HIF-1 alpha Antibody - BSA Free
Novus Biologicals | Catalog # NB100-134

![Western Blot: HIF-1 alpha AntibodyBSA Free [NB100-134] Western Blot: HIF-1 alpha AntibodyBSA Free [NB100-134]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody---BSA-Free-Western-Blot-NB100-134-img0013.jpg)
Key Product Details
Validated by
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Format
Product Specifications
Immunogen
Reactivity Notes
Localization
Clonality
Host
Isotype
Theoretical MW
Disclaimer note: The observed molecular weight of the protein may vary from the listed predicted molecular weight due to post translational modifications, post translation cleavages, relative charges, and other experimental factors.
Scientific Data Images for HIF-1 alpha Antibody - BSA Free
Western Blot: HIF-1 alpha AntibodyBSA Free [NB100-134]
Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - Analysis of HIF-1 alpha in human retinal and choroidal primary endothelia cells exposed to hypoxic conditions in the times noted using NB100-134. Image from verified customer review.Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody - BSA Free [NB100-134]
HIF-1-alpha-Antibody---BSA-Free-Immunocytochemistry-Immunofluorescence-NB100-134-img0017.jpgImmunohistochemistry-Paraffin: HIF-1 alpha Antibody - BSA Free [NB100-134]
Immunohistochemistry-Paraffin: HIF-1 alpha Antibody - BSA Free [NB100-134] - Analysis of HIF-1 alpha in zebrafish retina tissue using NB100-134 HIF-1 alpha antibody. Mouse tissue was used as a control. Image from verified customer review.Western Blot: HIF-1 alpha AntibodyBSA Free [NB100-134]
Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - Analysis of HIF-1 alpha in overexpression and endogenous HIF-1 alpha & HIF-2 alpha using anti-HIF-1 alpha antibody NB100-134. The data showed that HIF-1 alpha antibody did not react to HIF-2 alpha overexpression. Image from verified customer review.Western Blot: HIF-1 alpha AntibodyBSA Free [NB100-134]
Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - Analysis of HIF-1 alpha using NB100-134, on normoxic and hypoxic nuclear rat cell lysates.Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody - BSA Free [NB100-134]
Immunocytochemistry/Immunofluorescence: HIF-1 alpha Antibody - BSA Free [NB100-134] - Analysis of HIF-1 alpha in Normoxic (N) and Hypoxic (H) ARPE-19 cells using anti-HIF-1 alpha antibody NB100-134. Image from verified customer review.Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody - BSA Free [NB100-134]
HIF-1-alpha-Antibody---BSA-Free-Immunocytochemistry-Immunofluorescence-NB100-134-img0016.jpgImmunohistochemistry-Paraffin: HIF-1 alpha Antibody - BSA Free [NB100-134]
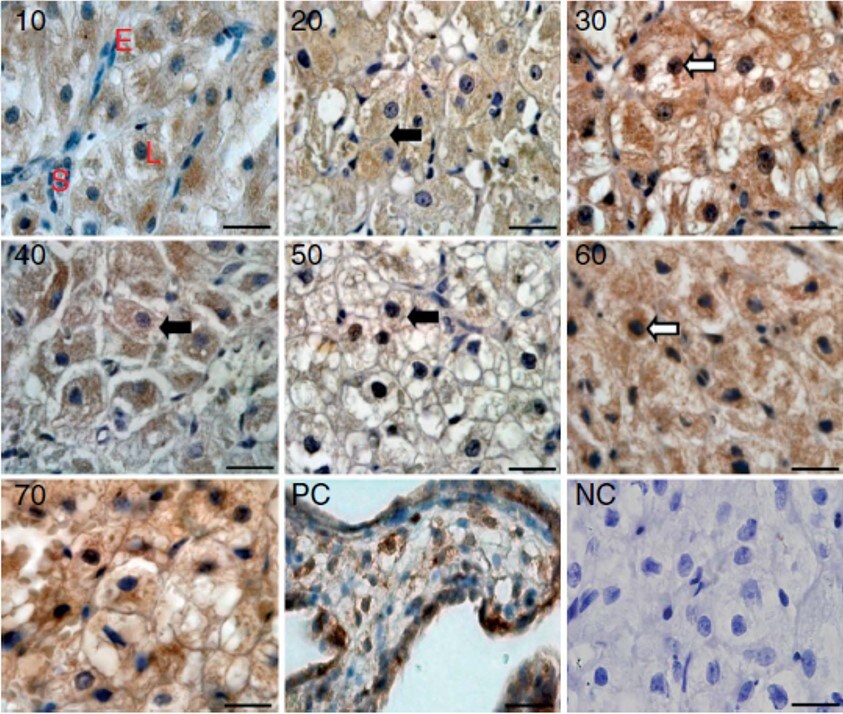
Immunohistochemistry-Paraffin: HIF-1 alpha Antibody - BSA Free [NB100-134] - Staining in the canine CL on days 10 to 70 after ovulation. PC = positive control (human placenta). NC = negative control. Image from verified customer review.Immunohistochemistry: HIF-1 alpha Antibody - BSA Free [NB100-134]
Immunohistochemistry: HIF-1 alpha Antibody - BSA Free [NB100-134] - Staining of human kidney, renal tubular epithelium in cortex using NB100-134.Immunohistochemistry-Paraffin: HIF-1 alpha Antibody - BSA Free [NB100-134]
Immunohistochemistry-Paraffin: HIF-1 alpha Antibody - BSA Free [NB100-134] - Analysis of FFPE tissue section of human endometrium carcinoma AN3CA cell line based xenograft using rabbit polyclonal HIF-1 alpha antibody NB100-134 at 1:300 dilution. The signal was developed using HRP-labelled secondary antibody and DAB reagent, and the section was further counterstained using hematoxylin. The tested section depicted mainly a diffused cytoplasmic staining but there were some cells which showed nuclear signal also (representing hypoxic cells).Simple Western: HIF-1 alpha AntibodyBSA Free [NB100-134]
Simple Western: HIF-1 alpha Antibody - BSA Free [NB100-134] - Image shows a specific band for HIF-1 alpha in 0.2 mg/mL of Hypoxic HeLa lysate. This experiment was performed under reducing conditions using the 12-230 kDa separation system.Immunoprecipitation: HIF-1 alpha Antibody - BSA Free [NB100-134]
Immunoprecipitation: HIF-1 alpha Antibody - BSA Free [NB100-134] - PC-3 cell lysates run with cobalt chloride treatments. Immunoprecipitation was performed with protein A/G agarose beads. This data was provided courtesy of Kelie Reece, Figg lab, NCI.Simple Western: HIF-1 alpha AntibodyBSA Free [NB100-134]
Simple Western: HIF-1 alpha Antibody - BSA Free [NB100-134] - Simple Western lane view shows lysates of MCF +/- DMOG, BioSpherix MSCs in hypoxic conditions, and BioSpherix MSCs in normoxic conditions loaded at 0.5 mg/ml. A band was detected for HIF-1 alpha at approximately 116 kDa (as indicated) using NB100-134 (1:100 dilution) followed by Anti-Rabbit Secondary Antibody (042-206, ProteinSimple). This experiment was conducted under standard assay conditions, and using the 12-230 kDa separation module (SW-W004). Non-specific interaction with the 230 kDa Simple Western standard may be seen with this antibody. Image from an internal validation.Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] -
P53 was increased response to IR in the fetal brain.Even though no significant change in protein level, total p53 and phosphorylation of P53 at ser15 were increased compared with HIF-1 alpha in fetal brains response to IR. Numbers in the pictures show the average of three independent experiments (n = 3 from 3 individual pregnant mice).Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] -
WNT11 is induced by hypoxia or hypoxic mimetics in different cell types.(A) Increased Wnt11 mRNA in EMSC adipocytes (Day 12) after hypoxia-mimetic treatments. EMSC adipocytes were treated with CoCl2 (0.1 mM), DFO (0.1 mM) or DMOG (0.1 mM) for 24 hrs. Values were normalized to Tbp mRNA & are expressed relative to control (n = 3). (B,C) Increased Wnt11 mRNA by hypoxia in EMSC preadipocytes & adipocytes (Day 0–12 after differentiation) (B), & C2C12 myoblast & myocyte (Day 0 & 8 after differentiation) (C). Wnt11 mRNA was assessed by quantitative PCR in cells exposed to air (21% O2) or hypoxia (1% O2) for 24 hrs. (n = 4). Values were normalized to Tbp mRNA & are expressed relative to 21% O2 samples (left panel). (D) Immunoblot analyses of HeLa cells under normal air or hypoxia for 24 hrs. (E,F) Induction of Wnt11 by increasing concentrations of DMOG in MDA-MB-231 cells (E) & 4T1 cells (F). (G) EMSCs treated with 0.1 mM DMOG for the indicated times. Wnt11 & Vegf mRNA expression was measured by qPCR & normalized to Tbp mRNA (n = 4). (H) WNT11 protein levels after DMOG treatment normalized to alpha -Tubulin (upper panel; n = 4). Representative immunoblots of EMSCs treated with 0.1 mM DMOG for the indicated times (Lower panel). (I) Protein expression in MDA-MB-231 cells treated with 0.1 mM DMOG. (J) Induction of Wnt11 promoter activity by hypoxia or hypoxia mimetics. pGL3-Wnt11 promoter plasmid was transfected into C2C12 cells. Cells were incubated with DMOG (left panel, n = 4) or under 21% O2 or 1% O2 (right panel, n = 8) for 24 hrs. For panels (A–C,G,H,J), values are mean ± s.e.m. *p < 0.05, **p < 0.01. For panels of immunoblotting, laminin, alpha -tubulin, & ERK were used as loading controls, WNT11 normalized to alpha -Tubulin was shown. Image collected & cropped by CiteAb from the following publication (https://www.nature.com/articles/srep21520), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] -
Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - LOXL2 abundance in EC‐derived exosomes is increased in hypoxia. (A) Immunoblots of endothelial cells for HIF‐1 alpha (control for hypoxia, top panel), LOXL2 (middle panel) & beta ‐actin (loading control, lower panel). (B) Immunoblots of EC‐derived exosomes for LOXL2 (upper panel), & beta ‐actin (loading control, lower panel). (C) Densitometric quantification of relative LOXL2 protein abundance in control & hypoxic EC‐derived exosomes (n = 4 ± SD, Student's t‐test; **P < 0.01). (D) Immunoblots of sucrose density gradient samples of EC‐derived exosomes for LOXL2 (upper panel) & exosome‐marker Flotillin‐1 (lower panel). Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/26612622), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody - BSA Free [NB100-134] -
Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody - BSA Free [NB100-134] - Hif1 alpha expression in 9.5dpc primordial germ cells.(A) Whole-mount staining of Hif1 alpha & GFP in Oct4-GFP embryo (lateral view). Scale bar: 100μm. (B) Hif1 alpha & GFP staining of FACS-sorted PGCs. Scale bar: 20μm. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/27148974), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] -
Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - Hypoxic setup for larval zebrafish. (A) (1) oxygen regulator; (2) hypoxic larval aquarium; (3) oxygen electrode; (4) air stone; & (5) solenoid valve. The compressed nitrogen source is shown as a blue gas cylinder. (B) Dissolved oxygen in the hypoxic aquarium decreased slowly over time as nitrogen perfusion increased until dissolved oxygen stabilized at 3.5 mg/L. (C) qRT-PCR analysis showing that hif1 alpha mRNA expression was significantly increased under hypoxia. (D) Western blot of Hif1 alpha protein expression showing that hypoxia enhanced Mbp protein translation. ∗P < 0.05; ∗∗P < 0.01; ∗∗∗P < 0.001. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/30337858), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] -
Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - P53 was increased response to IR in the fetal brain.Even though no significant change in protein level, total p53 & phosphorylation of P53 at ser15 were increased compared with HIF-1 alpha in fetal brains response to IR. Numbers in the pictures show the average of three independent experiments (n = 3 from 3 individual pregnant mice). Image collected & cropped by CiteAb from the following publication (https://dx.plos.org/10.1371/journal.pone.0110577), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody - BSA Free [NB100-134] -
Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody - BSA Free [NB100-134] - Hif1 alpha expression in 15.5dpc germ cells.(A) Sections of male (top) & female (bottom) gonads from 15.5 dpc Oct4-GFP embryos showing Oct4-GFP & Hif1 alpha expressions in germ cells. Scale bars: 50μm (male) & 20μm (female). (B) FACS-sorted male (top) & female (bottom) 15.5dpc germ cells showing Oct4-GFP & Hif1 alpha expressions. Scale bars: 100μm (male) & 50μm (female). Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/27148974), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody - BSA Free [NB100-134] -
Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody - BSA Free [NB100-134] - Hif1 alpha expression in preimplantation embryos.(A) Hif1 alpha staining in two-cell stage embryo & morula (top) & negative control images without primary antibody (bottom). (B) Co-staining of OCT4 & Hif1 alpha in inner cell mass of blastocyst (top) & negative control images without primary antibodies (bottom). Scale bar: 30μm. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/27148974), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] -
Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - HIF-1 alpha binds to TIM-3 promoter & regulates its expression in primary glia.(a) Cell surface expression of TIM-3 analysed in BV2 cells under 20% O2 or 1% O2 for 24 h by flow cytometry using PE-conjugated anti-TIM-3 antibody. Results from 3 independent experiments presented as a representative histogram & the mean fold change (± s.d.) relative to normoxic sample. (b) Mouse primary mixed glial cells incubated under hypoxia or normoxia for 24 h, & the cells examined by immunocytochemistry using an anti-TIM-3 antibody. (c,d) Mouse primary mixed glial cells & primary neuronal cells incubated under hypoxia or normoxia for 24 h, & then RT–PCR used to detect the levels of TIM-3 & actin. Relative transcript levels shown as the mean fold change (± s.d.) from 3 independent experiments (NS, not significant, Student–Newman–Keuls test). (e) Primary mixed glial cells incubated under hypoxia or normoxia for 24 h, & chromatin immunoprecipitation (ChIP) performed w/ anti-HIF-1 alpha or control IgG. Results presented as relative amounts representative of 3 independent experiments. (f) Primary mixed glial cells cultured from HIF-1 alpha +f/+f mice, infected w/ Ad-GFP or Ad-Cre/GFP, transfected w/ TIM-3-luciferase reporter constructs & incubated under hypoxic or normoxic conditions for 24 h. Relative promoter activity is expressed as the ratio of luciferase activity/ beta -galactosidase activity. (g,h) RT–PCR (g) & WB analysis (h) performed under hypoxia or normoxia for 24 h using the indicated primers & antibodies, respectively. The data shown representative of at least 3 independent experiments. The graphs show the % changes in TIM-3 transcript & protein levels in Ad-Cre/GFP- versus Ad-GFP-infected cells under hypoxia. IP, immunoprecipitation. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/25790768), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] -
Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - TIM-3 is highly expressed in hypoxic brain regions of a H/I mouse model.(a) TIM-3 transcript levels were examined in brain tissues from the contralateral cortex (C, boxed region) & ischaemic ipsilateral cortex (I, boxed region) of mouse model 24 h after H/I. The RT–PCR products were quantified with Image J & normalized with respect to the expression of actin. The HIF-1 alpha transcript level represents a positive control for hypoxia. The right panel shows representative TTC staining of three brain sections from the H/I mice. (b) Representative western blot analyses of the TIM-3 & HIF-1 alpha proteins (n=3). Relative levels of TIM-3 are shown as the mean±s.d. from three independent experiments. (c) Contralateral & ipsilateral cortical regions of coronal sections from the H/I mice were subjected to immunohistochemistry using an anti-TIM-3 antibody, & the number of TIM-3-expressing cells per mm2 was counted. (d) Immunohistochemistry was performed on brain sections from the H/I mice using anti-TIM-3 & hypoxyprobe-1 (red, to detect hypoxic regions). Scale bars, 50 μm ( × 20); 50 μm ( × 40). (e,f) Brain cells were isolated from the ipsilateral & contralateral hemispheres of three mice per group, processed for simultaneous detection of TIM-3 plus Iba-1 (e) or GFAP (f), & analysed by FACS. The results are presented as relative TIM-3 levels in the indicated gated populations, as determined from three independent experiments. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/25790768), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] -
Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - Hypoxia & HIF-1 trigger CREB activity.a Knocking down CREB with siRNA abolishes the effect of hypoxia on endogenous rat Gh transcription in GH3 cells as determined by real-time RT-PCR. Immunoblot shows the knockdown efficacy of the CREB siRNA (b) effect of hypoxia (1% O2 for 18 h) on CRE induced luciferase activity. Transfection with 100 nM HIF-1 alpha siRNA for 48 h abolished the effect of hypoxia. Luc/ beta Gal: luciferase: beta -galactosidase ratio. Data are means ± SEM of three experiments & expressed as percentage of each normoxia control. *P < 0.05 (Student’s t test). c Effect of HIF-1 alpha overexpression on CRE luciferase activity. Data are means ± SEM of three experiments & expressed as percentage of mock control. *P < 0.05 (Student’s t test). d Chromatin immunoprecipitation showing increased CREB binding to the endogenous rat Pou1f1 (encoding for Pit-1) promoter in GH3 cells overexpressing HIF-1 alpha. Rabbit IgG was used as a control. Data are arbitrary units from two independent experiments, presented as% of input. **P < 0.01 (Student’s t test). e Immunoblot showing that HIF-1 alpha overexpression increases basal & forskolin (5 µM, 1–6 h)-induced pCREB-Ser133 levels. It also shows that forskolin-induced pCREB-Ser133 remains elevated in HIF-1 alpha overexpressing GH3 cells, while it is back to basal after 6 h in the mock plasmid control transfected cells. f Hypoxia fails to increase rat Gh transcription in GH cells overexpressing CREB-M1 (CREBS133A) a mutant that cannot be phosphorylated by PKA. Data are Gh/TfIIb & presented as fold increase to each normoxia (NX). *P < 0.05 to each normoxia (Student’s t test). Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/32111982), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] -
Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - TIM-3 is highly expressed in hypoxic brain regions of a H/I mouse model.(a) TIM-3 transcript levels were examined in brain tissues from the contralateral cortex (C, boxed region) & ischaemic ipsilateral cortex (I, boxed region) of mouse model 24 h after H/I. The RT–PCR products were quantified with Image J & normalized with respect to the expression of actin. The HIF-1 alpha transcript level represents a positive control for hypoxia. The right panel shows representative TTC staining of three brain sections from the H/I mice. (b) Representative western blot analyses of the TIM-3 & HIF-1 alpha proteins (n=3). Relative levels of TIM-3 are shown as the mean±s.d. from three independent experiments. (c) Contralateral & ipsilateral cortical regions of coronal sections from the H/I mice were subjected to immunohistochemistry using an anti-TIM-3 antibody, & the number of TIM-3-expressing cells per mm2 was counted. (d) Immunohistochemistry was performed on brain sections from the H/I mice using anti-TIM-3 & hypoxyprobe-1 (red, to detect hypoxic regions). Scale bars, 50 μm ( × 20); 50 μm ( × 40). (e,f) Brain cells were isolated from the ipsilateral & contralateral hemispheres of three mice per group, processed for simultaneous detection of TIM-3 plus Iba-1 (e) or GFAP (f), & analysed by FACS. The results are presented as relative TIM-3 levels in the indicated gated populations, as determined from three independent experiments. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/25790768), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody - BSA Free [NB100-134] -
Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody - BSA Free [NB100-134] - Hif1 alpha expression in neonatal & adult testis.(A) Section of testis from 5-day old (P5) male new born pups showing Hif1 alpha expression in MVH+ gonocytes within the seminiferous tubules (top) & negative control images without primary antibodies (bottom). Scale bar: 50μm. (B) Western blot analysis of Hif1 alpha expression in P5 testes (left) compared to extract of the adult brain sub-ventricular zone (SVZ) (right). Loading control ( beta -Actin) is shown below. (C) Section of adult (3 month old) testis showing Hif1 alpha expression in spermatogonia. Scale bar: 30μm. (D) Western blot analysis of whole adult testis. HEK293 cells treated with DFX were used as a positive control & intestinal tissue was used as a negative control. Loading control ( beta -Actin) is shown below. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/27148974), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody - BSA Free [NB100-134] -
Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody - BSA Free [NB100-134] - HIF-1 alpha levels are increased by androgen stimulus & Tip60 overexpression in LNCaP cells. (A) Nuclear & (B) cytosolic HIF-1 alpha levels & (C) images in LNCaP cells & in LNCaP cells overexpressing Tip60, in the absence or presence of androgen (10 nM R1881, 72 h). HIF-1 alpha levels were detected by immunofluorescence using confocal imaging system. Images were acquired with 20x objective. Staining intensity levels in the nucleus & cytosolic region were obtained using Harmony software. Nucleus & cytosol were identified through Hoechst & CellMask staining, respectively. Scale is shown as 100 μm. White dotted frames indicate the section of the image that was enlarged. Values are expressed as mean ± SEM, from three independent culture preparations, each treatment performed in quadruplicate. Two-way ANOVA, Bonferroni post-test & p values comparisons are specified in the figures (* p < 0.05). HIF-1 alpha, hypoxia-inducible factor-1 alpha ; OE, overexpressing; R1881, synthetic androgen. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31671779), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Chromatin Immunoprecipitation: HIF-1 alpha Antibody - BSA Free [NB100-134] -
Chromatin Immunoprecipitation: HIF-1 alpha Antibody - BSA Free [NB100-134] - HIF-1 alpha binds to TIM-3 promoter & regulates its expression in primary glia.(a) Cell surface expression of TIM-3 analysed in BV2 cells under 20% O2 or 1% O2 for 24 h by flow cytometry using PE-conjugated anti-TIM-3 antibody. Results from 3 independent experiments presented as a representative histogram & the mean fold change (± s.d.) relative to normoxic sample. (b) Mouse primary mixed glial cells incubated under hypoxia or normoxia for 24 h, & the cells examined by immunocytochemistry using an anti-TIM-3 antibody. (c,d) Mouse primary mixed glial cells & primary neuronal cells incubated under hypoxia or normoxia for 24 h, & then RT–PCR used to detect the levels of TIM-3 & actin. Relative transcript levels shown as the mean fold change (± s.d.) from 3 independent experiments (NS, not significant, Student–Newman–Keuls test). (e) Primary mixed glial cells incubated under hypoxia or normoxia for 24 h, & chromatin immunoprecipitation (ChIP) performed w/ anti-HIF-1 alpha or control IgG. Results presented as relative amounts representative of 3 independent experiments. (f) Primary mixed glial cells cultured from HIF-1 alpha +f/+f mice, infected w/ Ad-GFP or Ad-Cre/GFP, transfected w/ TIM-3-luciferase reporter constructs & incubated under hypoxic or normoxic conditions for 24 h. Relative promoter activity is expressed as the ratio of luciferase activity/ beta -galactosidase activity. (g,h) RT–PCR (g) & WB analysis (h) performed under hypoxia or normoxia for 24 h using the indicated primers & antibodies, respectively. data shown representative of at least 3 independent experiments. The graphs show the % changes in TIM-3 transcript & protein levels in Ad-Cre/GFP- versus Ad-GFP-infected cells under hypoxia. IP, immunoprecipitation. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/25790768), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody - BSA Free [NB100-134] -
Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody - BSA Free [NB100-134] - NgBR overexpression (OE) suppresses cell proliferation through enhancing mitochondria-ER communication & reducing phosphorylation of IP3R3. (A) The transfection efficiency of NgBR OE plasmid was verified at 48 h post transfection. n = 3 (B) Representative images from three experiments show phosphorylation of endogenous IP3R3 (Akt substrate) in the immunoprecipitates of IP3R3 from crude mitochondrial extracts. (C) Representative confocal microscopy images show co-staining of Rhod-2 AM (red) & MitoTracker Green (green). Results were calculated as relative AFU using ImageJ. n = 20 pictures/group from four independent experiments. Scale bar = 20 µm. (D) Representative line & bar graphs of OCR of control & NgBR OE cells under normoxia. n = 15–16 wells from three individual experiments. (E) Representative confocal microscopy images showing staining of HIF-1 alpha (red) & DAPI (nuclear stain; blue). Percentage of HIF-1 alpha -positive nuclei was calculated using FV10-ASW3.1 software. n > 25 pictures/group from three separate experiments. Scale bar = 40 µm. (F) Cell proliferation was assessed by evaluating PCNA expression, n = 4. * p < 0.05, ** p < 0.01. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31083380), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry: HIF-1 alpha Antibody - BSA Free [NB100-134] -
Immunohistochemistry: HIF-1 alpha Antibody - BSA Free [NB100-134] - E2 enhances neutrophil apoptosis through suppressing the expression of hif1 alpha & c-myb under physiological conditions.A E2 decreased neutrophils in AB zebrafish embryos. (t-test, ***p < 0.001, n > 20). B May–Grunwald–Giemsa staining of whole KM blood cells in 6-month-old AB zebrafish after 4 days of E2 treatment (t-test, ***p < 0.001, n = 12). Red arrowheads, blue asterisks, black arrowheads & yellow lightning indicates neutrophils, precursors, lymphocytes & macrophages, respectively. C Staining of hif1 alpha with antibody with or without E2 treatment. D E2 exposure decreased c-myb in the CHT, as determined by WISH. E qPCR quantification of decreased c-myb expression in lyz:Dsred+ cells by E2 (t-test, mean ± SEM. ***p < 0.001, **p < 0.01, *p < 0.05, n > 20). F Effect of E2 on neutrophil proliferation in AB zebrafish embryos. (one-way ANOVA (LSD). ns, no significance, n > 10). G E2 promotes the apoptosis of myeloid lineage cells in AB zebrafish embryos (one-way ANOVA (LSD) **p < 0.01. n > 10). Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/35842445), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] -
Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - IGFBP5 inhibits HIF1 alpha expression through the MAPK-ERK signaling pathwayA. & B. Western blots using specific antibodies for the phosphorylation state of IGF1R, ERK1/2, & p38-MAPK from A375 IGFBP5 OE & control cells. Phosphorylation of IGF1R, ERK1/2, & p38-MAPK was decreased in A375 cells transfected with IGFBP5. GAPDH was used as a loading control. C. Overexpression of IGFBP5 inhibited HIF1 alpha expression visualized by western blots of A375 cells. The arrow points to the band of HIF1 alpha. D. Assessment of the reduced gene expression levels of VEGFA & MMP9, downstream genes regulated by HIF1 alpha, in A375 IGFBP5 OE compared to control cells by qRT-PCR analysis. Data were shown for the mean ± SD form three independent experiments. P values based on two-side Student t-test comparing A375 IGFBP5 OE tumor cells & vector control cells. *, P < 0.05. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/26010068), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] -
Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - WNT11 regulates MMPs activities.(A–C) (Top panels): Serum-free medium was conditioned for 24 hrs by the indicated cells, concentrated 20-fold & assayed by gelatin zymography. Gelatinolytic activity is indicated by clear zones against a dark background of stained substrate. (Bottom): Whole cell extracts were immunoblotted with indicated antibodies. (A) Overexpression of Wnt11 in EMSC or BT473 cells enhances activity of MMP-9 & MMP-2. (B) Impaired activity of MMP-9 & MMP-2 in MDA-MB-231 cells (left) or EMSCs (right) stably expressing Wnt11 shRNAs & treated with DMOG. (C) WNT11 is required for MMP-9 & MMP-2 activity in MDA-MB-231 cells (left) or EMSCs (right) under normoxic & hypoxic culture conditions. (D) WNT11 regulates MMP2 protein in media. (Top): conditioned media from indicated cells & treatments. (Bottom): whole cell lysates were immunoblotted with indicated antibodies. (E) Recombinant WNT11 induces both MMP-2 protein & MMP-2 activity in media. (Top panels): Gelatin zymography & immunoblot of serum-free medium conditioned for the indicated times after recombinant WNT11 (r-WNT11) treatment. (Bottom): Whole cell lysates were immunoblotted with indicated antibodies. (F) MMP-2 inhibitor attenuated induced migration by WNT11. MDA-MB-231 cells infected with lentiviruses for stable expression of Wnt11 or GFP (n = 4) were incubated with either vehicle or 1 μM of ARP100. Media in the lower compartment had same concentration of DMSO or inhibitor. Values are mean ± s.e.m. *p < 0.05, **p < 0.01. For panels (A–D), HIF-1 alpha & HIF-2 alpha were shown as a marker of hypoxia, WNT11 normalized to alpha -Tubulin was shown. Image collected & cropped by CiteAb from the following publication (https://www.nature.com/articles/srep21520), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] -
Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - ER-INP binds to PHD2, inhibits hydroxylation of HIF-1 alpha & increases HIF-1 alpha accumulation. (A) Co-immunoprecipitation assay. HEK293 cells were transfected with control vector or intrabody ER-INP for 48 hours & subsequently lysed. Co-immunoprecipitation & western blot assays were performed on the cell lysis. ER-INP recognized & bound to PHD2 in HEK293 cells (n = 3). (B) Western blot analysis to measure the effect of ER-INP on HIF-1 alpha & its hydroxylation level in transfected HEK293 & RAW264.7 cells pre-treated with MG132 (upper panel) & the protein ratio to beta -actin loading control by ImageJ densitometry analysis (lower panel; n = 3). (C) Immunofluorescence assay. Expression of ER-INP increases HIF-1 alpha accumulation in RAW264.7 cells. Cells were stained with anti-HIF-1 alpha antibody & DAPI & then visualized & photographed under immunofluorescence microscopy (left), & mean fluorescence intensity of HIF-1 alpha versus the mean fluorescence intensity of nuclear DAPI staining is displayed (right). Data represent the mean ± SD of 3 independent slides. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31413262), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] -
Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - HIF-1 alpha binds to TIM-3 promoter & regulates its expression in primary glia.(a) Cell surface expression of TIM-3 analysed in BV2 cells under 20% O2 or 1% O2 for 24 h by flow cytometry using PE-conjugated anti-TIM-3 antibody. Results from 3 independent experiments presented as a representative histogram & the mean fold change (± s.d.) relative to normoxic sample. (b) Mouse primary mixed glial cells incubated under hypoxia or normoxia for 24 h, & the cells examined by immunocytochemistry using an anti-TIM-3 antibody. (c,d) Mouse primary mixed glial cells & primary neuronal cells incubated under hypoxia or normoxia for 24 h, & then RT–PCR used to detect the levels of TIM-3 & actin. Relative transcript levels shown as the mean fold change (± s.d.) from 3 independent experiments (NS, not significant, Student–Newman–Keuls test). (e) Primary mixed glial cells incubated under hypoxia or normoxia for 24 h, & chromatin immunoprecipitation (ChIP) performed w/ anti-HIF-1 alpha or control IgG. Results presented as relative amounts representative of 3 independent experiments. (f) Primary mixed glial cells cultured from HIF-1 alpha +f/+f mice, infected w/ Ad-GFP or Ad-Cre/GFP, transfected w/ TIM-3-luciferase reporter constructs & incubated under hypoxic or normoxic conditions for 24 h. Relative promoter activity is expressed as the ratio of luciferase activity/ beta -galactosidase activity. (g,h) RT–PCR (g) & WB analysis (h) performed under hypoxia or normoxia for 24 h using the indicated primers & antibodies, respectively. The data shown representative of at least 3 independent experiments. The graphs show the % changes in TIM-3 transcript & protein levels in Ad-Cre/GFP- versus Ad-GFP-infected cells under hypoxia. IP, immunoprecipitation. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/25790768), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] -
Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - Regulation of catabolic genes by HIF-1 alpha. (A) Protein levels of Hif1a, & mRNA levels of Mmp13, & Hif2a in WT mouse primary chondrocytes transduced with GFP or HIF1A adenoviral vectors under the hypoxic condition (3% O2). GFP or HIF1A was transduced at a multiplicity of infection (MOI) of 100. The cells were treated with or without 10 ng/mL IL-1 beta for 2 days. Bars show the mean ± SD of three samples per group. *P < 0.05. (B) Protein levels of Hif1a, & mRNA levels of Mmp13, & Hif2a in WT mouse primary chondrocytes transfected with siRNA against GFP or Hif1a under the hypoxic condition. The cells were treated with or without 10 ng/mL IL-1 beta for 2 days. Bars show the mean ± SD of three samples per group. *P < 0.05. (C) Protein levels of Hif1a, & mRNA levels of Mmp13, & Hif2a in Hif1afl/fl primary chondrocytes transduced with GFP or Cre adenoviral vectors under the hypoxic condition. GFP or Cre was transduced at a MOI of 100. The cells were treated with or without 10 ng/mL IL-1 beta for 2 days. Bars show the mean ± SD of three samples per group. *P < 0.05. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/32214220), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] -
Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - NSCs Reduce Succinate Levels & Reprogram the Metabolism of Type 1 Pro-inflammatory Mφ toward Oxidative Phosphorylation In Vitro(A) Experimental setup for in vitro MφLPS co-cultures with iNSCs/NSCs.(B & C) Gene expression microarrays of MφLPS-iNSCs/NSCs. (B) Venn diagram of differentially expressed genes (adjusted p value < 0.1). (C) Heatmap of genes differentially expressed (adjusted p value < 0.1) in MφLPS-iNSCs or MφLPS-NSCs.(D & E) qRT-PCR independent validation of differentially expressed inflammatory genes as in (C). (D) Expression of genes related to type 1 inflammatory (E) & anti-inflammatory Mφ phenotypes relative to Actb. Data are mean fold change (±SEM) versus MφLPS from n ≥ 3 independent replicates per condition.(F) qRT-PCR of BV2LPS-iNSCs/NSCs (±SEM) from n ≥ 3 independent experiments per condition. BV2 & BV2LPS are shown as controls.(G & H) Extracellular flux (XF) assay of the oxygen consumption rate (OCR) (G) & extracellular acidification rate (ECAR) (H) in MφLPS-iNSCs/NSCs. Data were normalized on total protein content & are expressed as mean values (±SEM) from n ≥ 3 independent experiments per condition.(I & J) Levels of significantly changed extracellular (EXTRA_Metab, I) & intracellular (INTRA_Metab, J) metabolites in MφLPS versus Mφ at 25 hr. Data are mean a.u. (±SEM) from n ≥ 2 independent experiments per condition.(K & L) Hif-1 alpha (K), PKM2 (K), & IL-1 beta (L) expression levels relative to beta -actin. Data are mean fold change versus MφLPS (±SEM) from n ≥ 3 independent experiments per condition.∗p ≤ 0.05 & ∗∗p ≤ 0.01 versus MφLPS. See also Tables S2 & S3. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/29478844), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] -
Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - Enhanced expression of hypoxia-induced survival factors & angiogenic growth factors in hASC L-spheroids.(A) The light source used was LED (660 nm) designed to fit over a microplate (12.5 × 8.5 cm) for cell culture. (B) Formation of hASC L-spheroids. hASCs morphology on non–tissue culture–treated 24-well plates at day 3. Scale bar = 500 μm. (C) Western blot analysis & quantification of HIF1-alpha in hASCs cultured as spheroids, L-spheroids & monolayers (*p < 0.01, compared to the L-spheroid group). (D) Angiogenesis-related protein analysis of L-spheroids (*, p < 0.05, compared to the spheroid group, t-test, n = 3 in each group). (E) ELISA measurement of spheroids cultured for 3 days. Concentrations of VEGF are presented as pg-corrected for 104 cells. (*, p < 0.05, compared with spheroid 6J/cm2 group, t-test, n = 3 in each group). Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/26065900), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Applications for HIF-1 alpha Antibody - BSA Free
Chromatin Immunoprecipitation Sequencing
Gel Super Shift Assays
Immunohistochemistry
Immunohistochemistry-Frozen
Immunohistochemistry-Paraffin
Immunoprecipitation
Simple Western
Western Blot
Reviewed Applications
Read 14 reviews rated 4.8 using NB100-134 in the following applications:
Formulation, Preparation, and Storage
Purification
Formulation
Format
Preservative
Concentration
Shipping
Stability & Storage
Background: HIF-1 alpha/HIF1A
HIF-1 or hypoxia inducible factor 1 (predicted molecular weight 93kDa), is a transcription factor commonly referred to as a "master regulator of the hypoxic response" for its central role in the regulation of cellular adaptations to hypoxia. In its active form under hypoxic conditions, HIF-1 is stabilized by the formation of a heterodimer of HIF-1 alpha and ARNT/HIF-1 beta subunits. Nuclear HIF-1 engages p300/CBP for binding to hypoxic response elements (HREs). This process induces transcription and regulation of genes including EPO, VEGF, iNOS2, ANGPT1 and OCT4 (4,5).
