Butyrylcholinesterase/BCHE Overexpression Lysate [NBL1-07942] Western Blot: Butyrylcholinesterase/BCHE Overexpression Lysate [NBL1-07942]](https://resources.rndsystems.com/images/products/Butyrylcholinesterase-Overexpression-Lysate-Adult-Normal-Western-Blot-NBL1-07942-img0002.jpg)
![Western Blot: Butyrylcholinesterase/BCHE Antibody (4N2N9) [NBP3-16712] Western Blot: Butyrylcholinesterase/BCHE Antibody (4N2N9) [NBP3-16712]](https://resources.rndsystems.com/images/products/Butyrylcholinesterase-BCHE-Antibody-4N2N9-Western-Blot-NBP3-16712-img0001.jpg)
![Western Blot: Butyrylcholinesterase/BCHE Antibody (1E9) [NBP3-15164] Western Blot: Butyrylcholinesterase/BCHE Antibody (1E9) [NBP3-15164]](https://resources.rndsystems.com/images/products/Butyrylcholinesterase-BCHE-Antibody-1E9-Western-Blot-NBP3-15164-img0001.jpg)
![Western Blot: Butyrylcholinesterase/BCHE Antibody (SR2272) [NBP3-21793] -](https://resources.rndsystems.com/images/products/nbp3-21793_rabbit-butyrylcholinesterase-bche-mab-sr2272-6720239533752.jpg)