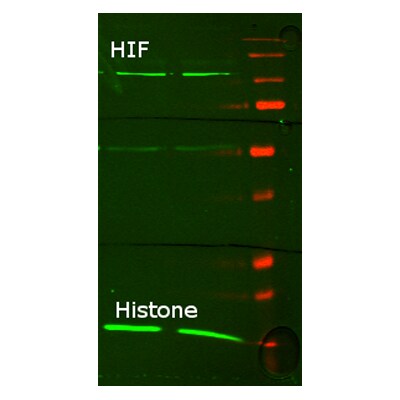
![Western Blot: HIF-1 alpha Antibody [NB100-449] Knockdown Validated: HIF-1 alpha Antibody [NB100-449]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-Knockdown-Validated-NB100-449-img0050.jpg)
Key Product Details
Validated by
Species Reactivity
Validated:
Cited:
Predicted:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Reactivity Notes
Clonality
Host
Isotype
Theoretical MW
Disclaimer note: The observed molecular weight of the protein may vary from the listed predicted molecular weight due to post translational modifications, post translation cleavages, relative charges, and other experimental factors.
Scientific Data Images for HIF-1 alpha Antibody
Simple Western: HIF-1 alpha Antibody [NB100-449]
Simple Western: HIF-1 alpha Antibody [NB100-449] - Simple Western lane view shows a specific band for HIF-1 alpha in 0.5 mg/ml of hypoxic HeLa lysate. This experiment was performed under reducing conditions using the 12-230 kDa separation system.Immunohistochemistry: HIF-1 alpha Antibody [NB100-449]
Immunohistochemistry: HIF-1 alpha Antibody [NB100-449] - Mouse Brain, Neurons 40XImmunohistochemistry: HIF-1 alpha Antibody [NB100-449]
Immunohistochemistry: HIF-1 alpha Antibody [NB100-449] - Detection of human HIF1-alpha by immunohistochemistry. Sample: FFPE section of renal cell carcinoma. Antibody: Affinity purified rabbit anti-HIF1-alpha antibody (NB110-58773). Detection: DABWestern Blot: HIF-1 alpha Antibody [NB100-449]
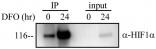
Western Blot: HIF-1 alpha Antibody [NB100-449] - Detection of Human HIF-1 alpha by Western Blot and Immunoprecipitation. Samples: Whole cell lysate (5, 15 and 50 ug for WB; 1 mg for IP, 20% of IP loaded) from HeLa cells that were either treated with cobalt chloride (+; 200 uM) or mock treated (-). Antibodies: Affinity purified rabbit anti-HIF1 alpha antibody used for WB at 0.1 ug/mL(A) and 1 ug/mL (B) and used for IP at 3 ug/mg lysate. HIF-1 alpha was also immunoprecipitated by a previous lot of this antibody. Detection: Chemiluminescence with exposure times of 30 seconds (A) and 10 seconds (B).Western Blot: HIF-1 alpha Antibody [NB100-449]
HIF-1-alpha-Antibody-Western-Blot-NB100-449-img0053.jpgImmunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody [NB100-449]
Immunocytochemistry/Immunofluorescence: HIF-1 alpha Antibody [NB100-449] - Murine primary bone marrow derived macrophages stained with HIF1-alpha antibody (red). Nuclei were counterstained with DAPI (blue). Image from verified customer review.Flow Cytometry: HIF-1 alpha Antibody [NB100-449]
Flow Cytometry: HIF-1 alpha Antibody [NB100-449] - HeLa cells were treated for 15 hrs with 200 uM CoCl2, fixed in PFA, and permeabilized in 90% MeOH. 1 X 10^6 cells were stained with 0.125 ug anti-HIF-1 alpha and secondary FITC-conjugated goat anti-rabbit (in a 150 uL reaction). Black: treated, anti-KLH control IgG; Red: untreated, anti-HIF-1 alpha; Blue: treated, anti-HIF-1 alpha.Western Blot: HIF-1 alpha Antibody [NB100-449]
Western Blot: HIF-1 alpha Antibody [NB100-449] - Samples: Whole cell lysate (5, 15 and 50 ug) from HeLa cells that were treated with cobalt chloride (+; 200 uM) or mock treated (-). Antibodies: Affinity purified rabbit anti-HIF1 alpha antibody NB100-449 used for WB at 0.1 ug/ml. Detection: Chemiluminescence with exposure times of 30 seconds.Western Blot: HIF-1 alpha Antibody [NB100-449]
Western Blot: HIF-1 alpha Antibody [NB100-449] - Analysis of total protein using anti-HIF-1 alpha, -CAIX, -ISCU antibodies and VDACs antibody (PMID: 29596470).Western Blot: HIF-1 alpha Antibody [NB100-449]
Western Blot: HIF-1 alpha Antibody [NB100-449] - Detection of human HIF1 alpha by western blot. Samples: Whole cell lysate (15 and 50 ug) from HEK293T cells that were either treated with 200 uM cobalt chloride (+) or mock treated (-). Antibodies: Affinity purified rabbit anti-HIF1 alpha antibody NB100-449 used for WB at 0.1 ug/ml. Detection: Chemiluminescence with exposure times of 3 minutes.Western Blot: HIF-1 alpha Antibody [NB100-449]
Western Blot: HIF-1 alpha Antibody [NB100-449] - 3T3-L1 mouse embryonic fibroblast adipose-like cell line. Antibody at 1:2000. WB image submitted by a verified customer review.Immunoprecipitation: HIF-1 alpha Antibody [NB100-449]
Immunoprecipitation: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha analysis in HEK293 cells. Image from verified customer review.Immunoprecipitation: HIF-1 alpha Antibody [NB100-449]
Immunoprecipitation: HIF-1 alpha Antibody [NB100-449] - Detection of human HIF1 alpha by western blot of immunoprecipitates. Samples: Whole cell lysate (1 mg for IP, 20% of IP loaded) from HEK293T cells that were treated with 200 uM cobalt chloride. Antibodies: Affinity purified rabbit anti-HIF1 alpha antibody NB100-449 used for WB at 1 ug/ml and for IP at 6 ug/mg lysate. Detection: Chemiluminescence with exposure times of 3 minutes.Western Blot: HIF-1 alpha Antibody [NB100-449] -
beta -Glucan size affects the requirement for reactive oxygen species (ROS) in IL-1 beta induction.(B) Human mDCs from healthy donors or chronic granulomatous disease (CGD) patients were stimulated with curdlan or glucan-mp for 8 h. HIF-1 alpha and pro-IL-1 beta protein expression measured by immunoblot (n = 2 donors).Western Blot: Rabbit Polyclonal HIF-1 alpha Antibody -
Western Blot: Rabbit Polyclonal HIF-1 alpha Antibody - Analysis of HIF-1 alpha antibody on mouse liver nuclear extracts. Image from a verified customer review.Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody [NB100-449] -
HIF-1 alpha /2 alpha expression in myeloid-specific KO mice targeting the HIF pathway. (A) Images of the colon of wild-type (WT), myeloid-specific Hif-1a KO (hMRP8 Hif-1a KO) or von Hippel Lindau (Vhl) KO (hMRP8 Vhl KO) mice, immunostained for MRP8 (green) & the DNA-binding regions of Hif-1a mRNA (red). Mice were fed with 5% DSS for 4 days prior to immunostaining analyses. Note that there were no MRP8-positive cells that were positive for Hif-1a mRNA in hMRP8 Hif-1a KO (middle column) mice, but we observed many cells that were double positive for MRP8 & Hif-1a mRNA in hMRP8 Vhl KO mice (right column). (B) Images of the colon of hMRP8 Vhl KO mice fed with 5% DSS as in A, immunostained for MRP8 (green) & HIF-1 alpha (red, upper row) or HIF-2 alpha (red, bottom row). DAPI-stained nuclei are shown in blue. White boxes in A & B indicate the regions magnified in the lower or right images, respectively. Yellow arrowheads in A & B indicate cells positive for both markers. Scale bars: 100 μm. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/29967068), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry: HIF-1 alpha Antibody [NB100-449] -
Immunohistochemistry: HIF-1 alpha Antibody [NB100-449] - Analysis of microvascular density & HIF-1 alpha activity. Microvascular density of pulmonary tumors was significantly higher in hyperplastic (B) & tumoral lesions (D) of NNK/NTHi treated mice compared to the NNK treated mice (A, & C) detected by CD105 immunostaining (10X, scale bar = 100 μm). HIF-1 alpha immunostaining after NNK/NTHi combined treatment showed hot-spots of high stromal expression in tumors (F), & high expression in perivascular-peribronchiolar lymphocytes (H). In contrast, low, homogenous expression of HIF-1 alpha was detected in the tumors (E) & perivascular-peribronchiolar lymphocytes (G) of NNK treated mice (40X, scale bar = 25 μm). Image collected & cropped by CiteAb from the following publication (https://molecular-cancer.biomedcentral.com/articles/10.1186/1476-4598-1…), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody [NB100-449] -
Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody [NB100-449] - Chronic ethanol feeding enhances tumorigenesis markers during AOM/DSS-induced tumorigenesis. AOM/DSS-induced colonic tumorigenesis was induced with (EF) or without (PF) 4 % ethanol feeding as described in Methods section. Cryosections of distal colon were stained for VEGF (a) pSmad (b), or HIF1 alpha (c), & co-stained for F-actin & nucleus. Images presented are representative of n of 5 for each group Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/26951793), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry-Paraffin: HIF-1 alpha Antibody [NB100-449] -
Immunohistochemistry-Paraffin: HIF-1 alpha Antibody [NB100-449] - Treatment with HBOT or PFD decreased HIF-1 alpha expression in the AD lesions.IHC for HIF-1 alpha (A). Scale bar is 100 µm. HIF-1 alpha + area was measured in five fields chosen at random by using image analysis software (B). RT-PCR for HIF-1 alpha (C). NT, not treated. Data are expressed as the mean ± SD (n = 12). *P<0.05, compared with the control group; ¶P<0.05, compared with the AD-NT group; §, P<0.05, compared with the AD-Steroid group. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/25275529), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry-Paraffin: HIF-1 alpha Antibody [NB100-449] -
Immunohistochemistry-Paraffin: HIF-1 alpha Antibody [NB100-449] - Elevated PIK3/AKT/HIF-1 alpha pathways in KL SqCC tumours.(a,b) Analysis of TCGA gene expression (normalized TPM) & PIK3CA (a) & PTEN (b) genomic copy number alteration profiles. Each dot represents one SqCC patient (n=501). Boxes represent the interquartile range & whiskers are drawn to the minimum & maximum. Kruskal–Wallis non-parametric ANOVA, ****P<0.0001, ***P<0.001, **P<0.01. (c) IHC analysis (top) & quantification (bottom) of p63, p-AKT, p-S6, p-4EBP1, HIF-1 alpha & GLUT1 in KL tumours (n=6 each group). Two-tailed t-test, ****P<0.0001, ***P<0.001, **P<0.01. Scale bar, 50 μm. (d) Immunoblot analysis of HIF-1 alpha & GLUT1 in control shGFP & shHIF-1 alpha knockdown SqCC cell lines, HCC95 & HCC1588. All error bars represent the mean±s.e.m. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/28548087), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry: HIF-1 alpha Antibody [NB100-449] -
Immunohistochemistry: HIF-1 alpha Antibody [NB100-449] - Analysis of microvascular density & HIF-1 alpha activity. Microvascular density of pulmonary tumors was significantly higher in hyperplastic (B) & tumoral lesions (D) of NNK/NTHi treated mice compared to the NNK treated mice (A, & C) detected by CD105 immunostaining (10X, scale bar = 100 μm). HIF-1 alpha immunostaining after NNK/NTHi combined treatment showed hot-spots of high stromal expression in tumors (F), & high expression in perivascular-peribronchiolar lymphocytes (H). In contrast, low, homogenous expression of HIF-1 alpha was detected in the tumors (E) & perivascular-peribronchiolar lymphocytes (G) of NNK treated mice (40X, scale bar = 25 μm). Image collected & cropped by CiteAb from the following publication (https://molecular-cancer.biomedcentral.com/articles/10.1186/1476-4598-1…), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry: HIF-1 alpha Antibody [NB100-449] -
Immunohistochemistry: HIF-1 alpha Antibody [NB100-449] - Immunolocalization of HIF1A in limbs.Immunoreactivity was detected in control limbs in the apical ectodermal ridge & interdigital regions (I.R.). The mesenchymal condensations/developing cartilaginous anlagen (Digits) were not immunoreactive. 4-OOHCPA treatment resulted in a concentration- & time-dependent increase in HIF1A immunoreactivity in this area in the 3 & 6 h exposure groups; staining was diminished by 24 h. Four separate replicates were done. Image collected & cropped by CiteAb from the following publication (https://dx.plos.org/10.1371/journal.pone.0051937), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody [NB100-449] -
Western Blot: HIF-1 alpha Antibody [NB100-449] - Effects of cell stress on VEGF-A & HIF1 alpha expression in Müller cells & Y79 photoreceptors. (A) VEGF-A was measured by ELISA using conditioned media collected from MIO-M1 & Y79 cells, n = 4–5/group in Müller cells & n = 8/group in Y79 photoreceptors, ** p < 0.01; (B) Western blots for HIF1 alpha using cellular proteins, n = 4/group, ** p < 0.01. For MIO-M1 Müller cells: Grey bars: 5 mM glucose. Black bars: 25 mM glucose. For Y79 photoreceptors: Grey bars: 11 mM glucose. Black bars: 25 mM glucose. NS, not significant; LG, low glucose; HG, high glucose. Image collected & cropped by CiteAb from the following publication (http://www.mdpi.com/1422-0067/18/3/533), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry: HIF-1 alpha Antibody [NB100-449] -
Immunohistochemistry: HIF-1 alpha Antibody [NB100-449] - Analysis of microvascular density & HIF-1 alpha activity. Microvascular density of pulmonary tumors was significantly higher in hyperplastic (B) & tumoral lesions (D) of NNK/NTHi treated mice compared to the NNK treated mice (A, & C) detected by CD105 immunostaining (10X, scale bar = 100 μm). HIF-1 alpha immunostaining after NNK/NTHi combined treatment showed hot-spots of high stromal expression in tumors (F), & high expression in perivascular-peribronchiolar lymphocytes (H). In contrast, low, homogenous expression of HIF-1 alpha was detected in the tumors (E) & perivascular-peribronchiolar lymphocytes (G) of NNK treated mice (40X, scale bar = 25 μm). Image collected & cropped by CiteAb from the following publication (https://molecular-cancer.biomedcentral.com/articles/10.1186/1476-4598-1…), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody [NB100-449] -
Western Blot: HIF-1 alpha Antibody [NB100-449] - Phenylhydrazine administration results in hypoxia in retinal vessels & in HIF-1 alpha & HIF-2 alpha stabilization in peripheral blood leukocytes.(A) 3-dimensional reconstructed imaging of the superficial vascular plexus of flat mounted retinae of mice (steady state, treated with PHZ & treated with PHZ & peak EIU induction) stained with DAPI, hypoxyprobe & Isolectin B4. (B) Representative analysis of HIFs by western blot: 50ug of protein from total cell lysate of blood leukocytes isolated from mice treated with PHZ compared to untreated; (C) relative densitometry quantification of HIF western blots from PBMC showing mean, n = 3, error bar showing SD, unpaired t test, ***P < 0.001. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/28112274), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry-Frozen: HIF-1 alpha Antibody [NB100-449] -
Immunohistochemistry-Frozen: HIF-1 alpha Antibody [NB100-449] - Ethanol elevates tumorigenic markers & nuclear localization of HIF1 alpha in colonic crypts during AOM/DSS-induced tumorigenesis: AOM/DSS-induced colonic tumorigenesis was induced with or without 4 % ethanol feeding as described in Methods section. a & b Colonic mucosal extracts were immunoblotted for pSmad & VEGF (a), & the band density evaluated using Image J software (b). Values are mean ± SE (n = 3). Asterisks indicate the values that are significantly different (p < 0.05) from corresponding values for AOM/DSS group. c Cryosections of colon were stained for HIF1 alpha (red) & nucleus (blue). Sections of the images from Fig. 2c are enlarged to show the detailed localization of HIF1 alpha in the nucleus. While arrows indicate nuclear co-localization of HIF1 alpha Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/26951793), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody [NB100-449] -