Under normoxic conditions, the HIF-1 alpha subunit is rapidly targeted and degraded by the ubiquitin proteasome system. This process is mediated by prolyl hydroxylase domain enzymes (PHDs), which catalyze the hydroxylation of key proline residues (Pro-402 and Pro-564) within the oxygen-dependent degradation domain of HIF-1 alpha. Once hydroxylated, HIF-1 alpha binds the von Hippel-Lindau tumor suppressor protein (pVHL) for subsequent ubiquitination and proteasomal degradation (4). pVHL dependent regulation of HIF-1 alpha plays a role in normal physiology and disease states. Regulation of HIF-1 alpha by pVHL is critical for the suppressive function of FoxP3+ regulatory Tcells (6). Repression of pVHL expression in chronic lymphocytic leukemia (CLL) B cells leads to HIF-1 alpha stabilization and increased VEGF secretion (7).
References
1. Semenza, G. L., Agani, F., Feldser, D., Iyer, N., Kotch, L., Laughner, E., & Yu, A. (2000). Hypoxia, HIF-1, and the pathophysiology of common human diseases. Advances in Experimental Medicine and Biology.
2. Muz, B., de la Puente, P., Azab, F., & Azab, A. K. (2015). The role of hypoxia in cancer progression, angiogenesis, metastasis, and resistance to therapy. Hypoxia. https://doi.org/10.2147/hp.s93413
3. Huang, Y., Lin, D., & Taniguchi, C. M. (2017). Hypoxia inducible factor (HIF) in the tumor microenvironment: friend or foe? Science China Life Sciences. https://doi.org/10.1007/s11427-017-9178-y
4. Koyasu, S., Kobayashi, M., Goto, Y., Hiraoka, M., & Harada, H. (2018). Regulatory mechanisms of hypoxia-inducible factor 1 activity: Two decades of knowledge. Cancer Science. https://doi.org/10.1111/cas.13483
5. Dengler, V. L., Galbraith, M. D., & Espinosa, J. M. (2014). Transcriptional regulation by hypoxia inducible factors. Critical Reviews in Biochemistry and Molecular Biology. https://doi.org/10.3109/10409238.2013.838205
6. Lee, J. H., Elly, C., Park, Y., & Liu, Y. C. (2015). E3Ubiquitin Ligase VHL Regulates Hypoxia-Inducible Factor-1 alpha to Maintain Regulatory T Cell Stability and Suppressive Capacity. Immunity. https://doi.org/10.1016/j.immuni.2015.05.016
7. Ghosh, A. K., Shanafelt, T. D., Cimmino, A., Taccioli, C., Volinia, S., Liu, C. G.,... Kay, N. E. (2009). Aberrant regulation of pVHL levels by microRNA promotes the HIF/VEGF axis in CLL B cells. Blood. https://doi.org/10.1182/blood-2008-10-185686
Long Name
Alternate Names
Gene Symbol
UniProt
Additional HIF-1 alpha/HIF1A Products
Product Documents for HIF-1 alpha Antibody - BSA Free
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for HIF-1 alpha Antibody - BSA Free
This product is for research use only and is not approved for use in humans or in clinical diagnosis. Primary Antibodies are guaranteed for 1 year from date of receipt.
Citations for HIF-1 alpha Antibody - BSA Free
Customer Reviews for HIF-1 alpha Antibody - BSA Free (14)
Have you used HIF-1 alpha Antibody - BSA Free?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Application: Western BlotSample Tested: Zebrafish embryo lysatesSpecies: ZebrafishVerified Customer | Posted 02/07/2020
-
Application: Western BlotSample Tested: Human MD-MB-231 cells and bt549Species: HumanVerified Customer | Posted 10/28/2018
-
Application: Western BlotSample Tested: 20 ug whole cell lysateSpecies: HumanVerified Customer | Posted 04/30/2018
-
Application: Western BlotSample Tested: HCT116 whole cell lysateSpecies: HumanVerified Customer | Posted 03/22/2017
-
Application: Immunohistochemistry-ParaffinSample Tested: Zebrafish retina with mouse tissue as controlSpecies: OtherVerified Customer | Posted 08/02/2016HIF-1alpha staining on Zebrafish retina
-
Application: Western BlotSample Tested: Retinal and Choroidal primary endotheliaSpecies: HumanVerified Customer | Posted 07/07/2016Endogenous HIF-1alpha in Retinal and Choroidal Endothelial Cells
-
Application: Western BlotSample Tested: Vascular Smooth Muscle CellsSpecies: RatVerified Customer | Posted 07/23/2015
-
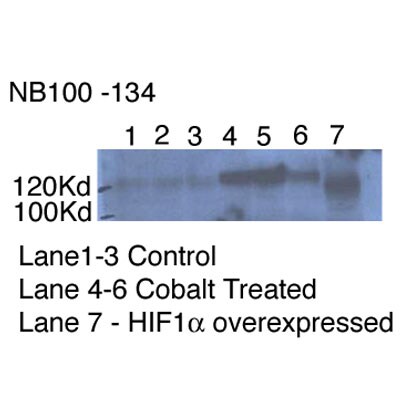
Application: Western BlotSample Tested: MDA-MB-231 Cell LysateSpecies: HumanVerified Customer | Posted 06/01/2015overexpression and endogenous HIF1a & HIF2a
-
Application: ImmunofluorescenceSample Tested: ARPE-19 cellsSpecies: HumanVerified Customer | Posted 05/14/2015hARPE-19 cells
-
Application: Western BlotSample Tested: HEK293 cell lysateSpecies: HumanVerified Customer | Posted 09/18/2014WB of NB100-134
-
Application: Western BlotSample Tested: Clear cell renal cell carcinoma samplesSpecies: HumanVerified Customer | Posted 08/30/2014Hif1a expression in ccRCC samples
-
Application: Immunohistochemistry-ParaffinSample Tested: Canine corpus luteumSpecies: OtherVerified Customer | Posted 01/30/2014HIF1A staining in the canine CL on days 10 to 70 after ovulation. PC = positive control (human placenta). NC = negative control
-
Application: Western BlotSample Tested: PC12 nuclear extract, Sample Amount: 50ugSpecies: RatVerified Customer | Posted 02/05/2012
-
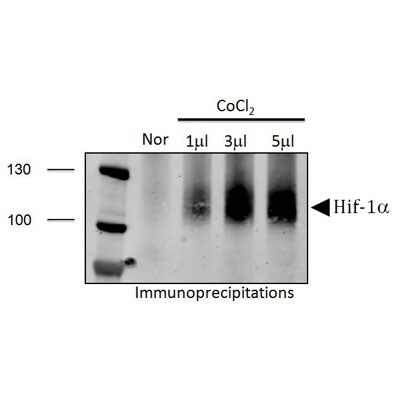
Application: ImmunoprecipitationSample Tested: PC-3 CellsSpecies: HumanVerified Customer | Posted 10/27/2011
There are no reviews that match your criteria.
Protocols
View specific protocols for HIF-1 alpha Antibody - BSA Free (NB100-134):
Immunohistochemistry-Paraffin Embedded Sections
Antigen Unmasking:
Bring slides to a boil in 10 mM sodium citrate buffer (pH 6.0) then maintain at a sub-boiling temperature for 10 minutes. Cool slides on bench-top for 30 minutes.
Staining:
1. Wash sections in deionized water three times for 5 minutes each.
2. Wash sections in wash buffer for 5 minutes.
3. Block each section with 100-400 ul blocking solution for 1 hour at room temperature.
4. Remove blocking solution and add 100-400 ul diluted primary antibody. Incubate overnight at 4 C.
5. Remove antibody solution and wash sections in wash buffer three times for 5 minutes each.
6. Add 100-400 uL biotinylated diluted secondary antibody. Incubate 30 minutes at room temperature.
7. Remove secondary antibody solution and wash sections three times with wash buffer for 5 minutes each.
8. Add 100-400 uL Streptavidin-HRP reagent to each section and incubate for 30 minutes at room temperature.
9. Wash sections three times in wash buffer for 5 minutes each.
10. Add 100-400 uL DAB substrate to each section and monitor staining closely.
11. As soon as the sections develop, immerse slides in deionized water.
12. Counterstain sections in hematoxylin.
13. Wash sections in deionized water two times for 5 minutes each.
14. Dehydrate sections.
15. Mount coverslips.
1. HIF-1alpha is largely undetectable in cells or tissues grown under normoxic conditions. It is stabilized only at O2 concentrations below 5% or with treatment using certain agents (CoCl2, DFO, etc.), therefore proper sample preparation is critical. We recommend lysing cells quickly and directly into the Laemmli sample buffer with DTT or BME.
2. Since stabilized HIF-1alpha translocates to the nucleus, using nuclear extracts is recommended for western blot analysis.
3. Positive and negative controls should always be run side by side in a Western blot to accurately identify the protein band upregulated in the hypoxic sample. (HeLa Hypoxic/Normoxic Cell Lysate: NBP2-36452; HeLa Hypoxic (CoCl2)/Normoxic Lysate: NBP2-36450)
4. To accurately compare treated and untreated samples and to ensure equal loading of samples the expression of a loading control should be evaluated. (alpha Tubulin Antibody (DM1A): NB100-690)
5. Unprocessed HIF-1alpha is ~95 kDa, while the fully post-translationally modified form is ~116 kDa, or larger.
6. HIF-1alpha may form a heterodimer with HIF-1beta (Duan, et al. Circulation. 2005; 111:2227-2232.). However, this is not typically seen under denaturing conditions.
7. Depending on the sample and treatment, a single band or a doublet may be present.
Western Blot Protocol
1. Load samples of treated and untreated cell lysates, 10-40 mg of total protein per lane on a 7.5%polyacrylamide
gel (SDS-PAGE). Alternatively, gradient gels can be used for better resolution of lower molecular weight loading
controls.
2. Resolve proteins by electrophoresis as required.
3. Transfer proteins to 0.45 mm PVDF membrane for 1 hour at 100V or equivalent.
4. Stain the blot using Ponceau S for 1-2 minutes to confirm efficient protein transfer onto the membrane.
5. Rinse the blot in distilled water to remove excess stain and mark the lanes and locations of molecular weight
markers using a pencil.
6. Block the membrane using 5% non-fat dry milk in TBST (0.1% Tween) for 1 hour.
7. Dilute the mouse anti-HIF-1 alpha primary antibody (NB100-105) at 2ug/ml in blocking solution and incubate 1
hour at room temperature or overnight at 4C.
8. Wash the membrane 3X 5 min in TBST.
9. Incubate in the appropriate diluted mouse-IgG HRP-conjugated secondary antibody in blocking solution (as per
manufacturer's instructions) for 1 hour at room temperature.
10. Wash the membrane 3X 5 min in TBST.
11. Incubate with ECL detection reagent (Supersignal West Pico Plus, or more sensitive) for 5 min.
12. Image the blot. That may require up to 5min of exposure due to weak signal.
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ChIP Protocol Video
- Chromatin Immunoprecipitation (ChIP) Protocol
- Chromatin Immunoprecipitation Protocol
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ELISA Sample Preparation & Collection Guide
- ELISA Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- How to Run an R&D Systems DuoSet ELISA
- How to Run an R&D Systems Quantikine ELISA
- How to Run an R&D Systems Quantikine™ QuicKit™ ELISA
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- ISH-IHC Protocol for Chromogenic Detection on Formalin Fixed Paraffin Embedded (FFPE) Tissue
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Immunoprecipitation Protocol
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Quantikine HS ELISA Kit Assay Principle, Alkaline Phosphatase
- Quantikine HS ELISA Kit Principle, Streptavidin-HRP Polymer
- R&D Systems Quality Control Western Blot Protocol
- Sandwich ELISA (Colorimetric) – Biotin/Streptavidin Detection Protocol
- Sandwich ELISA (Colorimetric) – Direct Detection Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: ELISA
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
FAQs for HIF-1 alpha Antibody - BSA Free
-
Q: Are there temperature requirements for storage?
A: We recommend short-term storage at 4C, and any long term storage at -20C. We recommend you aliquot any long-term storage and only remove what is needed for testing, as we recommend avoiding freeze-thaw cycles.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Does HIF1 alpha antibody (NB100-134) detect in the cytoplasm or the nucleus?
A: HIF1 alpha protein can be detected in both the cytoplasm and nucleus depending on the oxygen exposure of your tissues or cells. Cell that are more normoxic will have more signal in the cytoplasm while cells that are in hypoxic conditions will have a higher signal in the nucleus. Please see our publications for item NB100-134 for additional information regarding staining in IHC and ICC with this antibody.
-
Q: How much should I use for ChIP, 10E7 cells?
A: For ChIP, we recommend a dilution of 25 ul for 15 million cells. For 1 x 10^7 cells, you can use approximately 16-18 ul of this antibody.
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I am going to do a Western blot to detect HIF-1a protein on HTR8/SVneo trophoblast cells. I have used NB100-134 HIF-1 a antibody for immunostaining and will use the same Ab for Western. Now my questions are 1. What should I use for blocking? 2. Can I use HIF-1 alpha recombinant protein (H00003091-P01) as positive control? 3. What can I use as loading control? 4. Can I use DAB as substrate or anything else?
A: 1. You can use standard blocking solution which is 5% non-fat milk in TBST. 2. We recommend using Cos-7 Hypoxia Induced Nuclear Extract Kit (catalog #NB800-PC26) as the positive control for this antibody. 3. Since HIF-1 is a nuclear protein, you can use either alpha-Tubulin or beta-Tubulin as your loading control. 4. You can use DAB as your detection system.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I ordered this antibody but do not have a freezer to store it -20 degrees Celsius. Is it possible to store it for a short time (few days or weeks) at 4 degrees Celsius?
A: NB100-134 does contain Sodium Azide and should be fine at 4 degrees Celsius for about a week. We do recommend -20 degrees Celsius for storage and cannot guarantee this product will not degrade if kept at 4 degrees Celsius for more than a week.
-
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is the species reactivity of this antibody to Guinea pig based on computational homology?
A: The species reactivity reported for Guinea pig is based on published data. Here is the link for your reference. If you go through this paper carefully, on Figure 6, HIF1 expression has been tested across species including Cavia porcellus (Guinea pig).
