19 results for "GABA-A R beta 1" in Products
GABA-A R beta 1 Products
GABA A (γ-aminobutyric acid-type A) receptors are members of the cysteine-loop family of neurotransmitter-gated ion channels. GABA binding to A-type receptors induces anion-selective ion channel opening. These receptors are the principal fast inhibitory neurotransmitter receptors in the CNS. GABA A receptors are heteropentamer combinations of seven subunit types; α, β, γ, δ, ε, θ, and π. Three subunits, α, β, and γ, have at least ...
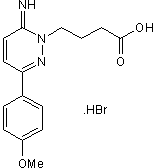
Competitive and selective GABAA antagonist
| Alternate Names: | Gabazine |
| Chemical Name: | 6-Imino-3-(4-methoxyphenyl)-1(6H)-pyridazinebutanoic acid hydrobromide |
| Purity: | ≥98% |
| Reactivity: | Human, Mouse, Rat |
| Details: | Mouse IgG1 Monoclonal Clone #S96-55 |
| Applications: | IHC, WB, ICC/IF, MA |
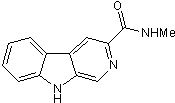
Benzodiazepine inverse agonist
| Chemical Name: | N-Methyl-β-carboline-3-carboxamide |
| Purity: | ≥99% |
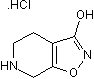
GABAA agonist
| Alternate Names: | Gaboxadol |
| Chemical Name: | 4,5,6,7-Tetrahydroisoxazolo[5,4-c]pyridin-3-ol hydrochloride |
| Purity: | ≥98% |
| Reactivity: | Mouse, Rat |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB |
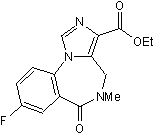
Benzodiazepine antagonist
| Alternate Names: | Ro 15-1788 |
| Chemical Name: | 8-Fluoro-5,6-dihydro-5-methyl-6-oxo-4H-imidazo[1,5-a][1,4]benzodiazepine-3-carboxylic acid, ethyl ester |
| Purity: | ≥99% |
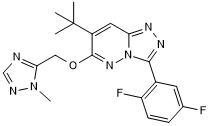
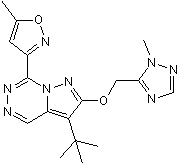
GABAA partial agonist; displays subtype selectivity
| Chemical Name: | 3-(2,5-Difluorophenyl)-7-(1,1-dimethylethyl)-6-[(1-methyl-1H-1,2,4-triazol-5-yl)methoxy]-1,2,4-triazolo[4,3-b]pyridazine |
| Purity: | ≥98% |
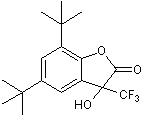
Potent and selective postitive allosteric modulator of GABAB receptors
| Chemical Name: | 5,7-Bis(1,1-dimethylethyl)-3-hydroxy-3(trifluoromethyl)-2(3H)-benzofuranone |
| Purity: | ≥98% |
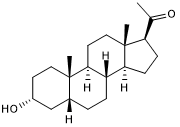
Postitive allosteric modulator of GABAA receptors
| Chemical Name: | (3α,5β)-3-Hydroxy-pregnan-20-one |
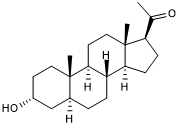
Positive allosteric modulator of GABAA receptors
| Alternate Names: | 3a,5a-THPROG |
| Chemical Name: | (3α,5α)-3-Hydroxy-pregnan-20-one |
α5-selective GABAA inverse agonist
| Chemical Name: | 3-(1,1-Dimethylethyl)-7-(5-methyl-3-isoxazolyl)-2-[(1-methyl-1H-1,2,4-triazol-5-yl)methoxy]-pyrazolo[1,5-d][1,2,4]triazine |
| Purity: | ≥98% |
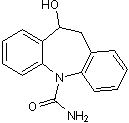
Active metabolite of oxcarbazepine (Cat. No. 3864)
| Alternate Names: | GP 47779 |
| Chemical Name: | 10,11-Dihydro-10-hydroxy-5H-dibenz(Z)[b,f]azepin-5-carboxamide |
| Purity: | ≥98% |
| Reactivity: | Human |
Recombinant Monoclonal Antibody
| Reactivity: | Human, Mouse, Rat |
| Details: | Rabbit IgG Monoclonal Clone #1Q3A9 |
| Applications: | WB |
Recombinant Monoclonal Antibody
| Reactivity: | Human, Mouse, Rat |
| Details: | Rabbit IgG Monoclonal Clone #SR1833 |
| Applications: | WB |
| Reactivity: | Human |
| Source: | Wheat germ |
| Applications: | WB, ELISA, AP, MA |
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC |
| Reactivity: | Human |
| Applications: | WB |
| Reactivity: | Human |
| Source: | E. coli |
| Applications: | AC |



![Western Blot: GABA-A R beta 1 Antibody (S96-55) [NBP1-48319] Western Blot: GABA-A R beta 1 Antibody (S96-55) [NBP1-48319]](https://resources.rndsystems.com/images/products/GABA-A-R-beta-1-Antibody-S96-55-Western-Blot-NBP1-48319-img0004.jpg)
![Western Blot: GABA-A R beta 1 Antibody [NB300-197] Western Blot: GABA-A R beta 1 Antibody [NB300-197]](https://resources.rndsystems.com/images/products/GABA-A-R-beta-1-Antibody-Western-Blot-NB300-197-img0005.jpg)


![Western Blot: GABA-A R beta 1 Antibody (1Q3A9) [NBP3-15857] Western Blot: GABA-A R beta 1 Antibody (1Q3A9) [NBP3-15857]](https://resources.rndsystems.com/images/products/GABA-A-R-beta-1-Antibody-1Q3A9-Western-Blot-NBP3-15857-img0001.jpg)
![Western Blot: GABA-A R beta 1 Antibody (SR1833) [NBP3-21964] -](https://resources.rndsystems.com/images/products/nbp3-21964_rabbit-gaba-a-r-beta-1-mab-sr1833-672023106253.jpg)
![Immunohistochemistry-Paraffin: GABA-A R beta 1 Antibody [NBP2-14034] Immunohistochemistry-Paraffin: GABA-A R beta 1 Antibody [NBP2-14034]](https://resources.rndsystems.com/images/products/GABA-A-R-beta-1-Antibody-Immunohistochemistry-Paraffin-NBP2-14034-img0002.jpg)
![Western Blot: GABA-A R beta 1 Overexpression Lysate [NBL1-10920] Western Blot: GABA-A R beta 1 Overexpression Lysate [NBL1-10920]](https://resources.rndsystems.com/images/products/GABA-A-Receptor-beta-1-Overexpression-Lysate-Adult-Normal-Western-Blot-NBL1-10920-img0002.jpg)