Western Blot: HIF-1 alpha Antibody [NB100-449] - Dectin-1-induced NADPH oxidase-derived ROS is required for augmented IL-1 beta during TSLPR inhibition. (A) Human mDC derived from healthy donors (HD) or CGD patients were stimulated with SC glucan for 8 h (n = 2 representative donors presented, three separate experiments performed). Pro-IL-1 beta, IL-1 beta, HIF-1 alpha, phospho-AMPK, AMPK & beta -actin were measured by immunoblot. (B–E) Densitometry of cumulative data was performed using Image Studio Lite software with pro-IL-1 beta, IL-1 beta & HIF-1 alpha normalized to beta -actin & phospho-AMPK normalized to total AMPK. Data is reported as percentage of maximal signal observed within each donor (n = 3 independent donors, presented as pooled data). (F–H) Human mDC derived from HD or CGD patients were stimulated with SC glucan, CA glucan or heat killed C. albicans hyphae with anti-TSLPR or IgG isotype control antibodies for 24 h (n = 3 independent donors, presented as pooled data). IL-1 beta was measured in 24-h cell culture supernatants by ELISA. Cumulative data displayed as mean +SEM. Statistical analysis calculated using one-way ANOVA with Bonferroni post-tests (***p = 0.001,*p = 0.05). Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31139177), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody [NB100-449] -
Western Blot: HIF-1 alpha Antibody [NB100-449] - Reactive oxygen species production & expression of stabilized HIF1 alpha & AMPK subunit alpha 2 & in H1299 & H1299r cells(A) H1299, H1299r, P31 & P31r cell were seeded at a density of 250,000 cells/well & left for 24 h. The cells were then stained with 10 μmol/L dichloro-dihydro-fluorescein diacetate (DCFH-DA). Fluorescence was analysed for 10,000 events. Whole cell lysates were prepared from confluent cultures of H1299, H1299r cells. Proteins (90 μg) were resolved in 10% SDS-PAGE gels & transferred to a PVDF membrane. (B) Representative blot of HIF1 alpha protein expression. (C) Representative blot of AMPK subunit alpha 2 protein expression from four independent experiments. (D) Bar chart of AMPK subunit alpha 2 mean for four separate measurements. Statistical analyses was carried out using the student t-test. ** = p<0.01, * = p<0.05. Image collected & cropped by CiteAb from the following publication (https://www.oncotarget.com/lookup/doi/10.18632/oncotarget.21885), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody [NB100-449] -
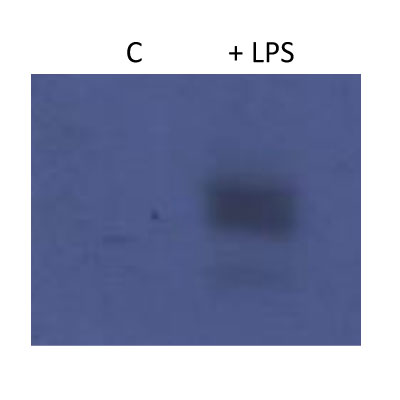
Western Blot: HIF-1 alpha Antibody [NB100-449] - Pyruvate kinase isoform M2 (PKM2) & hypoxia-inducible factor 1 alpha (Hif-1 alpha ) bind directly to the promoter region of PDL1 in primary murine BMDM cells. BMDM cells were treated with TEPP-46 at 50 µM for 1 h prior to stimulation with LPS (100 ng/ml, 24 h) (A). Binding of PKM2 (left) or Hif-1 alpha (right) to HRE1 or HRE4 of the PD-L1 promoter was detected by incubating cell lysates with biotinylated oligonucleotides spanning the relevant HRE promoter region. Protein–oligonucleotide complexes were isolated using streptavidin agarose beads, & proteins were detected by western blotting. Representative of n = 3. Chromatin immunoprecipitation (ChIP)-PCR (B) using PKM2 & HIF-1 alpha antibodies & primers specific for three promoter regions of PD-L1 (HRE1, 2–3, & 4) & a known Hif-1 alpha binding region of vascular endothelial growth factor as a positive control showing binding of Hif-1 alpha (left) & PKM2 (right) to the PD-L1 promoter in LPS-treated BMDMs (100 ng/ml, 24 h). (C) Sequential ChIP assays measuring simultaneous endogenous binding of PKM2 & Hif-1 alpha to chromatin in response to LPS (100 ng/ml, 24 h) ±TEPP-46 (pretreatment using 50 µM, 60 min). ChIP data are calculated as percent of input, error bar represents mean ± SEM, & statistics are performed as two-tailed unpaired t-test *P < 0.05, **P < 0.01, & ***P < 0.001. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/29081778), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody [NB100-449] -
Western Blot: HIF-1 alpha Antibody [NB100-449] - Changes in Hepatic Oxygen Saturation & Hypoxia-related Protein Expression. (A) In the representative case, the hepatic oxygen saturation in the experimental group was lower than that in the control group at four, six, & eight weeks (51.6 vs. 54.5%, 48.4 vs. 53.9%, 32.3 vs. 43.2%, & 31.2 vs. 42.9%). (B) There was significant decrease of oxygen saturation of liver parenchyma in the experimental group compared to those of control group (45.2 vs. 51.9%, 43.0 vs. 49.0%, & 31.3 vs. 49.7%) at the four, six, & eight weeks, respectively (P < 0.001). (C) In the Western blot analysis, the experimental group was relatively higher expression of hypoxia-inducible factor-1 alpha (HIF-1 alpha ) & vascular endothelial growth factor (VEGF) protein than the control group at eight weeks. The full-length blots with these antibodies were presented in supplementary Figure S1. (D,E) The experimental group showed significantly higher expression of HIF-1 alpha & VEGF protein than the control group at eight-week (1.47 ± 0.48 vs. 0.19 ± 0.08 & 0.96 ± 0.23 vs. 0.33 ± 0.06, P = 0.019 & P < 0.018). *P < 0.05. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/29079853), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody [NB100-449] -
Western Blot: HIF-1 alpha Antibody [NB100-449] - BCAT1 expression & inhibition in human macrophages.(a) RNA-sequencing in human monocyte-derived macrophages (hMDMs) shows mRNA copies of BCAT enzymes in normalized counts. n=14 healthy donor hMDMs were used for RNA-seq. (b) Chemical structure of ERG240 & leucine. (c) Enzymatic activity of recombinant human BCAT1 & BCAT2 in presence of ERG240. (d) Relative expression of IRG1, HIF1A & IL1B measured by qRT-PCR in control, ERG240-treated (20 mM, 3 h), LPS treated (100 ng ml−1, 3 h), & LPS+ERG240 treated (LPS, 100 ng ml−1; ERG240, 20 mM for 3 h) hMDMs. ns, non-significant. All expression values are normalized to those obtained for HPRT gene expression. n=3 healthy donor hMDMs were used in each group. (e) Western blot analysis of IRG1, IL-1 beta, HIF-1 alpha & beta-actin (ACTB) in control (untreated), ERG240-treated (20 mM, 3 h), LPS treated (100 ng ml−1, 3 h) & LPS+ERG240 treated (LPS, 100 ng ml−1; ERG240, 20 mM for 3 h) hMDMs. The experiment is representative of three independent experiments using n=3 healthy donor hMDMs each. (f) GC/MS results showing itaconic acid production in hMDMs in presence or absence of ERG240 (20 mM). LPS treatment was for 8 h (100 ng ml−1). GC/MS plot for each donor is shown separately where itaconate abundance denotes arbitrary units. Error bars are s.e.m. Significance was tested using two-tailed Student’s t-test or one-way ANOVA. * P<10−3 following two-way ANOVA. Image collected & cropped by CiteAb from the following publication (https://www.nature.com/articles/ncomms16040), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody [NB100-449] -
Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha /2 alpha expression in myeloid-specific KO mice targeting the HIF pathway. (A) Images of the colon of wild-type (WT), myeloid-specific Hif-1a KO (hMRP8 Hif-1a KO) or von Hippel Lindau (Vhl) KO (hMRP8 Vhl KO) mice, immunostained for MRP8 (green) & the DNA-binding regions of Hif-1a mRNA (red). Mice were fed with 5% DSS for 4 days prior to immunostaining analyses. Note that there were no MRP8-positive cells that were positive for Hif-1a mRNA in hMRP8 Hif-1a KO (middle column) mice, but we observed many cells that were double positive for MRP8 & Hif-1a mRNA in hMRP8 Vhl KO mice (right column). (B) Images of the colon of hMRP8 Vhl KO mice fed with 5% DSS as in A, immunostained for MRP8 (green) & HIF-1 alpha (red, upper row) or HIF-2 alpha (red, bottom row). DAPI-stained nuclei are shown in blue. White boxes in A & B indicate the regions magnified in the lower or right images, respectively. Yellow arrowheads in A & B indicate cells positive for both markers. Scale bars: 100 μm. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/29967068), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody [NB100-449] -
Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody [NB100-449] - Increased infiltration of myeloid cells expressing HIF-1 alpha in DSS-induced colitis. (A) Western blot analysis of MRP8, F4/80 or actin using the whole colon tissue lysate from mice fed with water or 5% DSS for 4 days. (B) Images of the colon of mice fed with water or 5% DSS, immunostained for myeloid cells using anti-MRP8 (green) or anti-HIF-1 alpha (red) antibodies. Nuclei were counterstained with DAPI. White rectangles indicate the areas magnified in the images shown below. Yellow arrowheads indicate cells with colocalization of MRP8 & HIF-1 alpha. Quantification of MRP8-positive cells & percentage of those expressing HIF-1 alpha are shown as bar graphs on the right-hand side. Data are mean±s.e.m. for at least three independent fields examined per mouse (n≥3 per group). **P<0.01 & ***P<0.001, assessed by Student's t-test. HPF, high-powered field. (C) Images of the colon of mice fed with 5% DSS, immunostained for MRP8 (red) & CD11b (green). DAPI-stained nuclei are shown in blue & a merged image is also shown. Scale bars: 100 μm. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/29967068), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody [NB100-449] -
Western Blot: HIF-1 alpha Antibody [NB100-449] - Increased HIF-1 alpha expression is regulated by TSLPR inhibition & not IL-1 beta secretion (A) Human mDC were stimulated with SC glucan with either anti-TSLPR or IgG isotype control antibodies for 8 h in the presence or absence of IL-1 beta neutralization antibodies (n = 1 representative donor presented, three separate experiments performed). Pro-IL-1 beta, IL-1 beta, HIF-1 alpha, phospho-p38 MAPK, p38 MAPK, phospho-AMPK, AMPK & beta -actin were measured by immunoblot. (B,C) Densitometry of cumulative data was performed using Image Studio Lite software with HIF-1 alpha normalized to beta -actin & phospho-AMPK normalized to AMPK. Data is reported as percentage of maximal signal observed within each donor (n = 3 independent donors, presented as pooled data). Cumulative data displayed as mean +SEM. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31139177), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody [NB100-449] -
Western Blot: HIF-1 alpha Antibody [NB100-449] - miR-18b (miR-18b-5p) regulates Hif1 alpha & reduces apoptosis in mtNSC-34 cells. a Overexpressed miR-18b (miR-18b-5p) decreased Hif1 alpha & Mef2c proteins. Both Mctp1 & Rarb expression were increased by miR-18b (miR-18b-5p). Downregulated Bax & upregulated Bcl2 by miR-18b (miR-18b-5p) diminished apoptosis in mtNSC-34 cells. b RT-qPCR analysis showed low expression of Hif1 alpha & Mef2c mRNAs. c Mctp1 & Rarb transcripts were highly expressed by miR-18b (miR-18b-5p). d Bax mRNAs were decreased & Bcl2 mRNAs were increased by overexpressed miR-18b. e miR-18b (miR-18b-5p) was overexpressed in mtNSC-34 cells. f miR-206 was reduced by miR-18b (miR-18b-5p). g LDH release analysis explained that transfected miR-18b (miR-18b-5p) restores apoptosis. h Luciferases assay with 3′ UTR of Hif1 alpha showed that Hif1 alpha is target of miR-18b in contNSC-34 cells. i & j Overexpression of miR-18b (miR-18b-5p) enhanced neuronal differentiation (MAP2) & attenuated intracellular Ca2+ levels (Cont (0.098) versus miR-18b (miR-18b-5p) (0.051) in fluorescence intensities from baseline 490/525 ratio) in mtNSC-34 cells. Empty vector served as a negative control (Cont). Arrow represents SOD1 aggregation (green). Scale bar, 40 μm. Significantly different at *, p < 0.05; **, p < 0.005. The experiments were replicated 5 times Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/32605607), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody [NB100-449] -
Western Blot: HIF-1 alpha Antibody [NB100-449] - Hypoxic conditions upregulate CNN3 in BeWo cells.BeWo cells were cultured either under normoxic or under hypoxic conditions for 24(A) Total protein lysates were examined with the Western Blot technique to detect CNN3 protein levels. For normalization, HPRT was stained on the same membrane. As hypoxia marker, HIF-1 alpha was detected as well. (B) Protein bands of CNN3 & HPRT were densitometrically measured on the Western Blot membrane & CNN3/HPRT levels are plotted in the graph (white column: normoxia; black column: hypoxia). n = 3. (C) BeWo cells were serum starved for 16 h & then treated with either PBS as control or 200 µM CoCl2 for 6 h in serum free medium. Then total protein was isolated & a Western Blot was performed. The HIF-1 alpha, CNN3 & HPRT protein was detected on the membrane with specific antibodies. (D) A densitometric analysis was performed to determine CNN3/HPRT levels (white column: normoxia; black column: hypoxia). n = 3. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/25050546), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody [NB100-449] -
Western Blot: HIF-1 alpha Antibody [NB100-449] - Dynamics of the hypoxia-triggered decrease in MET phosphorylation & its reversal upon reoxygenation(A) MCF10A cells were incubated under normoxia or hypoxia for 1 h. They were then treated, under the same oxygen pressure, with 10 ng/mL HGF/SF for 5, 10, 20, 30, 60, 90 & 120 minutes. A control (Ctrl) without any HGF/SF stimulation was also performed. The same amount of protein was analyzed by western blotting with antibodies directed against: phosphorylated residues in the MET kinase domain, the MET kinase domain, the hypoxia marker HIF1a, phosphorylated Akt, Akt, Erk2, phosphorylated Erk, & actine. The positions of prestained molecular weight markers are indicated. Arrows indicate the positions of precursor & mature full-length MET. (B) MCF10A cells were incubated under hypoxia for 5, 10, 15 or 30 minutes. Another set of cells were incubated under hypoxia for 1 h & then returned to normoxia for 5, 10, 15 or 30 min (re-oxygenation). A control under normoxic (N) conditions was also included. The same amount of protein was analyzed by western blotting as previously described with the addition of GAB1 & its phosphorylated form. (C) MCF10A cells were placed under normoxic or hypoxic conditions for 1 h or hypoxia for 1 h & then normoxia for 10 minutes (reoxygenation). Cells were then treated at the indicated time with 10 ng/mL of HGF/SF. Cell lysates were incubated for AlphaScreen specific phospho-Erk & phospho-Akt quantitation. Error bars represent standard deviations (n = 3; ± SD). Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/29930749), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody [NB100-449] -
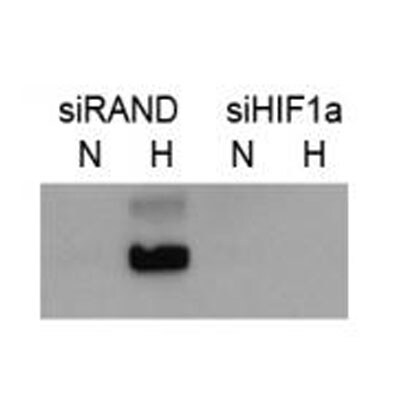