-
Q: Our customer would like to know the shelf life of your HIF-1 alpha Antibody (Cat. # NB100-134). We would appreciate if you can confirm this information for me as soon as possible. Moreover, I was wondering whether your antibody products have a general shelf life?
A: Unless otherwise stated on our website, our antibodies are guaranteed for 1 year from the date of receipt, whist peptides and proteins are guaranteed for three months from the date of receipt. Novus guarantees that our products will work in the applications and species listed on our website and on our product datasheets. If a Novus product fails to meet this criteria, our technical service team will work with you to troubleshoot the issue. If this process is not successful, Novus will provide you with a free replacement or a 100% refund for the product.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the difference in the following two HIF-1 alpha antibodies? Catalog # NB100-105 and NB100-134
A: NB100-105 is a mouse monoclonal antibody, whereas NB100-134 is a rabbit polyclonal. They both use the same immunogen.
-
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Are there temperature requirements for storage?
A: We recommend short-term storage at 4C, and any long term storage at -20C. We recommend you aliquot any long-term storage and only remove what is needed for testing, as we recommend avoiding freeze-thaw cycles.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Does HIF1 alpha antibody (NB100-134) detect in the cytoplasm or the nucleus?
A: HIF1 alpha protein can be detected in both the cytoplasm and nucleus depending on the oxygen exposure of your tissues or cells. Cell that are more normoxic will have more signal in the cytoplasm while cells that are in hypoxic conditions will have a higher signal in the nucleus. Please see our publications for item NB100-134 for additional information regarding staining in IHC and ICC with this antibody.
-
Q: How much should I use for ChIP, 10E7 cells?
A: For ChIP, we recommend a dilution of 25 ul for 15 million cells. For 1 x 10^7 cells, you can use approximately 16-18 ul of this antibody.
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I am going to do a Western blot to detect HIF-1a protein on HTR8/SVneo trophoblast cells. I have used NB100-134 HIF-1 a antibody for immunostaining and will use the same Ab for Western. Now my questions are 1. What should I use for blocking? 2. Can I use HIF-1 alpha recombinant protein (H00003091-P01) as positive control? 3. What can I use as loading control? 4. Can I use DAB as substrate or anything else?
A: 1. You can use standard blocking solution which is 5% non-fat milk in TBST. 2. We recommend using Cos-7 Hypoxia Induced Nuclear Extract Kit (catalog #NB800-PC26) as the positive control for this antibody. 3. Since HIF-1 is a nuclear protein, you can use either alpha-Tubulin or beta-Tubulin as your loading control. 4. You can use DAB as your detection system.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I ordered this antibody but do not have a freezer to store it -20 degrees Celsius. Is it possible to store it for a short time (few days or weeks) at 4 degrees Celsius?
A: NB100-134 does contain Sodium Azide and should be fine at 4 degrees Celsius for about a week. We do recommend -20 degrees Celsius for storage and cannot guarantee this product will not degrade if kept at 4 degrees Celsius for more than a week.
-
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is the species reactivity of this antibody to Guinea pig based on computational homology?
A: The species reactivity reported for Guinea pig is based on published data. Here is the link for your reference. If you go through this paper carefully, on Figure 6, HIF1 expression has been tested across species including Cavia porcellus (Guinea pig).
-
Q: Our customer would like to know the shelf life of your HIF-1 alpha Antibody (Cat. # NB100-134). We would appreciate if you can confirm this information for me as soon as possible. Moreover, I was wondering whether your antibody products have a general shelf life?
A: Unless otherwise stated on our website, our antibodies are guaranteed for 1 year from the date of receipt, whist peptides and proteins are guaranteed for three months from the date of receipt. Novus guarantees that our products will work in the applications and species listed on our website and on our product datasheets. If a Novus product fails to meet this criteria, our technical service team will work with you to troubleshoot the issue. If this process is not successful, Novus will provide you with a free replacement or a 100% refund for the product.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the difference in the following two HIF-1 alpha antibodies? Catalog # NB100-105 and NB100-134
A: NB100-105 is a mouse monoclonal antibody, whereas NB100-134 is a rabbit polyclonal. They both use the same immunogen.
-
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Are there temperature requirements for storage?
A: We recommend short-term storage at 4C, and any long term storage at -20C. We recommend you aliquot any long-term storage and only remove what is needed for testing, as we recommend avoiding freeze-thaw cycles.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Does HIF1 alpha antibody (NB100-134) detect in the cytoplasm or the nucleus?
A: HIF1 alpha protein can be detected in both the cytoplasm and nucleus depending on the oxygen exposure of your tissues or cells. Cell that are more normoxic will have more signal in the cytoplasm while cells that are in hypoxic conditions will have a higher signal in the nucleus. Please see our publications for item NB100-134 for additional information regarding staining in IHC and ICC with this antibody.
-
Q: How much should I use for ChIP, 10E7 cells?
A: For ChIP, we recommend a dilution of 25 ul for 15 million cells. For 1 x 10^7 cells, you can use approximately 16-18 ul of this antibody.
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I am going to do a Western blot to detect HIF-1a protein on HTR8/SVneo trophoblast cells. I have used NB100-134 HIF-1 a antibody for immunostaining and will use the same Ab for Western. Now my questions are 1. What should I use for blocking? 2. Can I use HIF-1 alpha recombinant protein (H00003091-P01) as positive control? 3. What can I use as loading control? 4. Can I use DAB as substrate or anything else?
A: 1. You can use standard blocking solution which is 5% non-fat milk in TBST. 2. We recommend using Cos-7 Hypoxia Induced Nuclear Extract Kit (catalog #NB800-PC26) as the positive control for this antibody. 3. Since HIF-1 is a nuclear protein, you can use either alpha-Tubulin or beta-Tubulin as your loading control. 4. You can use DAB as your detection system.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I ordered this antibody but do not have a freezer to store it -20 degrees Celsius. Is it possible to store it for a short time (few days or weeks) at 4 degrees Celsius?
A: NB100-134 does contain Sodium Azide and should be fine at 4 degrees Celsius for about a week. We do recommend -20 degrees Celsius for storage and cannot guarantee this product will not degrade if kept at 4 degrees Celsius for more than a week.
-
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is the species reactivity of this antibody to Guinea pig based on computational homology?
A: The species reactivity reported for Guinea pig is based on published data. Here is the link for your reference. If you go through this paper carefully, on Figure 6, HIF1 expression has been tested across species including Cavia porcellus (Guinea pig).
-
Q: Our customer would like to know the shelf life of your HIF-1 alpha Antibody (Cat. # NB100-134). We would appreciate if you can confirm this information for me as soon as possible. Moreover, I was wondering whether your antibody products have a general shelf life?
A: Unless otherwise stated on our website, our antibodies are guaranteed for 1 year from the date of receipt, whist peptides and proteins are guaranteed for three months from the date of receipt. Novus guarantees that our products will work in the applications and species listed on our website and on our product datasheets. If a Novus product fails to meet this criteria, our technical service team will work with you to troubleshoot the issue. If this process is not successful, Novus will provide you with a free replacement or a 100% refund for the product.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the difference in the following two HIF-1 alpha antibodies? Catalog # NB100-105 and NB100-134
A: NB100-105 is a mouse monoclonal antibody, whereas NB100-134 is a rabbit polyclonal. They both use the same immunogen.
-
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Are there temperature requirements for storage?
A: We recommend short-term storage at 4C, and any long term storage at -20C. We recommend you aliquot any long-term storage and only remove what is needed for testing, as we recommend avoiding freeze-thaw cycles.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Does HIF1 alpha antibody (NB100-134) detect in the cytoplasm or the nucleus?
A: HIF1 alpha protein can be detected in both the cytoplasm and nucleus depending on the oxygen exposure of your tissues or cells. Cell that are more normoxic will have more signal in the cytoplasm while cells that are in hypoxic conditions will have a higher signal in the nucleus. Please see our publications for item NB100-134 for additional information regarding staining in IHC and ICC with this antibody.
-
Q: How much should I use for ChIP, 10E7 cells?
A: For ChIP, we recommend a dilution of 25 ul for 15 million cells. For 1 x 10^7 cells, you can use approximately 16-18 ul of this antibody.
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I am going to do a Western blot to detect HIF-1a protein on HTR8/SVneo trophoblast cells. I have used NB100-134 HIF-1 a antibody for immunostaining and will use the same Ab for Western. Now my questions are 1. What should I use for blocking? 2. Can I use HIF-1 alpha recombinant protein (H00003091-P01) as positive control? 3. What can I use as loading control? 4. Can I use DAB as substrate or anything else?
A: 1. You can use standard blocking solution which is 5% non-fat milk in TBST. 2. We recommend using Cos-7 Hypoxia Induced Nuclear Extract Kit (catalog #NB800-PC26) as the positive control for this antibody. 3. Since HIF-1 is a nuclear protein, you can use either alpha-Tubulin or beta-Tubulin as your loading control. 4. You can use DAB as your detection system.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I ordered this antibody but do not have a freezer to store it -20 degrees Celsius. Is it possible to store it for a short time (few days or weeks) at 4 degrees Celsius?
A: NB100-134 does contain Sodium Azide and should be fine at 4 degrees Celsius for about a week. We do recommend -20 degrees Celsius for storage and cannot guarantee this product will not degrade if kept at 4 degrees Celsius for more than a week.
-
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is the species reactivity of this antibody to Guinea pig based on computational homology?
A: The species reactivity reported for Guinea pig is based on published data. Here is the link for your reference. If you go through this paper carefully, on Figure 6, HIF1 expression has been tested across species including Cavia porcellus (Guinea pig).
-
Q: Our customer would like to know the shelf life of your HIF-1 alpha Antibody (Cat. # NB100-134). We would appreciate if you can confirm this information for me as soon as possible. Moreover, I was wondering whether your antibody products have a general shelf life?
A: Unless otherwise stated on our website, our antibodies are guaranteed for 1 year from the date of receipt, whist peptides and proteins are guaranteed for three months from the date of receipt. Novus guarantees that our products will work in the applications and species listed on our website and on our product datasheets. If a Novus product fails to meet this criteria, our technical service team will work with you to troubleshoot the issue. If this process is not successful, Novus will provide you with a free replacement or a 100% refund for the product.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the difference in the following two HIF-1 alpha antibodies? Catalog # NB100-105 and NB100-134
A: NB100-105 is a mouse monoclonal antibody, whereas NB100-134 is a rabbit polyclonal. They both use the same immunogen.
-
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Are there temperature requirements for storage?
A: We recommend short-term storage at 4C, and any long term storage at -20C. We recommend you aliquot any long-term storage and only remove what is needed for testing, as we recommend avoiding freeze-thaw cycles.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Does HIF1 alpha antibody (NB100-134) detect in the cytoplasm or the nucleus?
A: HIF1 alpha protein can be detected in both the cytoplasm and nucleus depending on the oxygen exposure of your tissues or cells. Cell that are more normoxic will have more signal in the cytoplasm while cells that are in hypoxic conditions will have a higher signal in the nucleus. Please see our publications for item NB100-134 for additional information regarding staining in IHC and ICC with this antibody.
-
Q: How much should I use for ChIP, 10E7 cells?
A: For ChIP, we recommend a dilution of 25 ul for 15 million cells. For 1 x 10^7 cells, you can use approximately 16-18 ul of this antibody.
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I am going to do a Western blot to detect HIF-1a protein on HTR8/SVneo trophoblast cells. I have used NB100-134 HIF-1 a antibody for immunostaining and will use the same Ab for Western. Now my questions are 1. What should I use for blocking? 2. Can I use HIF-1 alpha recombinant protein (H00003091-P01) as positive control? 3. What can I use as loading control? 4. Can I use DAB as substrate or anything else?
A: 1. You can use standard blocking solution which is 5% non-fat milk in TBST. 2. We recommend using Cos-7 Hypoxia Induced Nuclear Extract Kit (catalog #NB800-PC26) as the positive control for this antibody. 3. Since HIF-1 is a nuclear protein, you can use either alpha-Tubulin or beta-Tubulin as your loading control. 4. You can use DAB as your detection system.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I ordered this antibody but do not have a freezer to store it -20 degrees Celsius. Is it possible to store it for a short time (few days or weeks) at 4 degrees Celsius?
A: NB100-134 does contain Sodium Azide and should be fine at 4 degrees Celsius for about a week. We do recommend -20 degrees Celsius for storage and cannot guarantee this product will not degrade if kept at 4 degrees Celsius for more than a week.
-
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is the species reactivity of this antibody to Guinea pig based on computational homology?
A: The species reactivity reported for Guinea pig is based on published data. Here is the link for your reference. If you go through this paper carefully, on Figure 6, HIF1 expression has been tested across species including Cavia porcellus (Guinea pig).
-
Q: Our customer would like to know the shelf life of your HIF-1 alpha Antibody (Cat. # NB100-134). We would appreciate if you can confirm this information for me as soon as possible. Moreover, I was wondering whether your antibody products have a general shelf life?
A: Unless otherwise stated on our website, our antibodies are guaranteed for 1 year from the date of receipt, whist peptides and proteins are guaranteed for three months from the date of receipt. Novus guarantees that our products will work in the applications and species listed on our website and on our product datasheets. If a Novus product fails to meet this criteria, our technical service team will work with you to troubleshoot the issue. If this process is not successful, Novus will provide you with a free replacement or a 100% refund for the product.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the difference in the following two HIF-1 alpha antibodies? Catalog # NB100-105 and NB100-134
A: NB100-105 is a mouse monoclonal antibody, whereas NB100-134 is a rabbit polyclonal. They both use the same immunogen.
-
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Are there temperature requirements for storage?
A: We recommend short-term storage at 4C, and any long term storage at -20C. We recommend you aliquot any long-term storage and only remove what is needed for testing, as we recommend avoiding freeze-thaw cycles.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Does HIF1 alpha antibody (NB100-134) detect in the cytoplasm or the nucleus?
A: HIF1 alpha protein can be detected in both the cytoplasm and nucleus depending on the oxygen exposure of your tissues or cells. Cell that are more normoxic will have more signal in the cytoplasm while cells that are in hypoxic conditions will have a higher signal in the nucleus. Please see our publications for item NB100-134 for additional information regarding staining in IHC and ICC with this antibody.
-
Q: How much should I use for ChIP, 10E7 cells?
A: For ChIP, we recommend a dilution of 25 ul for 15 million cells. For 1 x 10^7 cells, you can use approximately 16-18 ul of this antibody.
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I am going to do a Western blot to detect HIF-1a protein on HTR8/SVneo trophoblast cells. I have used NB100-134 HIF-1 a antibody for immunostaining and will use the same Ab for Western. Now my questions are 1. What should I use for blocking? 2. Can I use HIF-1 alpha recombinant protein (H00003091-P01) as positive control? 3. What can I use as loading control? 4. Can I use DAB as substrate or anything else?
A: 1. You can use standard blocking solution which is 5% non-fat milk in TBST. 2. We recommend using Cos-7 Hypoxia Induced Nuclear Extract Kit (catalog #NB800-PC26) as the positive control for this antibody. 3. Since HIF-1 is a nuclear protein, you can use either alpha-Tubulin or beta-Tubulin as your loading control. 4. You can use DAB as your detection system.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I ordered this antibody but do not have a freezer to store it -20 degrees Celsius. Is it possible to store it for a short time (few days or weeks) at 4 degrees Celsius?
A: NB100-134 does contain Sodium Azide and should be fine at 4 degrees Celsius for about a week. We do recommend -20 degrees Celsius for storage and cannot guarantee this product will not degrade if kept at 4 degrees Celsius for more than a week.
-
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is the species reactivity of this antibody to Guinea pig based on computational homology?
A: The species reactivity reported for Guinea pig is based on published data. Here is the link for your reference. If you go through this paper carefully, on Figure 6, HIF1 expression has been tested across species including Cavia porcellus (Guinea pig).
-
Q: Our customer would like to know the shelf life of your HIF-1 alpha Antibody (Cat. # NB100-134). We would appreciate if you can confirm this information for me as soon as possible. Moreover, I was wondering whether your antibody products have a general shelf life?
A: Unless otherwise stated on our website, our antibodies are guaranteed for 1 year from the date of receipt, whist peptides and proteins are guaranteed for three months from the date of receipt. Novus guarantees that our products will work in the applications and species listed on our website and on our product datasheets. If a Novus product fails to meet this criteria, our technical service team will work with you to troubleshoot the issue. If this process is not successful, Novus will provide you with a free replacement or a 100% refund for the product.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the difference in the following two HIF-1 alpha antibodies? Catalog # NB100-105 and NB100-134
A: NB100-105 is a mouse monoclonal antibody, whereas NB100-134 is a rabbit polyclonal. They both use the same immunogen.
-
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Are there temperature requirements for storage?
A: We recommend short-term storage at 4C, and any long term storage at -20C. We recommend you aliquot any long-term storage and only remove what is needed for testing, as we recommend avoiding freeze-thaw cycles.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Does HIF1 alpha antibody (NB100-134) detect in the cytoplasm or the nucleus?
A: HIF1 alpha protein can be detected in both the cytoplasm and nucleus depending on the oxygen exposure of your tissues or cells. Cell that are more normoxic will have more signal in the cytoplasm while cells that are in hypoxic conditions will have a higher signal in the nucleus. Please see our publications for item NB100-134 for additional information regarding staining in IHC and ICC with this antibody.
-
Q: How much should I use for ChIP, 10E7 cells?
A: For ChIP, we recommend a dilution of 25 ul for 15 million cells. For 1 x 10^7 cells, you can use approximately 16-18 ul of this antibody.
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I am going to do a Western blot to detect HIF-1a protein on HTR8/SVneo trophoblast cells. I have used NB100-134 HIF-1 a antibody for immunostaining and will use the same Ab for Western. Now my questions are 1. What should I use for blocking? 2. Can I use HIF-1 alpha recombinant protein (H00003091-P01) as positive control? 3. What can I use as loading control? 4. Can I use DAB as substrate or anything else?
A: 1. You can use standard blocking solution which is 5% non-fat milk in TBST. 2. We recommend using Cos-7 Hypoxia Induced Nuclear Extract Kit (catalog #NB800-PC26) as the positive control for this antibody. 3. Since HIF-1 is a nuclear protein, you can use either alpha-Tubulin or beta-Tubulin as your loading control. 4. You can use DAB as your detection system.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I ordered this antibody but do not have a freezer to store it -20 degrees Celsius. Is it possible to store it for a short time (few days or weeks) at 4 degrees Celsius?
A: NB100-134 does contain Sodium Azide and should be fine at 4 degrees Celsius for about a week. We do recommend -20 degrees Celsius for storage and cannot guarantee this product will not degrade if kept at 4 degrees Celsius for more than a week.
-
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is the species reactivity of this antibody to Guinea pig based on computational homology?
A: The species reactivity reported for Guinea pig is based on published data. Here is the link for your reference. If you go through this paper carefully, on Figure 6, HIF1 expression has been tested across species including Cavia porcellus (Guinea pig).
-
Q: Our customer would like to know the shelf life of your HIF-1 alpha Antibody (Cat. # NB100-134). We would appreciate if you can confirm this information for me as soon as possible. Moreover, I was wondering whether your antibody products have a general shelf life?
A: Unless otherwise stated on our website, our antibodies are guaranteed for 1 year from the date of receipt, whist peptides and proteins are guaranteed for three months from the date of receipt. Novus guarantees that our products will work in the applications and species listed on our website and on our product datasheets. If a Novus product fails to meet this criteria, our technical service team will work with you to troubleshoot the issue. If this process is not successful, Novus will provide you with a free replacement or a 100% refund for the product.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the difference in the following two HIF-1 alpha antibodies? Catalog # NB100-105 and NB100-134
A: NB100-105 is a mouse monoclonal antibody, whereas NB100-134 is a rabbit polyclonal. They both use the same immunogen.
-
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Are there temperature requirements for storage?
A: We recommend short-term storage at 4C, and any long term storage at -20C. We recommend you aliquot any long-term storage and only remove what is needed for testing, as we recommend avoiding freeze-thaw cycles.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Does HIF1 alpha antibody (NB100-134) detect in the cytoplasm or the nucleus?
A: HIF1 alpha protein can be detected in both the cytoplasm and nucleus depending on the oxygen exposure of your tissues or cells. Cell that are more normoxic will have more signal in the cytoplasm while cells that are in hypoxic conditions will have a higher signal in the nucleus. Please see our publications for item NB100-134 for additional information regarding staining in IHC and ICC with this antibody.
-
Q: How much should I use for ChIP, 10E7 cells?
A: For ChIP, we recommend a dilution of 25 ul for 15 million cells. For 1 x 10^7 cells, you can use approximately 16-18 ul of this antibody.
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I am going to do a Western blot to detect HIF-1a protein on HTR8/SVneo trophoblast cells. I have used NB100-134 HIF-1 a antibody for immunostaining and will use the same Ab for Western. Now my questions are 1. What should I use for blocking? 2. Can I use HIF-1 alpha recombinant protein (H00003091-P01) as positive control? 3. What can I use as loading control? 4. Can I use DAB as substrate or anything else?
A: 1. You can use standard blocking solution which is 5% non-fat milk in TBST. 2. We recommend using Cos-7 Hypoxia Induced Nuclear Extract Kit (catalog #NB800-PC26) as the positive control for this antibody. 3. Since HIF-1 is a nuclear protein, you can use either alpha-Tubulin or beta-Tubulin as your loading control. 4. You can use DAB as your detection system.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I ordered this antibody but do not have a freezer to store it -20 degrees Celsius. Is it possible to store it for a short time (few days or weeks) at 4 degrees Celsius?
A: NB100-134 does contain Sodium Azide and should be fine at 4 degrees Celsius for about a week. We do recommend -20 degrees Celsius for storage and cannot guarantee this product will not degrade if kept at 4 degrees Celsius for more than a week.
-
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is the species reactivity of this antibody to Guinea pig based on computational homology?
A: The species reactivity reported for Guinea pig is based on published data. Here is the link for your reference. If you go through this paper carefully, on Figure 6, HIF1 expression has been tested across species including Cavia porcellus (Guinea pig).
-
Q: Our customer would like to know the shelf life of your HIF-1 alpha Antibody (Cat. # NB100-134). We would appreciate if you can confirm this information for me as soon as possible. Moreover, I was wondering whether your antibody products have a general shelf life?
A: Unless otherwise stated on our website, our antibodies are guaranteed for 1 year from the date of receipt, whist peptides and proteins are guaranteed for three months from the date of receipt. Novus guarantees that our products will work in the applications and species listed on our website and on our product datasheets. If a Novus product fails to meet this criteria, our technical service team will work with you to troubleshoot the issue. If this process is not successful, Novus will provide you with a free replacement or a 100% refund for the product.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the difference in the following two HIF-1 alpha antibodies? Catalog # NB100-105 and NB100-134
A: NB100-105 is a mouse monoclonal antibody, whereas NB100-134 is a rabbit polyclonal. They both use the same immunogen.
-
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Are there temperature requirements for storage?
A: We recommend short-term storage at 4C, and any long term storage at -20C. We recommend you aliquot any long-term storage and only remove what is needed for testing, as we recommend avoiding freeze-thaw cycles.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Does HIF1 alpha antibody (NB100-134) detect in the cytoplasm or the nucleus?
A: HIF1 alpha protein can be detected in both the cytoplasm and nucleus depending on the oxygen exposure of your tissues or cells. Cell that are more normoxic will have more signal in the cytoplasm while cells that are in hypoxic conditions will have a higher signal in the nucleus. Please see our publications for item NB100-134 for additional information regarding staining in IHC and ICC with this antibody.
-
Q: How much should I use for ChIP, 10E7 cells?
A: For ChIP, we recommend a dilution of 25 ul for 15 million cells. For 1 x 10^7 cells, you can use approximately 16-18 ul of this antibody.
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I am going to do a Western blot to detect HIF-1a protein on HTR8/SVneo trophoblast cells. I have used NB100-134 HIF-1 a antibody for immunostaining and will use the same Ab for Western. Now my questions are 1. What should I use for blocking? 2. Can I use HIF-1 alpha recombinant protein (H00003091-P01) as positive control? 3. What can I use as loading control? 4. Can I use DAB as substrate or anything else?
A: 1. You can use standard blocking solution which is 5% non-fat milk in TBST. 2. We recommend using Cos-7 Hypoxia Induced Nuclear Extract Kit (catalog #NB800-PC26) as the positive control for this antibody. 3. Since HIF-1 is a nuclear protein, you can use either alpha-Tubulin or beta-Tubulin as your loading control. 4. You can use DAB as your detection system.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I ordered this antibody but do not have a freezer to store it -20 degrees Celsius. Is it possible to store it for a short time (few days or weeks) at 4 degrees Celsius?
A: NB100-134 does contain Sodium Azide and should be fine at 4 degrees Celsius for about a week. We do recommend -20 degrees Celsius for storage and cannot guarantee this product will not degrade if kept at 4 degrees Celsius for more than a week.
-
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is the species reactivity of this antibody to Guinea pig based on computational homology?
A: The species reactivity reported for Guinea pig is based on published data. Here is the link for your reference. If you go through this paper carefully, on Figure 6, HIF1 expression has been tested across species including Cavia porcellus (Guinea pig).
-
Q: Our customer would like to know the shelf life of your HIF-1 alpha Antibody (Cat. # NB100-134). We would appreciate if you can confirm this information for me as soon as possible. Moreover, I was wondering whether your antibody products have a general shelf life?
A: Unless otherwise stated on our website, our antibodies are guaranteed for 1 year from the date of receipt, whist peptides and proteins are guaranteed for three months from the date of receipt. Novus guarantees that our products will work in the applications and species listed on our website and on our product datasheets. If a Novus product fails to meet this criteria, our technical service team will work with you to troubleshoot the issue. If this process is not successful, Novus will provide you with a free replacement or a 100% refund for the product.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the difference in the following two HIF-1 alpha antibodies? Catalog # NB100-105 and NB100-134
A: NB100-105 is a mouse monoclonal antibody, whereas NB100-134 is a rabbit polyclonal. They both use the same immunogen.
-
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Are there temperature requirements for storage?
A: We recommend short-term storage at 4C, and any long term storage at -20C. We recommend you aliquot any long-term storage and only remove what is needed for testing, as we recommend avoiding freeze-thaw cycles.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Does HIF1 alpha antibody (NB100-134) detect in the cytoplasm or the nucleus?
A: HIF1 alpha protein can be detected in both the cytoplasm and nucleus depending on the oxygen exposure of your tissues or cells. Cell that are more normoxic will have more signal in the cytoplasm while cells that are in hypoxic conditions will have a higher signal in the nucleus. Please see our publications for item NB100-134 for additional information regarding staining in IHC and ICC with this antibody.
-
Q: How much should I use for ChIP, 10E7 cells?
A: For ChIP, we recommend a dilution of 25 ul for 15 million cells. For 1 x 10^7 cells, you can use approximately 16-18 ul of this antibody.
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I am going to do a Western blot to detect HIF-1a protein on HTR8/SVneo trophoblast cells. I have used NB100-134 HIF-1 a antibody for immunostaining and will use the same Ab for Western. Now my questions are 1. What should I use for blocking? 2. Can I use HIF-1 alpha recombinant protein (H00003091-P01) as positive control? 3. What can I use as loading control? 4. Can I use DAB as substrate or anything else?
A: 1. You can use standard blocking solution which is 5% non-fat milk in TBST. 2. We recommend using Cos-7 Hypoxia Induced Nuclear Extract Kit (catalog #NB800-PC26) as the positive control for this antibody. 3. Since HIF-1 is a nuclear protein, you can use either alpha-Tubulin or beta-Tubulin as your loading control. 4. You can use DAB as your detection system.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I ordered this antibody but do not have a freezer to store it -20 degrees Celsius. Is it possible to store it for a short time (few days or weeks) at 4 degrees Celsius?
A: NB100-134 does contain Sodium Azide and should be fine at 4 degrees Celsius for about a week. We do recommend -20 degrees Celsius for storage and cannot guarantee this product will not degrade if kept at 4 degrees Celsius for more than a week.
-
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is the species reactivity of this antibody to Guinea pig based on computational homology?
A: The species reactivity reported for Guinea pig is based on published data. Here is the link for your reference. If you go through this paper carefully, on Figure 6, HIF1 expression has been tested across species including Cavia porcellus (Guinea pig).
-
Q: Our customer would like to know the shelf life of your HIF-1 alpha Antibody (Cat. # NB100-134). We would appreciate if you can confirm this information for me as soon as possible. Moreover, I was wondering whether your antibody products have a general shelf life?
A: Unless otherwise stated on our website, our antibodies are guaranteed for 1 year from the date of receipt, whist peptides and proteins are guaranteed for three months from the date of receipt. Novus guarantees that our products will work in the applications and species listed on our website and on our product datasheets. If a Novus product fails to meet this criteria, our technical service team will work with you to troubleshoot the issue. If this process is not successful, Novus will provide you with a free replacement or a 100% refund for the product.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the difference in the following two HIF-1 alpha antibodies? Catalog # NB100-105 and NB100-134
A: NB100-105 is a mouse monoclonal antibody, whereas NB100-134 is a rabbit polyclonal. They both use the same immunogen.
-
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Are there temperature requirements for storage?
A: We recommend short-term storage at 4C, and any long term storage at -20C. We recommend you aliquot any long-term storage and only remove what is needed for testing, as we recommend avoiding freeze-thaw cycles.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Does HIF1 alpha antibody (NB100-134) detect in the cytoplasm or the nucleus?
A: HIF1 alpha protein can be detected in both the cytoplasm and nucleus depending on the oxygen exposure of your tissues or cells. Cell that are more normoxic will have more signal in the cytoplasm while cells that are in hypoxic conditions will have a higher signal in the nucleus. Please see our publications for item NB100-134 for additional information regarding staining in IHC and ICC with this antibody.
-
Q: How much should I use for ChIP, 10E7 cells?
A: For ChIP, we recommend a dilution of 25 ul for 15 million cells. For 1 x 10^7 cells, you can use approximately 16-18 ul of this antibody.
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I am going to do a Western blot to detect HIF-1a protein on HTR8/SVneo trophoblast cells. I have used NB100-134 HIF-1 a antibody for immunostaining and will use the same Ab for Western. Now my questions are 1. What should I use for blocking? 2. Can I use HIF-1 alpha recombinant protein (H00003091-P01) as positive control? 3. What can I use as loading control? 4. Can I use DAB as substrate or anything else?
A: 1. You can use standard blocking solution which is 5% non-fat milk in TBST. 2. We recommend using Cos-7 Hypoxia Induced Nuclear Extract Kit (catalog #NB800-PC26) as the positive control for this antibody. 3. Since HIF-1 is a nuclear protein, you can use either alpha-Tubulin or beta-Tubulin as your loading control. 4. You can use DAB as your detection system.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I ordered this antibody but do not have a freezer to store it -20 degrees Celsius. Is it possible to store it for a short time (few days or weeks) at 4 degrees Celsius?
A: NB100-134 does contain Sodium Azide and should be fine at 4 degrees Celsius for about a week. We do recommend -20 degrees Celsius for storage and cannot guarantee this product will not degrade if kept at 4 degrees Celsius for more than a week.
-
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is the species reactivity of this antibody to Guinea pig based on computational homology?
A: The species reactivity reported for Guinea pig is based on published data. Here is the link for your reference. If you go through this paper carefully, on Figure 6, HIF1 expression has been tested across species including Cavia porcellus (Guinea pig).
-
Q: Our customer would like to know the shelf life of your HIF-1 alpha Antibody (Cat. # NB100-134). We would appreciate if you can confirm this information for me as soon as possible. Moreover, I was wondering whether your antibody products have a general shelf life?
A: Unless otherwise stated on our website, our antibodies are guaranteed for 1 year from the date of receipt, whist peptides and proteins are guaranteed for three months from the date of receipt. Novus guarantees that our products will work in the applications and species listed on our website and on our product datasheets. If a Novus product fails to meet this criteria, our technical service team will work with you to troubleshoot the issue. If this process is not successful, Novus will provide you with a free replacement or a 100% refund for the product.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the difference in the following two HIF-1 alpha antibodies? Catalog # NB100-105 and NB100-134
A: NB100-105 is a mouse monoclonal antibody, whereas NB100-134 is a rabbit polyclonal. They both use the same immunogen.
-
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Are there temperature requirements for storage?
A: We recommend short-term storage at 4C, and any long term storage at -20C. We recommend you aliquot any long-term storage and only remove what is needed for testing, as we recommend avoiding freeze-thaw cycles.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Does HIF1 alpha antibody (NB100-134) detect in the cytoplasm or the nucleus?
A: HIF1 alpha protein can be detected in both the cytoplasm and nucleus depending on the oxygen exposure of your tissues or cells. Cell that are more normoxic will have more signal in the cytoplasm while cells that are in hypoxic conditions will have a higher signal in the nucleus. Please see our publications for item NB100-134 for additional information regarding staining in IHC and ICC with this antibody.
-
Q: How much should I use for ChIP, 10E7 cells?
A: For ChIP, we recommend a dilution of 25 ul for 15 million cells. For 1 x 10^7 cells, you can use approximately 16-18 ul of this antibody.
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I am going to do a Western blot to detect HIF-1a protein on HTR8/SVneo trophoblast cells. I have used NB100-134 HIF-1 a antibody for immunostaining and will use the same Ab for Western. Now my questions are 1. What should I use for blocking? 2. Can I use HIF-1 alpha recombinant protein (H00003091-P01) as positive control? 3. What can I use as loading control? 4. Can I use DAB as substrate or anything else?