Western Blot: HIF-1 alpha Antibody [NB100-449] - The role of HIF-1 in purinosome formation.a, quantifying the number of purinosome-containing cells in normoxia or hypoxia (24 h) in purine-rich medium & normoxia in purine-depleted medium (purine -ve), cells in hypoxia transfected with siRNA to HIF-1 alpha (+ siRNA), & cells in purine-rich medium supplemented with DFX. Data shown are n = 3, mean ± S.E., total number of cells counted are shown in parentheses. b, time course of purinosome formation in hypoxia shows the number of purinosome-containing cells steadily increases after 3 h in hypoxia. Re-oxygenation of the samples after hypoxic incubation for 10 h reverts the number purinosome-containing cells back to normoxic levels. Data shown is n = 3, mean ± S.E., total number of cells counted are shown in parentheses. c, time course of HIF-1 alpha stabilization in hypoxia shows maximum HIF-1 alpha protein expression levels at 3 h in hypoxia, after which the HIF-1 alpha expression decreases. The positions of molecular markers are shown for each blot; uncropped blots with overlaid markers are deposited in the raw data files. d, the effect of hypoxia on the transcription of purine biosynthesis enzymes measured by qPCR. Vascular endothelial growth factor (VEGF) & HIF-1 alpha are controls. Data shown are n = 5, mean ± S.E. e, the effect of hypoxia on the protein expression levels of the purine biosynthetic enzymes. HIF-1 alpha is stabilized in hypoxia as expected, & no significant increase in the purine enzymes was detected between normoxic (21% oxygen) & hypoxic (1% oxygen) growth conditions. The positions of molecular markers surrounding each band of interest are shown for each blot; uncropped blots with overlaid markers are deposited in the raw data files. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/32439803), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody [NB100-449] -
Western Blot: HIF-1 alpha Antibody [NB100-449] - C/EBP delta promotes HIF-1 alpha expression in macrophages through inhibition of FBXW7 alpha a) RT-PCR analysis of FBXW7 isoform expression from different sources as follows. 1, primary peritoneal macrophages (PPMs); 2, RAW 264.7 cells; 3, MMTV-Neu mammary tumour tissue; 4, primary mouse embryo fibroblasts. Numbers indicate the position of size markers in base pairs. (b) RT-qPCR analysis of Fbxw7 transcript levels in PPMs from WT & Cebpd−/− KO mice, cultured +/− LPS (100 ng/ml, 24 h), compared to WT untreated (n=4, *P<0.05; **P<0.001). (c) Western analysis of nuclear extract (NE) from primary human monocytes nucleofected with siRNA oligos (C, control; D, CEBPD; F, FBXW7) & treated with LPS (100 ng/ml) & 1%O2 (16 h) as indicated. SE, short exposure; LE, long exposure. (d) RT-qPCR analysis of FBXW7 & CEBPD transcripts in primary human monocytes as in panel (c) (n=3, *P<0.05; **P<0.001). (e) Western analysis of NE from PPMs nucelofected with siRNA oligos & treated with LPS (100 ng/ml) & 1%O2 for 16 h as indicated. SE, short exposure; LE, long exposure.Where applicable, data are mean±S.E.M., evaluated by two-tailed unequal variance t-test. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/23575666), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody [NB100-449] -
Western Blot: HIF-1 alpha Antibody [NB100-449] - Hypoxia downregulates CFTR expression. (a) Phase-contrast photographs of MDCK cells treated with normoxia & hypoxia conditions; (b) Western blot showing downregulation of CFTR in the MDCK cells by hypoxia (Full-length blot is shown in Supplementary Figure S7b.); (c) Real time-PCR assay showing decreased mRNA expression of CFTR induced by hypoxia in HK-2 cells,*p < 0.05; (d) Western blot showing decreased expression of CFTR induced by hypoxia in HK-2 cells (Full-length blot is shown in Supplementary Figure S7c.); (e) Western blot showing the expression changes of HIF-1 alpha & EMT markers induced by hypoxia in MDCK cells, quantification analysis is shown in the lower panel, *p < 0.05; (Full-length blot is shown in Supplementary Figure S7d.) (f) Immunofluorescent staining showing dramatically increased HIF-1 alpha & reduced CFTR protein levels in tubular epithelial cells at inner cortices in UUO kidney (arrow). Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/28701694), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody [NB100-449] -
Western Blot: HIF-1 alpha Antibody [NB100-449] - NRP1 & MET on HT1080 cells are accessible for blood-borne rhodocetin-alpha beta (A) expression of NRP1 & MET by HT1080 cells was proven by flow cytometry. Gray, isotype-matched controls. (B) flow cytometry of NRP1-knockout HT1080 cells demonstrating their NRP1-deficiency & unaffected MET expression. (C) treatment with CoCl2, mimicking a hypoxic tumor micro-environment, induced upregulation of HIF-1 alpha. (D) NRP1, as a downstream target of HIF-1 alpha, is upregulated in HT1080 cells but not in NRP1-knockout HT1080 cells. beta -actin immunoblots show even loading. (E) increased HIF-1 alpha (red) levels in hypoxic tumor regions, which also contained partly (arrows) or completely (open arrows) EC-deficient VM vessels. ECs are stained green & nuclei blue. (F) immunostaining of NRP1 (red) & MET (blue) showed that both proteins were present on HT1080 cells & ECs in tumor tissue. Note the continuity between EC-lined vasculature & EC marker-deficient vessels (arrows in F’). F’, F’’, in oblique view, gating of the green CD31 signal also showed an apical absence of NRP1 on ECs in contrast to MET (open arrows). (G) the fluorescence intensity along a traceroute, averaged over a width of 5 pixels, (rectangle in F) through the endothelium revealed that in ECs NRP1, unlike MET, is absent from the apical side & restricted to the basolateral side. In contrast, on ATV/VM-lining cells (F’, arrows) both NRP1 & MET are accessible from the bloodstream (H). Vertical gray lines in G & H indicate the position of the apical cell border. Original magnification was 400× (E) & 630× (F-F’’). Representative images are shown. Image collected & cropped by CiteAb from the following publication (https://www.oncotarget.com/lookup/doi/10.18632/oncotarget.25032), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry: HIF-1 alpha Antibody [NB100-449] -
Immunohistochemistry: HIF-1 alpha Antibody [NB100-449] - NRP1 & MET on HT1080 cells are accessible for blood-borne rhodocetin-alpha beta (A) expression of NRP1 & MET by HT1080 cells was proven by flow cytometry. Gray, isotype-matched controls. (B) flow cytometry of NRP1-knockout HT1080 cells demonstrating their NRP1-deficiency & unaffected MET expression. (C) treatment with CoCl2, mimicking a hypoxic tumor micro-environment, induced upregulation of HIF-1 alpha. (D) NRP1, as a downstream target of HIF-1 alpha, is upregulated in HT1080 cells but not in NRP1-knockout HT1080 cells. beta -actin immunoblots show even loading. (E) increased HIF-1 alpha (red) levels in hypoxic tumor regions, which also contained partly (arrows) or completely (open arrows) EC-deficient VM vessels. ECs are stained green & nuclei blue. (F) immunostaining of NRP1 (red) & MET (blue) showed that both proteins were present on HT1080 cells & ECs in tumor tissue. Note the continuity between EC-lined vasculature & EC marker-deficient vessels (arrows in F’). F’, F’’, in oblique view, gating of the green CD31 signal also showed an apical absence of NRP1 on ECs in contrast to MET (open arrows). (G) the fluorescence intensity along a traceroute, averaged over a width of 5 pixels, (rectangle in F) through the endothelium revealed that in ECs NRP1, unlike MET, is absent from the apical side & restricted to the basolateral side. In contrast, on ATV/VM-lining cells (F’, arrows) both NRP1 & MET are accessible from the bloodstream (H). Vertical gray lines in G & H indicate the position of the apical cell border. Original magnification was 400× (E) & 630× (F-F’’). Representative images are shown. Image collected & cropped by CiteAb from the following publication (https://www.oncotarget.com/lookup/doi/10.18632/oncotarget.25032), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody [NB100-449] -