A: 1. You can use standard blocking solution which is 5% non-fat milk in TBST. 2. We recommend using Cos-7 Hypoxia Induced Nuclear Extract Kit (catalog #NB800-PC26) as the positive control for this antibody. 3. Since HIF-1 is a nuclear protein, you can use either alpha-Tubulin or beta-Tubulin as your loading control. 4. You can use DAB as your detection system.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I ordered this antibody but do not have a freezer to store it -20 degrees Celsius. Is it possible to store it for a short time (few days or weeks) at 4 degrees Celsius?
A: NB100-134 does contain Sodium Azide and should be fine at 4 degrees Celsius for about a week. We do recommend -20 degrees Celsius for storage and cannot guarantee this product will not degrade if kept at 4 degrees Celsius for more than a week.
-
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is the species reactivity of this antibody to Guinea pig based on computational homology?
A: The species reactivity reported for Guinea pig is based on published data. Here is the link for your reference. If you go through this paper carefully, on Figure 6, HIF1 expression has been tested across species including Cavia porcellus (Guinea pig).
-
Q: Our customer would like to know the shelf life of your HIF-1 alpha Antibody (Cat. # NB100-134). We would appreciate if you can confirm this information for me as soon as possible. Moreover, I was wondering whether your antibody products have a general shelf life?
A: Unless otherwise stated on our website, our antibodies are guaranteed for 1 year from the date of receipt, whist peptides and proteins are guaranteed for three months from the date of receipt. Novus guarantees that our products will work in the applications and species listed on our website and on our product datasheets. If a Novus product fails to meet this criteria, our technical service team will work with you to troubleshoot the issue. If this process is not successful, Novus will provide you with a free replacement or a 100% refund for the product.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the difference in the following two HIF-1 alpha antibodies? Catalog # NB100-105 and NB100-134
A: NB100-105 is a mouse monoclonal antibody, whereas NB100-134 is a rabbit polyclonal. They both use the same immunogen.
-
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Are there temperature requirements for storage?
A: We recommend short-term storage at 4C, and any long term storage at -20C. We recommend you aliquot any long-term storage and only remove what is needed for testing, as we recommend avoiding freeze-thaw cycles.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Does HIF1 alpha antibody (NB100-134) detect in the cytoplasm or the nucleus?
A: HIF1 alpha protein can be detected in both the cytoplasm and nucleus depending on the oxygen exposure of your tissues or cells. Cell that are more normoxic will have more signal in the cytoplasm while cells that are in hypoxic conditions will have a higher signal in the nucleus. Please see our publications for item NB100-134 for additional information regarding staining in IHC and ICC with this antibody.
-
Q: How much should I use for ChIP, 10E7 cells?
A: For ChIP, we recommend a dilution of 25 ul for 15 million cells. For 1 x 10^7 cells, you can use approximately 16-18 ul of this antibody.
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I am going to do a Western blot to detect HIF-1a protein on HTR8/SVneo trophoblast cells. I have used NB100-134 HIF-1 a antibody for immunostaining and will use the same Ab for Western. Now my questions are 1. What should I use for blocking? 2. Can I use HIF-1 alpha recombinant protein (H00003091-P01) as positive control? 3. What can I use as loading control? 4. Can I use DAB as substrate or anything else?
A: 1. You can use standard blocking solution which is 5% non-fat milk in TBST. 2. We recommend using Cos-7 Hypoxia Induced Nuclear Extract Kit (catalog #NB800-PC26) as the positive control for this antibody. 3. Since HIF-1 is a nuclear protein, you can use either alpha-Tubulin or beta-Tubulin as your loading control. 4. You can use DAB as your detection system.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I ordered this antibody but do not have a freezer to store it -20 degrees Celsius. Is it possible to store it for a short time (few days or weeks) at 4 degrees Celsius?
A: NB100-134 does contain Sodium Azide and should be fine at 4 degrees Celsius for about a week. We do recommend -20 degrees Celsius for storage and cannot guarantee this product will not degrade if kept at 4 degrees Celsius for more than a week.
-
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is the species reactivity of this antibody to Guinea pig based on computational homology?
A: The species reactivity reported for Guinea pig is based on published data. Here is the link for your reference. If you go through this paper carefully, on Figure 6, HIF1 expression has been tested across species including Cavia porcellus (Guinea pig).
-
Q: Our customer would like to know the shelf life of your HIF-1 alpha Antibody (Cat. # NB100-134). We would appreciate if you can confirm this information for me as soon as possible. Moreover, I was wondering whether your antibody products have a general shelf life?
A: Unless otherwise stated on our website, our antibodies are guaranteed for 1 year from the date of receipt, whist peptides and proteins are guaranteed for three months from the date of receipt. Novus guarantees that our products will work in the applications and species listed on our website and on our product datasheets. If a Novus product fails to meet this criteria, our technical service team will work with you to troubleshoot the issue. If this process is not successful, Novus will provide you with a free replacement or a 100% refund for the product.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the difference in the following two HIF-1 alpha antibodies? Catalog # NB100-105 and NB100-134
A: NB100-105 is a mouse monoclonal antibody, whereas NB100-134 is a rabbit polyclonal. They both use the same immunogen.
-
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Are there temperature requirements for storage?
A: We recommend short-term storage at 4C, and any long term storage at -20C. We recommend you aliquot any long-term storage and only remove what is needed for testing, as we recommend avoiding freeze-thaw cycles.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Does HIF1 alpha antibody (NB100-134) detect in the cytoplasm or the nucleus?
A: HIF1 alpha protein can be detected in both the cytoplasm and nucleus depending on the oxygen exposure of your tissues or cells. Cell that are more normoxic will have more signal in the cytoplasm while cells that are in hypoxic conditions will have a higher signal in the nucleus. Please see our publications for item NB100-134 for additional information regarding staining in IHC and ICC with this antibody.
-
Q: How much should I use for ChIP, 10E7 cells?
A: For ChIP, we recommend a dilution of 25 ul for 15 million cells. For 1 x 10^7 cells, you can use approximately 16-18 ul of this antibody.
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I am going to do a Western blot to detect HIF-1a protein on HTR8/SVneo trophoblast cells. I have used NB100-134 HIF-1 a antibody for immunostaining and will use the same Ab for Western. Now my questions are 1. What should I use for blocking? 2. Can I use HIF-1 alpha recombinant protein (H00003091-P01) as positive control? 3. What can I use as loading control? 4. Can I use DAB as substrate or anything else?
A: 1. You can use standard blocking solution which is 5% non-fat milk in TBST. 2. We recommend using Cos-7 Hypoxia Induced Nuclear Extract Kit (catalog #NB800-PC26) as the positive control for this antibody. 3. Since HIF-1 is a nuclear protein, you can use either alpha-Tubulin or beta-Tubulin as your loading control. 4. You can use DAB as your detection system.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I ordered this antibody but do not have a freezer to store it -20 degrees Celsius. Is it possible to store it for a short time (few days or weeks) at 4 degrees Celsius?
A: NB100-134 does contain Sodium Azide and should be fine at 4 degrees Celsius for about a week. We do recommend -20 degrees Celsius for storage and cannot guarantee this product will not degrade if kept at 4 degrees Celsius for more than a week.
-
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is the species reactivity of this antibody to Guinea pig based on computational homology?
A: The species reactivity reported for Guinea pig is based on published data. Here is the link for your reference. If you go through this paper carefully, on Figure 6, HIF1 expression has been tested across species including Cavia porcellus (Guinea pig).
-
Q: Our customer would like to know the shelf life of your HIF-1 alpha Antibody (Cat. # NB100-134). We would appreciate if you can confirm this information for me as soon as possible. Moreover, I was wondering whether your antibody products have a general shelf life?
A: Unless otherwise stated on our website, our antibodies are guaranteed for 1 year from the date of receipt, whist peptides and proteins are guaranteed for three months from the date of receipt. Novus guarantees that our products will work in the applications and species listed on our website and on our product datasheets. If a Novus product fails to meet this criteria, our technical service team will work with you to troubleshoot the issue. If this process is not successful, Novus will provide you with a free replacement or a 100% refund for the product.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the difference in the following two HIF-1 alpha antibodies? Catalog # NB100-105 and NB100-134
A: NB100-105 is a mouse monoclonal antibody, whereas NB100-134 is a rabbit polyclonal. They both use the same immunogen.
-
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Are there temperature requirements for storage?
A: We recommend short-term storage at 4C, and any long term storage at -20C. We recommend you aliquot any long-term storage and only remove what is needed for testing, as we recommend avoiding freeze-thaw cycles.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Does HIF1 alpha antibody (NB100-134) detect in the cytoplasm or the nucleus?
A: HIF1 alpha protein can be detected in both the cytoplasm and nucleus depending on the oxygen exposure of your tissues or cells. Cell that are more normoxic will have more signal in the cytoplasm while cells that are in hypoxic conditions will have a higher signal in the nucleus. Please see our publications for item NB100-134 for additional information regarding staining in IHC and ICC with this antibody.
-
Q: How much should I use for ChIP, 10E7 cells?
A: For ChIP, we recommend a dilution of 25 ul for 15 million cells. For 1 x 10^7 cells, you can use approximately 16-18 ul of this antibody.
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I am going to do a Western blot to detect HIF-1a protein on HTR8/SVneo trophoblast cells. I have used NB100-134 HIF-1 a antibody for immunostaining and will use the same Ab for Western. Now my questions are 1. What should I use for blocking? 2. Can I use HIF-1 alpha recombinant protein (H00003091-P01) as positive control? 3. What can I use as loading control? 4. Can I use DAB as substrate or anything else?
A: 1. You can use standard blocking solution which is 5% non-fat milk in TBST. 2. We recommend using Cos-7 Hypoxia Induced Nuclear Extract Kit (catalog #NB800-PC26) as the positive control for this antibody. 3. Since HIF-1 is a nuclear protein, you can use either alpha-Tubulin or beta-Tubulin as your loading control. 4. You can use DAB as your detection system.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I ordered this antibody but do not have a freezer to store it -20 degrees Celsius. Is it possible to store it for a short time (few days or weeks) at 4 degrees Celsius?
A: NB100-134 does contain Sodium Azide and should be fine at 4 degrees Celsius for about a week. We do recommend -20 degrees Celsius for storage and cannot guarantee this product will not degrade if kept at 4 degrees Celsius for more than a week.
-
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is the species reactivity of this antibody to Guinea pig based on computational homology?
A: The species reactivity reported for Guinea pig is based on published data. Here is the link for your reference. If you go through this paper carefully, on Figure 6, HIF1 expression has been tested across species including Cavia porcellus (Guinea pig).
-
Q: Our customer would like to know the shelf life of your HIF-1 alpha Antibody (Cat. # NB100-134). We would appreciate if you can confirm this information for me as soon as possible. Moreover, I was wondering whether your antibody products have a general shelf life?
A: Unless otherwise stated on our website, our antibodies are guaranteed for 1 year from the date of receipt, whist peptides and proteins are guaranteed for three months from the date of receipt. Novus guarantees that our products will work in the applications and species listed on our website and on our product datasheets. If a Novus product fails to meet this criteria, our technical service team will work with you to troubleshoot the issue. If this process is not successful, Novus will provide you with a free replacement or a 100% refund for the product.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the difference in the following two HIF-1 alpha antibodies? Catalog # NB100-105 and NB100-134
A: NB100-105 is a mouse monoclonal antibody, whereas NB100-134 is a rabbit polyclonal. They both use the same immunogen.
-
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Are there temperature requirements for storage?
A: We recommend short-term storage at 4C, and any long term storage at -20C. We recommend you aliquot any long-term storage and only remove what is needed for testing, as we recommend avoiding freeze-thaw cycles.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Does HIF1 alpha antibody (NB100-134) detect in the cytoplasm or the nucleus?
A: HIF1 alpha protein can be detected in both the cytoplasm and nucleus depending on the oxygen exposure of your tissues or cells. Cell that are more normoxic will have more signal in the cytoplasm while cells that are in hypoxic conditions will have a higher signal in the nucleus. Please see our publications for item NB100-134 for additional information regarding staining in IHC and ICC with this antibody.
-
Q: How much should I use for ChIP, 10E7 cells?
A: For ChIP, we recommend a dilution of 25 ul for 15 million cells. For 1 x 10^7 cells, you can use approximately 16-18 ul of this antibody.
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I am going to do a Western blot to detect HIF-1a protein on HTR8/SVneo trophoblast cells. I have used NB100-134 HIF-1 a antibody for immunostaining and will use the same Ab for Western. Now my questions are 1. What should I use for blocking? 2. Can I use HIF-1 alpha recombinant protein (H00003091-P01) as positive control? 3. What can I use as loading control? 4. Can I use DAB as substrate or anything else?
A: 1. You can use standard blocking solution which is 5% non-fat milk in TBST. 2. We recommend using Cos-7 Hypoxia Induced Nuclear Extract Kit (catalog #NB800-PC26) as the positive control for this antibody. 3. Since HIF-1 is a nuclear protein, you can use either alpha-Tubulin or beta-Tubulin as your loading control. 4. You can use DAB as your detection system.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I ordered this antibody but do not have a freezer to store it -20 degrees Celsius. Is it possible to store it for a short time (few days or weeks) at 4 degrees Celsius?
A: NB100-134 does contain Sodium Azide and should be fine at 4 degrees Celsius for about a week. We do recommend -20 degrees Celsius for storage and cannot guarantee this product will not degrade if kept at 4 degrees Celsius for more than a week.
-
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is the species reactivity of this antibody to Guinea pig based on computational homology?
A: The species reactivity reported for Guinea pig is based on published data. Here is the link for your reference. If you go through this paper carefully, on Figure 6, HIF1 expression has been tested across species including Cavia porcellus (Guinea pig).
-
Q: Our customer would like to know the shelf life of your HIF-1 alpha Antibody (Cat. # NB100-134). We would appreciate if you can confirm this information for me as soon as possible. Moreover, I was wondering whether your antibody products have a general shelf life?
A: Unless otherwise stated on our website, our antibodies are guaranteed for 1 year from the date of receipt, whist peptides and proteins are guaranteed for three months from the date of receipt. Novus guarantees that our products will work in the applications and species listed on our website and on our product datasheets. If a Novus product fails to meet this criteria, our technical service team will work with you to troubleshoot the issue. If this process is not successful, Novus will provide you with a free replacement or a 100% refund for the product.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the difference in the following two HIF-1 alpha antibodies? Catalog # NB100-105 and NB100-134
A: NB100-105 is a mouse monoclonal antibody, whereas NB100-134 is a rabbit polyclonal. They both use the same immunogen.
-
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Are there temperature requirements for storage?
A: We recommend short-term storage at 4C, and any long term storage at -20C. We recommend you aliquot any long-term storage and only remove what is needed for testing, as we recommend avoiding freeze-thaw cycles.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Does HIF1 alpha antibody (NB100-134) detect in the cytoplasm or the nucleus?
A: HIF1 alpha protein can be detected in both the cytoplasm and nucleus depending on the oxygen exposure of your tissues or cells. Cell that are more normoxic will have more signal in the cytoplasm while cells that are in hypoxic conditions will have a higher signal in the nucleus. Please see our publications for item NB100-134 for additional information regarding staining in IHC and ICC with this antibody.
-
Q: How much should I use for ChIP, 10E7 cells?
A: For ChIP, we recommend a dilution of 25 ul for 15 million cells. For 1 x 10^7 cells, you can use approximately 16-18 ul of this antibody.
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I am going to do a Western blot to detect HIF-1a protein on HTR8/SVneo trophoblast cells. I have used NB100-134 HIF-1 a antibody for immunostaining and will use the same Ab for Western. Now my questions are 1. What should I use for blocking? 2. Can I use HIF-1 alpha recombinant protein (H00003091-P01) as positive control? 3. What can I use as loading control? 4. Can I use DAB as substrate or anything else?
A: 1. You can use standard blocking solution which is 5% non-fat milk in TBST. 2. We recommend using Cos-7 Hypoxia Induced Nuclear Extract Kit (catalog #NB800-PC26) as the positive control for this antibody. 3. Since HIF-1 is a nuclear protein, you can use either alpha-Tubulin or beta-Tubulin as your loading control. 4. You can use DAB as your detection system.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I ordered this antibody but do not have a freezer to store it -20 degrees Celsius. Is it possible to store it for a short time (few days or weeks) at 4 degrees Celsius?
A: NB100-134 does contain Sodium Azide and should be fine at 4 degrees Celsius for about a week. We do recommend -20 degrees Celsius for storage and cannot guarantee this product will not degrade if kept at 4 degrees Celsius for more than a week.
-
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is the species reactivity of this antibody to Guinea pig based on computational homology?
A: The species reactivity reported for Guinea pig is based on published data. Here is the link for your reference. If you go through this paper carefully, on Figure 6, HIF1 expression has been tested across species including Cavia porcellus (Guinea pig).