Western Blot: HIF-1 alpha Antibody [NB100-449] - C/EBP delta promotes HIF-1 alpha expression in macrophages through inhibition of FBXW7 alpha a) RT-PCR analysis of FBXW7 isoform expression from different sources as follows. 1, primary peritoneal macrophages (PPMs); 2, RAW 264.7 cells; 3, MMTV-Neu mammary tumour tissue; 4, primary mouse embryo fibroblasts. Numbers indicate the position of size markers in base pairs. (b) RT-qPCR analysis of Fbxw7 transcript levels in PPMs from WT & Cebpd−/− KO mice, cultured +/− LPS (100 ng/ml, 24 h), compared to WT untreated (n=4, *P<0.05; **P<0.001). (c) Western analysis of nuclear extract (NE) from primary human monocytes nucleofected with siRNA oligos (C, control; D, CEBPD; F, FBXW7) & treated with LPS (100 ng/ml) & 1%O2 (16 h) as indicated. SE, short exposure; LE, long exposure. (d) RT-qPCR analysis of FBXW7 & CEBPD transcripts in primary human monocytes as in panel (c) (n=3, *P<0.05; **P<0.001). (e) Western analysis of NE from PPMs nucelofected with siRNA oligos & treated with LPS (100 ng/ml) & 1%O2 for 16 h as indicated. SE, short exposure; LE, long exposure.Where applicable, data are mean±S.E.M., evaluated by two-tailed unequal variance t-test. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/23575666), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody [NB100-449] -
Western Blot: HIF-1 alpha Antibody [NB100-449] - Dectin-1-induced TSLP negatively regulates pro-IL-1 beta & HIF-1 alpha. (A) Human mDC were stimulated with SC glucan, CA glucan or heat killed C. albicans hyphae with anti-TSLPR antibodies or IgG isotype control for 24 h (n = 6 independent donors, presented as pooled data). Lactate production was measured in cell-culture supernatants using colourmetric L-lactate detection kit. (B) Human mDC were stimulated SC glucan with either anti-TSLP, anti-TSLPR or IgG isotype control antibodies for 8 h (n = 1 representative donor presented, three separate experiments performed). Pro-IL-1 beta, IL-1 beta, HIF-1 alpha, phospho-p38 MAPK, p38 MAPK, phospho-AMPK, AMPK & beta -actin were measured by immunoblot. (C–G) Densitometry of cumulative data was performed using Image Studio Lite software with pro-IL-1 beta, IL-1 beta & HIF-1 alpha normalized to beta -actin & phospho-p38 MAPK & phospho-AMPK normalized to total p38 MAPK & AMPK respectively. Data is reported as percentage of maximal signal observed within each donor (n = 3 independent donors, presented as pooled data). Cumulative data displayed as mean +SEM. Statistical analysis calculated using one-way ANOVA with Bonferroni post-tests (***p = 0.001). Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31139177), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody [NB100-449] -
Western Blot: HIF-1 alpha Antibody [NB100-449] - Involvement of MET in Akt & Erk pathway activation under hypoxiaMCF10A cells were transfected with a pool of three MET-targeting siRNAs (20 nM) or a control siRNA (siCtrl). A control without siRNA was also included (Ctrl) (A). MCF10A cells were transfected with two HIF1a-targeting siRNAs (20 nM), independently or together, or a control siRNA (siCtrl) (B). The cells were then placed for 1 h under normoxic or hypoxic conditions & treated or not for 10 min with 10 ng/mL HGF/SF. In each experiment, the same amount of protein was analyzed by western blotting with antibodies directed against: phosphorylated residues in the MET kinase domain, the MET kinase domain, phosphorylated Akt, Akt, phosphorylated Erk, Erk2, phosphorylated GAB1, GAB1, or hypoxia marker HIF1a. The positions of prestained molecular weight markers are indicated. Arrows indicate the positions of precursor & mature full-length MET & Erk1/2 proteins. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/29930749), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody [NB100-449] -
Western Blot: HIF-1 alpha Antibody [NB100-449] - beta -Glucan size affects the requirement for reactive oxygen species (ROS) in IL-1 beta induction. (A) Human monocyte-derived dendritic cell (mDC) stimulated with curdlan or glucan-mp for 8 h. ROS were detected by incubating cells with CellRox Green fluorescence dye & analysis by flow cytometry (representative experiment of two) presented, (B) Human mDCs from healthy donors or chronic granulomatous disease (CGD) patients were stimulated with curdlan or glucan-mp for 8 h. HIF-1 alpha & pro-IL-1 beta protein expression measured by immunoblot (n = 2 donors). (C–L) Human mDC from healthy donors or CGD patients were stimulated with curdlan or glucan-mp for 24 h (n = 6 donors). IL-1 beta, IL-6, IL-23, TSLP, & CCL22 secretion were measured by enzyme-linked immunosorbent assay, with cumulative data displayed as mean ± SEM. Image collected & cropped by CiteAb from the following publication (http://journal.frontiersin.org/article/10.3389/fimmu.2017.00791/full), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Applications for HIF-1 alpha Antibody
Chromatin Immunoprecipitation (ChIP)
ELISA
Flow Cytometry
Immunocytochemistry/ Immunofluorescence
Immunohistochemistry
Immunohistochemistry-Frozen
Immunohistochemistry-Paraffin
Immunoprecipitation
Simple Western
Western Blot
See Simple Western Antibody Database for Simple Western validation: Tested in Hypoxic HeLa lysate 0.5 mg/mL, separated by Size, antibody dilution of 1:200, apparent MW was 115 kDa. For IHC-P, Tris-EDTA pH 9.0 buffer is recommended for the heat induced epitope retrieval. ELISA (PMID: 17556599 and 16966370). Knockout data (PMID: 31793879). Use in Flow-intracellular reported in scientific literature (PMID:31722203).
Reviewed Applications
Read 29 reviews rated 4.3 using NB100-449 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Formulation
Preservative
Concentration
Shipping
Stability & Storage
Background: HIF-1 alpha/HIF1A
HIF-1 or hypoxia inducible factor 1 (predicted molecular weight 93kDa), is a transcription factor commonly referred to as a "master regulator of the hypoxic response" for its central role in the regulation of cellular adaptations to hypoxia. In its active form under hypoxic conditions, HIF-1 is stabilized by the formation of a heterodimer of HIF-1 alpha and ARNT/HIF-1 beta subunits. Nuclear HIF-1 engages p300/CBP for binding to hypoxic response elements (HREs). This process induces transcription and regulation of genes including EPO, VEGF, iNOS2, ANGPT1 and OCT4 (4,5).
Under normoxic conditions, the HIF-1 alpha subunit is rapidly targeted and degraded by the ubiquitin proteasome system. This process is mediated by prolyl hydroxylase domain enzymes (PHDs), which catalyze the hydroxylation of key proline residues (Pro-402 and Pro-564) within the oxygen-dependent degradation domain of HIF-1 alpha. Once hydroxylated, HIF-1 alpha binds the von Hippel-Lindau tumor suppressor protein (pVHL) for subsequent ubiquitination and proteasomal degradation (4). pVHL dependent regulation of HIF-1 alpha plays a role in normal physiology and disease states. Regulation of HIF-1 alpha by pVHL is critical for the suppressive function of FoxP3+ regulatory Tcells (6). Repression of pVHL expression in chronic lymphocytic leukemia (CLL) B cells leads to HIF-1 alpha stabilization and increased VEGF secretion (7).
References
1. Semenza, G. L., Agani, F., Feldser, D., Iyer, N., Kotch, L., Laughner, E., & Yu, A. (2000). Hypoxia, HIF-1, and the pathophysiology of common human diseases. Advances in Experimental Medicine and Biology.