-
Q: Our customer would like to know the shelf life of your HIF-1 alpha Antibody (Cat. # NB100-134). We would appreciate if you can confirm this information for me as soon as possible. Moreover, I was wondering whether your antibody products have a general shelf life?
A: Unless otherwise stated on our website, our antibodies are guaranteed for 1 year from the date of receipt, whist peptides and proteins are guaranteed for three months from the date of receipt. Novus guarantees that our products will work in the applications and species listed on our website and on our product datasheets. If a Novus product fails to meet this criteria, our technical service team will work with you to troubleshoot the issue. If this process is not successful, Novus will provide you with a free replacement or a 100% refund for the product.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the difference in the following two HIF-1 alpha antibodies? Catalog # NB100-105 and NB100-134
A: NB100-105 is a mouse monoclonal antibody, whereas NB100-134 is a rabbit polyclonal. They both use the same immunogen.
-
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Are there temperature requirements for storage?
A: We recommend short-term storage at 4C, and any long term storage at -20C. We recommend you aliquot any long-term storage and only remove what is needed for testing, as we recommend avoiding freeze-thaw cycles.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Does HIF1 alpha antibody (NB100-134) detect in the cytoplasm or the nucleus?
A: HIF1 alpha protein can be detected in both the cytoplasm and nucleus depending on the oxygen exposure of your tissues or cells. Cell that are more normoxic will have more signal in the cytoplasm while cells that are in hypoxic conditions will have a higher signal in the nucleus. Please see our publications for item NB100-134 for additional information regarding staining in IHC and ICC with this antibody.
-
Q: How much should I use for ChIP, 10E7 cells?
A: For ChIP, we recommend a dilution of 25 ul for 15 million cells. For 1 x 10^7 cells, you can use approximately 16-18 ul of this antibody.
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I am going to do a Western blot to detect HIF-1a protein on HTR8/SVneo trophoblast cells. I have used NB100-134 HIF-1 a antibody for immunostaining and will use the same Ab for Western. Now my questions are 1. What should I use for blocking? 2. Can I use HIF-1 alpha recombinant protein (H00003091-P01) as positive control? 3. What can I use as loading control? 4. Can I use DAB as substrate or anything else?
A: 1. You can use standard blocking solution which is 5% non-fat milk in TBST. 2. We recommend using Cos-7 Hypoxia Induced Nuclear Extract Kit (catalog #NB800-PC26) as the positive control for this antibody. 3. Since HIF-1 is a nuclear protein, you can use either alpha-Tubulin or beta-Tubulin as your loading control. 4. You can use DAB as your detection system.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I ordered this antibody but do not have a freezer to store it -20 degrees Celsius. Is it possible to store it for a short time (few days or weeks) at 4 degrees Celsius?
A: NB100-134 does contain Sodium Azide and should be fine at 4 degrees Celsius for about a week. We do recommend -20 degrees Celsius for storage and cannot guarantee this product will not degrade if kept at 4 degrees Celsius for more than a week.
-
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is the species reactivity of this antibody to Guinea pig based on computational homology?
A: The species reactivity reported for Guinea pig is based on published data. Here is the link for your reference. If you go through this paper carefully, on Figure 6, HIF1 expression has been tested across species including Cavia porcellus (Guinea pig).
-
Q: Our customer would like to know the shelf life of your HIF-1 alpha Antibody (Cat. # NB100-134). We would appreciate if you can confirm this information for me as soon as possible. Moreover, I was wondering whether your antibody products have a general shelf life?
A: Unless otherwise stated on our website, our antibodies are guaranteed for 1 year from the date of receipt, whist peptides and proteins are guaranteed for three months from the date of receipt. Novus guarantees that our products will work in the applications and species listed on our website and on our product datasheets. If a Novus product fails to meet this criteria, our technical service team will work with you to troubleshoot the issue. If this process is not successful, Novus will provide you with a free replacement or a 100% refund for the product.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the difference in the following two HIF-1 alpha antibodies? Catalog # NB100-105 and NB100-134
A: NB100-105 is a mouse monoclonal antibody, whereas NB100-134 is a rabbit polyclonal. They both use the same immunogen.
-
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Are there temperature requirements for storage?
A: We recommend short-term storage at 4C, and any long term storage at -20C. We recommend you aliquot any long-term storage and only remove what is needed for testing, as we recommend avoiding freeze-thaw cycles.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Does HIF1 alpha antibody (NB100-134) detect in the cytoplasm or the nucleus?
A: HIF1 alpha protein can be detected in both the cytoplasm and nucleus depending on the oxygen exposure of your tissues or cells. Cell that are more normoxic will have more signal in the cytoplasm while cells that are in hypoxic conditions will have a higher signal in the nucleus. Please see our publications for item NB100-134 for additional information regarding staining in IHC and ICC with this antibody.
-
Q: How much should I use for ChIP, 10E7 cells?
A: For ChIP, we recommend a dilution of 25 ul for 15 million cells. For 1 x 10^7 cells, you can use approximately 16-18 ul of this antibody.
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I am going to do a Western blot to detect HIF-1a protein on HTR8/SVneo trophoblast cells. I have used NB100-134 HIF-1 a antibody for immunostaining and will use the same Ab for Western. Now my questions are 1. What should I use for blocking? 2. Can I use HIF-1 alpha recombinant protein (H00003091-P01) as positive control? 3. What can I use as loading control? 4. Can I use DAB as substrate or anything else?
A: 1. You can use standard blocking solution which is 5% non-fat milk in TBST. 2. We recommend using Cos-7 Hypoxia Induced Nuclear Extract Kit (catalog #NB800-PC26) as the positive control for this antibody. 3. Since HIF-1 is a nuclear protein, you can use either alpha-Tubulin or beta-Tubulin as your loading control. 4. You can use DAB as your detection system.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I ordered this antibody but do not have a freezer to store it -20 degrees Celsius. Is it possible to store it for a short time (few days or weeks) at 4 degrees Celsius?
A: NB100-134 does contain Sodium Azide and should be fine at 4 degrees Celsius for about a week. We do recommend -20 degrees Celsius for storage and cannot guarantee this product will not degrade if kept at 4 degrees Celsius for more than a week.
-
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is the species reactivity of this antibody to Guinea pig based on computational homology?
A: The species reactivity reported for Guinea pig is based on published data. Here is the link for your reference. If you go through this paper carefully, on Figure 6, HIF1 expression has been tested across species including Cavia porcellus (Guinea pig).
-
Q: Our customer would like to know the shelf life of your HIF-1 alpha Antibody (Cat. # NB100-134). We would appreciate if you can confirm this information for me as soon as possible. Moreover, I was wondering whether your antibody products have a general shelf life?
A: Unless otherwise stated on our website, our antibodies are guaranteed for 1 year from the date of receipt, whist peptides and proteins are guaranteed for three months from the date of receipt. Novus guarantees that our products will work in the applications and species listed on our website and on our product datasheets. If a Novus product fails to meet this criteria, our technical service team will work with you to troubleshoot the issue. If this process is not successful, Novus will provide you with a free replacement or a 100% refund for the product.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the difference in the following two HIF-1 alpha antibodies? Catalog # NB100-105 and NB100-134
A: NB100-105 is a mouse monoclonal antibody, whereas NB100-134 is a rabbit polyclonal. They both use the same immunogen.
-
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Are there temperature requirements for storage?
A: We recommend short-term storage at 4C, and any long term storage at -20C. We recommend you aliquot any long-term storage and only remove what is needed for testing, as we recommend avoiding freeze-thaw cycles.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Does HIF1 alpha antibody (NB100-134) detect in the cytoplasm or the nucleus?
A: HIF1 alpha protein can be detected in both the cytoplasm and nucleus depending on the oxygen exposure of your tissues or cells. Cell that are more normoxic will have more signal in the cytoplasm while cells that are in hypoxic conditions will have a higher signal in the nucleus. Please see our publications for item NB100-134 for additional information regarding staining in IHC and ICC with this antibody.
-
Q: How much should I use for ChIP, 10E7 cells?
A: For ChIP, we recommend a dilution of 25 ul for 15 million cells. For 1 x 10^7 cells, you can use approximately 16-18 ul of this antibody.
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I am going to do a Western blot to detect HIF-1a protein on HTR8/SVneo trophoblast cells. I have used NB100-134 HIF-1 a antibody for immunostaining and will use the same Ab for Western. Now my questions are 1. What should I use for blocking? 2. Can I use HIF-1 alpha recombinant protein (H00003091-P01) as positive control? 3. What can I use as loading control? 4. Can I use DAB as substrate or anything else?
A: 1. You can use standard blocking solution which is 5% non-fat milk in TBST. 2. We recommend using Cos-7 Hypoxia Induced Nuclear Extract Kit (catalog #NB800-PC26) as the positive control for this antibody. 3. Since HIF-1 is a nuclear protein, you can use either alpha-Tubulin or beta-Tubulin as your loading control. 4. You can use DAB as your detection system.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I ordered this antibody but do not have a freezer to store it -20 degrees Celsius. Is it possible to store it for a short time (few days or weeks) at 4 degrees Celsius?
A: NB100-134 does contain Sodium Azide and should be fine at 4 degrees Celsius for about a week. We do recommend -20 degrees Celsius for storage and cannot guarantee this product will not degrade if kept at 4 degrees Celsius for more than a week.
-
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is the species reactivity of this antibody to Guinea pig based on computational homology?
A: The species reactivity reported for Guinea pig is based on published data. Here is the link for your reference. If you go through this paper carefully, on Figure 6, HIF1 expression has been tested across species including Cavia porcellus (Guinea pig).
-
Q: Our customer would like to know the shelf life of your HIF-1 alpha Antibody (Cat. # NB100-134). We would appreciate if you can confirm this information for me as soon as possible. Moreover, I was wondering whether your antibody products have a general shelf life?
A: Unless otherwise stated on our website, our antibodies are guaranteed for 1 year from the date of receipt, whist peptides and proteins are guaranteed for three months from the date of receipt. Novus guarantees that our products will work in the applications and species listed on our website and on our product datasheets. If a Novus product fails to meet this criteria, our technical service team will work with you to troubleshoot the issue. If this process is not successful, Novus will provide you with a free replacement or a 100% refund for the product.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the difference in the following two HIF-1 alpha antibodies? Catalog # NB100-105 and NB100-134
A: NB100-105 is a mouse monoclonal antibody, whereas NB100-134 is a rabbit polyclonal. They both use the same immunogen.
-
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Are there temperature requirements for storage?
A: We recommend short-term storage at 4C, and any long term storage at -20C. We recommend you aliquot any long-term storage and only remove what is needed for testing, as we recommend avoiding freeze-thaw cycles.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Does HIF1 alpha antibody (NB100-134) detect in the cytoplasm or the nucleus?
A: HIF1 alpha protein can be detected in both the cytoplasm and nucleus depending on the oxygen exposure of your tissues or cells. Cell that are more normoxic will have more signal in the cytoplasm while cells that are in hypoxic conditions will have a higher signal in the nucleus. Please see our publications for item NB100-134 for additional information regarding staining in IHC and ICC with this antibody.
-
Q: How much should I use for ChIP, 10E7 cells?
A: For ChIP, we recommend a dilution of 25 ul for 15 million cells. For 1 x 10^7 cells, you can use approximately 16-18 ul of this antibody.
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I am going to do a Western blot to detect HIF-1a protein on HTR8/SVneo trophoblast cells. I have used NB100-134 HIF-1 a antibody for immunostaining and will use the same Ab for Western. Now my questions are 1. What should I use for blocking? 2. Can I use HIF-1 alpha recombinant protein (H00003091-P01) as positive control? 3. What can I use as loading control? 4. Can I use DAB as substrate or anything else?
A: 1. You can use standard blocking solution which is 5% non-fat milk in TBST. 2. We recommend using Cos-7 Hypoxia Induced Nuclear Extract Kit (catalog #NB800-PC26) as the positive control for this antibody. 3. Since HIF-1 is a nuclear protein, you can use either alpha-Tubulin or beta-Tubulin as your loading control. 4. You can use DAB as your detection system.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I ordered this antibody but do not have a freezer to store it -20 degrees Celsius. Is it possible to store it for a short time (few days or weeks) at 4 degrees Celsius?
A: NB100-134 does contain Sodium Azide and should be fine at 4 degrees Celsius for about a week. We do recommend -20 degrees Celsius for storage and cannot guarantee this product will not degrade if kept at 4 degrees Celsius for more than a week.
-
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is the species reactivity of this antibody to Guinea pig based on computational homology?
A: The species reactivity reported for Guinea pig is based on published data. Here is the link for your reference. If you go through this paper carefully, on Figure 6, HIF1 expression has been tested across species including Cavia porcellus (Guinea pig).
-
Q: Our customer would like to know the shelf life of your HIF-1 alpha Antibody (Cat. # NB100-134). We would appreciate if you can confirm this information for me as soon as possible. Moreover, I was wondering whether your antibody products have a general shelf life?
A: Unless otherwise stated on our website, our antibodies are guaranteed for 1 year from the date of receipt, whist peptides and proteins are guaranteed for three months from the date of receipt. Novus guarantees that our products will work in the applications and species listed on our website and on our product datasheets. If a Novus product fails to meet this criteria, our technical service team will work with you to troubleshoot the issue. If this process is not successful, Novus will provide you with a free replacement or a 100% refund for the product.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the difference in the following two HIF-1 alpha antibodies? Catalog # NB100-105 and NB100-134
A: NB100-105 is a mouse monoclonal antibody, whereas NB100-134 is a rabbit polyclonal. They both use the same immunogen.
-
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Are there temperature requirements for storage?
A: We recommend short-term storage at 4C, and any long term storage at -20C. We recommend you aliquot any long-term storage and only remove what is needed for testing, as we recommend avoiding freeze-thaw cycles.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Does HIF1 alpha antibody (NB100-134) detect in the cytoplasm or the nucleus?
A: HIF1 alpha protein can be detected in both the cytoplasm and nucleus depending on the oxygen exposure of your tissues or cells. Cell that are more normoxic will have more signal in the cytoplasm while cells that are in hypoxic conditions will have a higher signal in the nucleus. Please see our publications for item NB100-134 for additional information regarding staining in IHC and ICC with this antibody.
-
Q: How much should I use for ChIP, 10E7 cells?
A: For ChIP, we recommend a dilution of 25 ul for 15 million cells. For 1 x 10^7 cells, you can use approximately 16-18 ul of this antibody.
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I am going to do a Western blot to detect HIF-1a protein on HTR8/SVneo trophoblast cells. I have used NB100-134 HIF-1 a antibody for immunostaining and will use the same Ab for Western. Now my questions are 1. What should I use for blocking? 2. Can I use HIF-1 alpha recombinant protein (H00003091-P01) as positive control? 3. What can I use as loading control? 4. Can I use DAB as substrate or anything else?
A: 1. You can use standard blocking solution which is 5% non-fat milk in TBST. 2. We recommend using Cos-7 Hypoxia Induced Nuclear Extract Kit (catalog #NB800-PC26) as the positive control for this antibody. 3. Since HIF-1 is a nuclear protein, you can use either alpha-Tubulin or beta-Tubulin as your loading control. 4. You can use DAB as your detection system.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I ordered this antibody but do not have a freezer to store it -20 degrees Celsius. Is it possible to store it for a short time (few days or weeks) at 4 degrees Celsius?
A: NB100-134 does contain Sodium Azide and should be fine at 4 degrees Celsius for about a week. We do recommend -20 degrees Celsius for storage and cannot guarantee this product will not degrade if kept at 4 degrees Celsius for more than a week.
-
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is the species reactivity of this antibody to Guinea pig based on computational homology?
A: The species reactivity reported for Guinea pig is based on published data. Here is the link for your reference. If you go through this paper carefully, on Figure 6, HIF1 expression has been tested across species including Cavia porcellus (Guinea pig).
-
Q: Our customer would like to know the shelf life of your HIF-1 alpha Antibody (Cat. # NB100-134). We would appreciate if you can confirm this information for me as soon as possible. Moreover, I was wondering whether your antibody products have a general shelf life?
A: Unless otherwise stated on our website, our antibodies are guaranteed for 1 year from the date of receipt, whist peptides and proteins are guaranteed for three months from the date of receipt. Novus guarantees that our products will work in the applications and species listed on our website and on our product datasheets. If a Novus product fails to meet this criteria, our technical service team will work with you to troubleshoot the issue. If this process is not successful, Novus will provide you with a free replacement or a 100% refund for the product.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the difference in the following two HIF-1 alpha antibodies? Catalog # NB100-105 and NB100-134
A: NB100-105 is a mouse monoclonal antibody, whereas NB100-134 is a rabbit polyclonal. They both use the same immunogen.
-
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Are there temperature requirements for storage?
A: We recommend short-term storage at 4C, and any long term storage at -20C. We recommend you aliquot any long-term storage and only remove what is needed for testing, as we recommend avoiding freeze-thaw cycles.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Does HIF1 alpha antibody (NB100-134) detect in the cytoplasm or the nucleus?
A: HIF1 alpha protein can be detected in both the cytoplasm and nucleus depending on the oxygen exposure of your tissues or cells. Cell that are more normoxic will have more signal in the cytoplasm while cells that are in hypoxic conditions will have a higher signal in the nucleus. Please see our publications for item NB100-134 for additional information regarding staining in IHC and ICC with this antibody.
-
Q: How much should I use for ChIP, 10E7 cells?
A: For ChIP, we recommend a dilution of 25 ul for 15 million cells. For 1 x 10^7 cells, you can use approximately 16-18 ul of this antibody.
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I am going to do a Western blot to detect HIF-1a protein on HTR8/SVneo trophoblast cells. I have used NB100-134 HIF-1 a antibody for immunostaining and will use the same Ab for Western. Now my questions are 1. What should I use for blocking? 2. Can I use HIF-1 alpha recombinant protein (H00003091-P01) as positive control? 3. What can I use as loading control? 4. Can I use DAB as substrate or anything else?
A: 1. You can use standard blocking solution which is 5% non-fat milk in TBST. 2. We recommend using Cos-7 Hypoxia Induced Nuclear Extract Kit (catalog #NB800-PC26) as the positive control for this antibody. 3. Since HIF-1 is a nuclear protein, you can use either alpha-Tubulin or beta-Tubulin as your loading control. 4. You can use DAB as your detection system.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I ordered this antibody but do not have a freezer to store it -20 degrees Celsius. Is it possible to store it for a short time (few days or weeks) at 4 degrees Celsius?
A: NB100-134 does contain Sodium Azide and should be fine at 4 degrees Celsius for about a week. We do recommend -20 degrees Celsius for storage and cannot guarantee this product will not degrade if kept at 4 degrees Celsius for more than a week.
-
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is the species reactivity of this antibody to Guinea pig based on computational homology?
A: The species reactivity reported for Guinea pig is based on published data. Here is the link for your reference. If you go through this paper carefully, on Figure 6, HIF1 expression has been tested across species including Cavia porcellus (Guinea pig).
-
Q: Our customer would like to know the shelf life of your HIF-1 alpha Antibody (Cat. # NB100-134). We would appreciate if you can confirm this information for me as soon as possible. Moreover, I was wondering whether your antibody products have a general shelf life?
A: Unless otherwise stated on our website, our antibodies are guaranteed for 1 year from the date of receipt, whist peptides and proteins are guaranteed for three months from the date of receipt. Novus guarantees that our products will work in the applications and species listed on our website and on our product datasheets. If a Novus product fails to meet this criteria, our technical service team will work with you to troubleshoot the issue. If this process is not successful, Novus will provide you with a free replacement or a 100% refund for the product.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the difference in the following two HIF-1 alpha antibodies? Catalog # NB100-105 and NB100-134
A: NB100-105 is a mouse monoclonal antibody, whereas NB100-134 is a rabbit polyclonal. They both use the same immunogen.
-
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
Associated Pathways
 HIF Repressor Pathways
HIF Repressor Pathways
 Notch Signaling Pathways
Notch Signaling Pathways


![Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody - BSA Free [NB100-134] Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody - BSA Free [NB100-134]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody---BSA-Free-Immunocytochemistry-Immunofluorescence-NB100-134-img0017.jpg)
![Immunohistochemistry-Paraffin: HIF-1 alpha Antibody - BSA Free [NB100-134] Immunohistochemistry-Paraffin: HIF-1 alpha Antibody - BSA Free [NB100-134]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody---BSA-Free-Immunohistochemistry-Paraffin-NB100-134-img0014.jpg)
![Western Blot: HIF-1 alpha AntibodyBSA Free [NB100-134] Western Blot: HIF-1 alpha AntibodyBSA Free [NB100-134]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody---BSA-Free-Western-Blot-NB100-134-img0011.jpg)
![Western Blot: HIF-1 alpha AntibodyBSA Free [NB100-134] Western Blot: HIF-1 alpha AntibodyBSA Free [NB100-134]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody---BSA-Free-Western-Blot-NB100-134-img0007.jpg)
![Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody - BSA Free [NB100-134] Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody - BSA Free [NB100-134]](https://resources.rndsystems.com/images/products/HIF-1%20alpha%20Antibody%20-%20BSA%20Free-Immunocytochemistry-Immunofluorescence-NB100-134-img0020.jpg)
![Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody - BSA Free [NB100-134] Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody - BSA Free [NB100-134]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody---BSA-Free-Immunocytochemistry-Immunofluorescence-NB100-134-img0010.jpg)
![Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody - BSA Free [NB100-134] Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody - BSA Free [NB100-134]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody---BSA-Free-Immunocytochemistry-Immunofluorescence-NB100-134-img0016.jpg)
![Immunohistochemistry-Paraffin: HIF-1 alpha Antibody - BSA Free [NB100-134] Immunohistochemistry-Paraffin: HIF-1 alpha Antibody - BSA Free [NB100-134]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody---BSA-Free-Immunohistochemistry-Paraffin-NB100-134-img0008.jpg)
![Immunohistochemistry: HIF-1 alpha Antibody - BSA Free [NB100-134] Immunohistochemistry: HIF-1 alpha Antibody - BSA Free [NB100-134]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody---BSA-Free-Immunohistochemistry-NB100-134-img0006.jpg)
![Immunohistochemistry-Paraffin: HIF-1 alpha Antibody - BSA Free [NB100-134] Immunohistochemistry-Paraffin: HIF-1 alpha Antibody - BSA Free [NB100-134]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody---BSA-Free-Immunohistochemistry-Paraffin-NB100-134-img0012.jpg)
![Simple Western: HIF-1 alpha AntibodyBSA Free [NB100-134] Simple Western: HIF-1 alpha AntibodyBSA Free [NB100-134]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody---BSA-Free-Simple-Western-NB100-134-img0009.jpg)
![Immunoprecipitation: HIF-1 alpha Antibody - BSA Free [NB100-134] Immunoprecipitation: HIF-1 alpha Antibody - BSA Free [NB100-134]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody---BSA-Free-Immunoprecipitation-NB100-134-img0001.jpg)
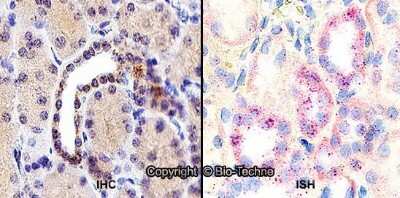
![Simple Western: HIF-1 alpha AntibodyBSA Free [NB100-134] Simple Western: HIF-1 alpha AntibodyBSA Free [NB100-134]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody---BSA-Free-Simple-Western-NB100-134-img0019.jpg)
![Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - HIF-1 alpha Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-134_rabbit-polyclonal-hif-1-alpha-antibody-271220231253384.jpg)
![Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - HIF-1 alpha Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-134_rabbit-polyclonal-hif-1-alpha-antibody-210202423454829.jpg)
![Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - HIF-1 alpha Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-134_rabbit-polyclonal-hif-1-alpha-antibody-310202415394545.jpg)
![Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody - BSA Free [NB100-134] - HIF-1 alpha Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-134_rabbit-polyclonal-hif-1-alpha-antibody-310202415384157.jpg)
![Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - HIF-1 alpha Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-134_rabbit-polyclonal-hif-1-alpha-antibody-310202415363748.jpg)
![Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - HIF-1 alpha Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-134_rabbit-polyclonal-hif-1-alpha-antibody-310202415384174.jpg)
![Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody - BSA Free [NB100-134] - HIF-1 alpha Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-134_rabbit-polyclonal-hif-1-alpha-antibody-31020241535619.jpg)
![Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody - BSA Free [NB100-134] - HIF-1 alpha Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-134_rabbit-polyclonal-hif-1-alpha-antibody-310202415382410.jpg)
![Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - HIF-1 alpha Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-134_rabbit-polyclonal-hif-1-alpha-antibody-310202415523948.jpg)
![Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - HIF-1 alpha Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-134_rabbit-polyclonal-hif-1-alpha-antibody-31020241555217.jpg)
![Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - HIF-1 alpha Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-134_rabbit-polyclonal-hif-1-alpha-antibody-31020241682166.jpg)
![Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - HIF-1 alpha Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-134_rabbit-polyclonal-hif-1-alpha-antibody-310202415532066.jpg)
![Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody - BSA Free [NB100-134] - HIF-1 alpha Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-134_rabbit-polyclonal-hif-1-alpha-antibody-310202415523935.jpg)
![Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody - BSA Free [NB100-134] - HIF-1 alpha Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-134_rabbit-polyclonal-hif-1-alpha-antibody-31020241552525.jpg)
![Chromatin Immunoprecipitation: HIF-1 alpha Antibody - BSA Free [NB100-134] - HIF-1 alpha Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-134_rabbit-polyclonal-hif-1-alpha-antibody-31020241612683.jpg)
![Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody - BSA Free [NB100-134] - HIF-1 alpha Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-134_rabbit-polyclonal-hif-1-alpha-antibody-31020241555210.jpg)
![Immunohistochemistry: HIF-1 alpha Antibody - BSA Free [NB100-134] - HIF-1 alpha Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-134_rabbit-polyclonal-hif-1-alpha-antibody-310202415535161.jpg)
![Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - HIF-1 alpha Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-134_rabbit-polyclonal-hif-1-alpha-antibody-31020241682161.jpg)
![Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - HIF-1 alpha Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-134_rabbit-polyclonal-hif-1-alpha-antibody-310202415525221.jpg)
![Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - HIF-1 alpha Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-134_rabbit-polyclonal-hif-1-alpha-antibody-310202415535194.jpg)
![Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - HIF-1 alpha Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-134_rabbit-polyclonal-hif-1-alpha-antibody-310202415541938.jpg)
![Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - HIF-1 alpha Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-134_rabbit-polyclonal-hif-1-alpha-antibody-31020241682181.jpg)
![Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - HIF-1 alpha Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-134_rabbit-polyclonal-hif-1-alpha-antibody-31020241554540.jpg)
![Western Blot: HIF-1 alpha Antibody - BSA Free [NB100-134] - HIF-1 alpha Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-134_rabbit-polyclonal-hif-1-alpha-antibody-310202415532035.jpg)
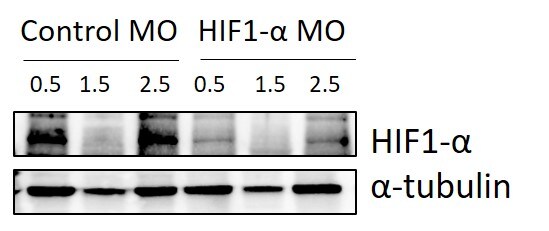

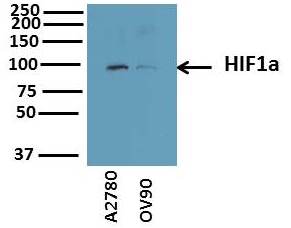
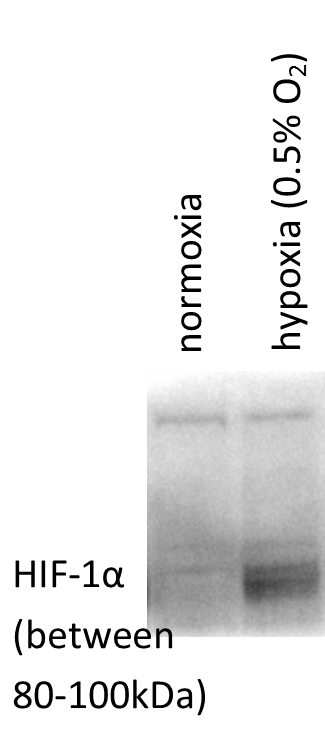
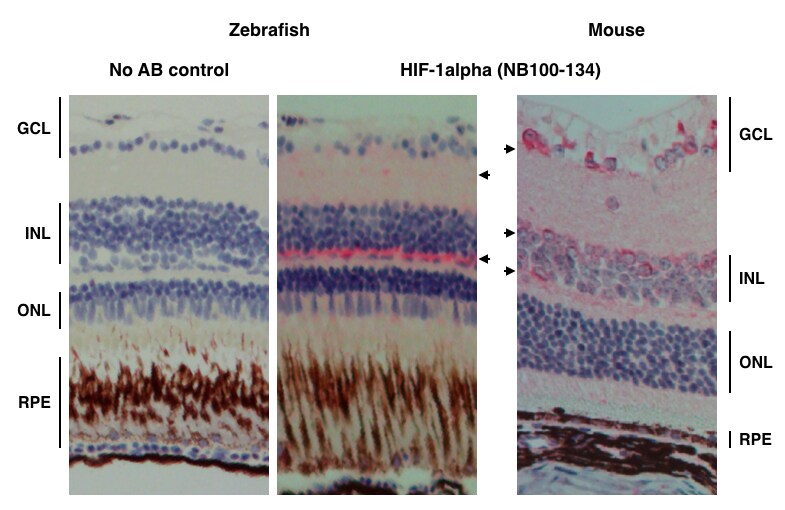
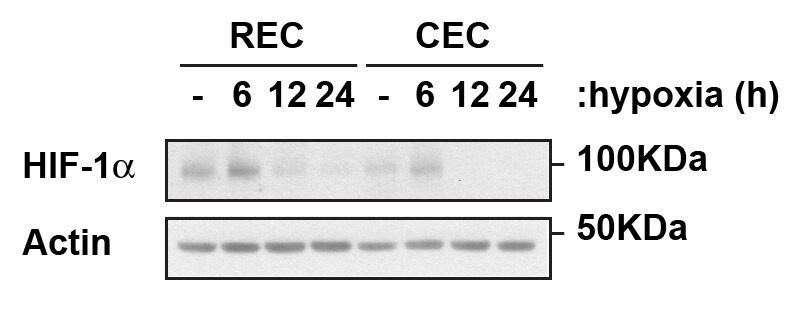
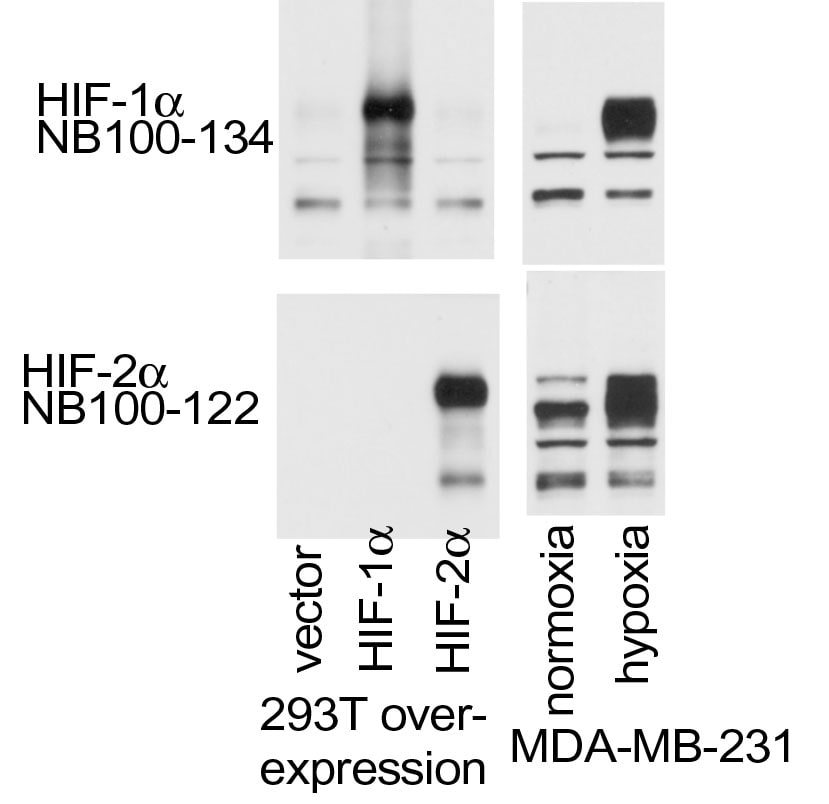
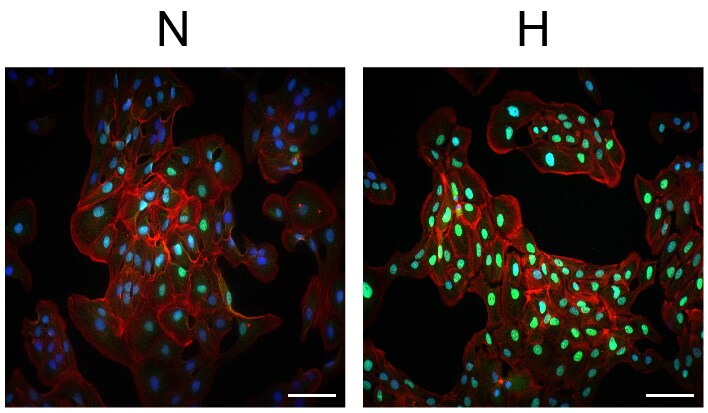
-(01-ml)_NB100-134_10191.gif)
-(01-ml)_NB100-134_9746.jpg)