2. Muz, B., de la Puente, P., Azab, F., & Azab, A. K. (2015). The role of hypoxia in cancer progression, angiogenesis, metastasis, and resistance to therapy. Hypoxia. https://doi.org/10.2147/hp.s93413
3. Huang, Y., Lin, D., & Taniguchi, C. M. (2017). Hypoxia inducible factor (HIF) in the tumor microenvironment: friend or foe? Science China Life Sciences. https://doi.org/10.1007/s11427-017-9178-y
4. Koyasu, S., Kobayashi, M., Goto, Y., Hiraoka, M., & Harada, H. (2018). Regulatory mechanisms of hypoxia-inducible factor 1 activity: Two decades of knowledge. Cancer Science. https://doi.org/10.1111/cas.13483
5. Dengler, V. L., Galbraith, M. D., & Espinosa, J. M. (2014). Transcriptional regulation by hypoxia inducible factors. Critical Reviews in Biochemistry and Molecular Biology. https://doi.org/10.3109/10409238.2013.838205
6. Lee, J. H., Elly, C., Park, Y., & Liu, Y. C. (2015). E3Ubiquitin Ligase VHL Regulates Hypoxia-Inducible Factor-1 alpha to Maintain Regulatory T Cell Stability and Suppressive Capacity. Immunity. https://doi.org/10.1016/j.immuni.2015.05.016
7. Ghosh, A. K., Shanafelt, T. D., Cimmino, A., Taccioli, C., Volinia, S., Liu, C. G.,... Kay, N. E. (2009). Aberrant regulation of pVHL levels by microRNA promotes the HIF/VEGF axis in CLL B cells. Blood. https://doi.org/10.1182/blood-2008-10-185686
Long Name
Alternate Names
Gene Symbol
UniProt
Additional HIF-1 alpha/HIF1A Products
Product Documents for HIF-1 alpha Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for HIF-1 alpha Antibody
This product is for research use only and is not approved for use in humans or in clinical diagnosis. Primary Antibodies are guaranteed for 1 year from date of receipt.
Citations for HIF-1 alpha Antibody
Customer Reviews for HIF-1 alpha Antibody (29)
Have you used HIF-1 alpha Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Application: Western BlotSample Tested: mouse liver and Hep3B human hepatocellular carcinoma cell lineSpecies: MouseVerified Customer | Posted 01/11/2024Must use nuclear extractsNuclear extraction: NE-PER NUCLEAR AND CYTOPLASMIC EXTRACTION REAGENTS
-
Application: Western BlotSample Tested: 3T3-L1 mouse embryonic fibroblast adipose-like cell lineSpecies: MouseVerified Customer | Posted 05/08/2020skimmed milk 5% primary : 1:2000 secondary: 1: 200000skimmed milk 5% primary : 1:2000 secondary: 1: 200000
-
Application: Immunohistochemistry-ParaffinSample Tested: PDX mouseSpecies: MouseVerified Customer | Posted 06/07/2019Mouse Tissue, 20x.Working Dilution 1:100, EDTA pH 9.0; detection kit Polymer-HRP.
-
Application: Western BlotSample Tested: laydig cellSpecies: GoatVerified Customer | Posted 05/15/201890-100dilution:1:500
-
Application: Western BlotSample Tested: BMDMSpecies: MouseVerified Customer | Posted 07/17/2017BMDM were seeded at 0.5x10^6 overnight. Cells were treated with 10 ng/ml LPS for 24 hrs, and a western blot was performed.
-
Application: ImmunoprecipitationSample Tested: PC-12 rat adrenal pheochromocytoma cell lineSpecies: RatVerified Customer | Posted 04/24/2017
-
Application: ImmunofluorescenceSample Tested: primary bone marrow derived macrophagesSpecies: MouseVerified Customer | Posted 01/12/2016Immunohistochemistry: HIF-1 alpha antibody
-
Application: Western BlotSample Tested: Human cancer cell whole cell lysateSpecies: HumanVerified Customer | Posted 09/07/2015
-
Application: Simple WesternSample Tested: Human mesenchymal stem cell whole cell lysateSpecies: HumanVerified Customer | Posted 06/09/20151.0 ug/uL human mesechymal stem cell lysate. Normoxia
-
Application: Western BlotSample Tested: Myeloma cells whole cell lysateSpecies: HumanVerified Customer | Posted 01/09/2015Myeloma cells whole cell lysate
-
Application: Western BlotSample Tested: See PMID 22923663Species: HumanVerified Customer | Posted 12/12/2014
-
Application: Western BlotSample Tested: Human pancreatic cancer cell lysatesSpecies: HumanVerified Customer | Posted 08/15/2014HIF1a NB100-449
-
Application: Western BlotSample Tested: Caki-1 cell lysateSpecies: HumanVerified Customer | Posted 07/18/2014HIF1alpha induction by CoCl2
-
Application: Western BlotSample Tested: large intestinal lamina propria leukocytes (LPLs).Species: MouseVerified Customer | Posted 07/05/2014Detection of HIF-1 alpha expression in a hypoxic large intestinal lamina propria leukocytes (LPLs).
-
Application: Western BlotSample Tested: MouseSpecies: MouseVerified Customer | Posted 06/30/2014
-
Application: Western BlotSample Tested: nuclear fractions from T47-D beast cancer cell lineSpecies: HumanVerified Customer | Posted 04/29/2014nuclear fraction, T47D cell line, samples in triplicate loaded in this order: normoxia, hypoxia for 16 hr, hypoxia for 24 hr
-
Application: Western BlotSample Tested:Species: OtherVerified Customer | Posted 03/14/2014HIF-1 alpha levels analyzed in cell lysates from bovine aortic endothelial cells
-
Application: Western BlotSample Tested: pig satellite cell nuclear proteinSpecies: OtherVerified Customer | Posted 08/26/2013
-
Application: Western BlotSample Tested: human cell lysatesSpecies: HumanVerified Customer | Posted 11/05/2012
-
Application: Western BlotSample Tested: mouse peritoneal and bone marrow derived macrophagesSpecies: MouseVerified Customer | Posted 08/24/2012
-
Application: Western BlotVerified Customer | Posted 08/07/2012
-
Application: Western BlotVerified Customer | Posted 05/09/2012
-
Application: Western BlotSample Tested: PC12 cell whole lysate, Sample Amount: 25ugSpecies: RatVerified Customer | Posted 01/03/2012
-
Application: Western BlotSample Tested: HUVEC whole cell lysate, Sample Amount: 20 microgSpecies: HumanVerified Customer | Posted 06/08/2011
-
Application: Western BlotSample Tested: cerebellar neuronsSpecies: MouseVerified Customer | Posted 12/22/2010
-
Application: ImmunoprecipitationSample Tested: HEK 293Species: HumanVerified Customer | Posted 12/03/2010
-
Application: Western BlotSample Tested: Colon Cancer Cell lines, Sample Amount: 50ugSpecies: OtherVerified Customer | Posted 03/11/2010
-
Application: Western BlotSample Tested: human tumor model mouse, Sample Amount: 12ugSpecies: MouseVerified Customer | Posted 12/21/2009
-
Application: Western BlotSample Tested: Mouse Neuroblastoma, Sample Amount: 25ugSpecies: MouseVerified Customer | Posted 04/08/2009
There are no reviews that match your criteria.
Protocols
View specific protocols for HIF-1 alpha Antibody (NB100-449):
IHC - Frozen 7 um mouse frozen sections were used.
Detection system: Vectors Anti-Rabbit Ig ImmPRESS Reagent Kit (cat # MP-7401)
1. Fix in ice cold acetone
2. Block for one hour at room temp. The block is provided by the vector kit; it is 2.5% horse serum.
3. Use NB 100-449 at a 1:100 dilution in PBS and incubate overnight in the fridge.
4. Perform a 15 min peroxidase block and incubated with the ImmPress anti-rabbit for 30 mins at RT.
5. Use DAB to detect staining and counterstained with Vectors Hemotoxylin. PBS washes (3X2 mins) were done in between all steps except in between the block and the primary
IHC-FFPE sections
I. Deparaffinization:
A. Treat slides with Xylene: 3 changes for 5 minutes each. Drain slides for 10 seconds between changes.
B. Treat slides with 100% Reagent Alcohol: 3 changes for 5 minutes each. Drain slides for 10 seconds between changes.
II. Quench Endogenous Peroxidase:
To Prepare 200 ml of Quenching Solution:
Add 3 ml of 30% Hydrogen Peroxide to 200 ml of Methanol.
**Use within 4 hours of preparation
A. Place slides in peroxidase quenching solution: 15-30 minutes.
B. Place slides in distilled water: 2 changes for 2 minutes each.
III. Retrieve Epitopes:
A. Preheat Citrate Buffer. Place 200 ml of Citrate Buffer Working Solution into container, cover and place into steamer. Heat to 90-96C.
B. Place rack of slides into hot Citrate Buffer for 20 minutes. Cover.
C. Carefully remove container with slides from steamer and cool on bench, uncovered, for 20 minutes.
D. Slowly add distilled water to further cool for 5 minutes.
E. Rinse slides with distilled water, 2 changes for 2 minutes each
IV. Immunostaining Procedure:
A. Remove each slide from rack and circle tissue section with a hydrophobic barrier pen (e.g. Liquid Blocker-Super Pap Pen).
B. Flood slide with Wash Solution.
Do not allow tissue sections to dry for the rest of the procedure.
C. Drain wash solution and apply 4 drops of Blocking Reagent to each slide and incubate for 15 minutes.
D. Drain Blocking Reagent (do not wash off the Blocking Reagent), apply 200 ul of Primary Antibody solution to each slide, and incubate for 1 hour.
E. Wash slides with Wash Solution: 3 changes for 5 minutes each.
F. Drain wash solution, apply 4 drops of Secondary antibody to each slide and incubate for 1 hour.
G. Wash slides with Wash Solution: 3 changes for 5 minutes each.
H. Drain wash solution, apply 4 drops of DAB Substrate to each slide and develop for 5-10 minutes.
Check development with microscope.
I. Wash slides with Wash Solution: 3 changes for 5 minutes each.
J. Drain wash solution, apply 4 drops of Hematoxylin to each slide and stain for 1-3 minutes. Increase time if darker counterstaining is desired.
K. Wash slides with Wash Solution: 2-3 changes for 2 minutes each.
L. Drain wash solution and apply 4 drops of Bluing Solution to each slide for 1-2 minutes.
M. Rinse slides in distilled water.
N. Soak slides in 70% reagent alcohol: 3 minutes with intermittent agitation.
O. Soak slides in 95% reagent alcohol: 2 changes for 3 minutes each with intermittent agitation.
P. Soak slides in 100% reagent alcohol: 3 changes for 3 minutes each with intermittent agitation. Drain slides for 10 seconds between each change.
Q. Soak slides in Xylene: 3 changes for 3 minutes each with intermittent agitation. Drain slides for 10 seconds between each change.
R. Apply 2-3 drops of non-aqueous mounting media to each slide and mount coverslip.
S. Lay slides on a flat surface to dry prior to viewing under microscope.
NOTES:
-Use treated slides (e.g. HistoBond) to assure adherence of FFPE sections to slide.
-Prior to Deparaffinization, heat slides overnight in a 60C oven.
-All steps in which Xylene is used should be performed in a fume hood.
-For Epitope Retrieval, a microwave or pressure cooker may be substituted for the steamer method. Adjust times as necessary depending on conditions.
-For the initial IHC run with a new primary antibody, test tissues with and without Epitope Retrieval. In some instances, Epitope Retrieval may not be necessary.
-200 ul is the recommended maximum volume to apply to a slide for full coverage. Using more than 200 ul may allow solutions to wick off the slide and create drying artifacts, for small tissue sections less than 200 ul may be used.
-5 minutes of development with DAB Substrate should be sufficient. Do not develop for more than 10 minutes. If 5 minutes of development causes background staining, further dilution of the primary antibody may be necessary.
-Hematoxylin should produce a light nuclear counterstain so as not to obscure the DAB staining. Counterstain for 1-1.5 minutes for nuclear antigens. Counterstain for 2-3 minutes for cytoplasmic and membranous antigens. If darker counterstaining is desired increase time (up to 10 minutes)
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ChIP Protocol Video
- Chromatin Immunoprecipitation (ChIP) Protocol
- Chromatin Immunoprecipitation Protocol
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ELISA Sample Preparation & Collection Guide
- ELISA Troubleshooting Guide
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- How to Run an R&D Systems DuoSet ELISA
- How to Run an R&D Systems Quantikine ELISA
- How to Run an R&D Systems Quantikine™ QuicKit™ ELISA
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Immunoprecipitation Protocol
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- Quantikine HS ELISA Kit Assay Principle, Alkaline Phosphatase
- Quantikine HS ELISA Kit Principle, Streptavidin-HRP Polymer
- R&D Systems Quality Control Western Blot Protocol
- Sandwich ELISA (Colorimetric) – Biotin/Streptavidin Detection Protocol
- Sandwich ELISA (Colorimetric) – Direct Detection Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: ELISA
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
FAQs for HIF-1 alpha Antibody
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Could you tell us if these antibodies need to use cell lysate treated with special extraction for WB? Do you have the recommended preparation method of cell lysate?
A:
Usually HIF1 alpha and HIF2 alpha are hard to detect endogenously and require some inducement. The most common is hypoxic treatment of the cells. This can be also be chemically induced by CoCl2 treatment as well. Our recommended protocols and lysate preparations can be found in our white paper here
-
Q: Do you have any specific protocol or relative literature for HIF1a FACS staining?
A: This particular antibody has not been tested in house, so unfortunately we do not have a specific protocol for this. We have a collaborator who tested this antibody in FACS and that image is on our datasheet. With the image is a brief of their protocol: 'Flow Cytometry: HIF-1 alpha Antibody [NB100-449] - Hela cells were treated for 15 hrs with 200uM CoCl2, fixed in PFA, and permeabilized in 90% MeOH. 1 X 10^6 cells were stained with 0.125ug anti- HIF-alpha and secondary FITC-conjugated goat anti-rabbit (in a 150ul reaction). Black- treated, anti-KLH control IgG; Red- untreated, anti-HIF1-alpha; Blue- treated, anti-HIF1-alpha.'
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I detect the 60 kDa band for HIF-1 alpha antibody (NB100-449), but no band at the right molecular weight. Do you know what this non-specific band is?
A: Most likely the 60kDa band is a degradation product of HIF1A in your samples. Since HIF-1 alpha degrades so rapidly in the presence of oxygen, our lab often sees bands at lower molecular weights. We have also experienced some non-specific banding around that molecular weight too in some tests.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Could you tell us if these antibodies need to use cell lysate treated with special extraction for WB? Do you have the recommended preparation method of cell lysate?
A:
Usually HIF1 alpha and HIF2 alpha are hard to detect endogenously and require some inducement. The most common is hypoxic treatment of the cells. This can be also be chemically induced by CoCl2 treatment as well. Our recommended protocols and lysate preparations can be found in our white paper here
-
Q: Do you have any specific protocol or relative literature for HIF1a FACS staining?
A: This particular antibody has not been tested in house, so unfortunately we do not have a specific protocol for this. We have a collaborator who tested this antibody in FACS and that image is on our datasheet. With the image is a brief of their protocol: 'Flow Cytometry: HIF-1 alpha Antibody [NB100-449] - Hela cells were treated for 15 hrs with 200uM CoCl2, fixed in PFA, and permeabilized in 90% MeOH. 1 X 10^6 cells were stained with 0.125ug anti- HIF-alpha and secondary FITC-conjugated goat anti-rabbit (in a 150ul reaction). Black- treated, anti-KLH control IgG; Red- untreated, anti-HIF1-alpha; Blue- treated, anti-HIF1-alpha.'
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I detect the 60 kDa band for HIF-1 alpha antibody (NB100-449), but no band at the right molecular weight. Do you know what this non-specific band is?
A: Most likely the 60kDa band is a degradation product of HIF1A in your samples. Since HIF-1 alpha degrades so rapidly in the presence of oxygen, our lab often sees bands at lower molecular weights. We have also experienced some non-specific banding around that molecular weight too in some tests.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Could you tell us if these antibodies need to use cell lysate treated with special extraction for WB? Do you have the recommended preparation method of cell lysate?
A:
Usually HIF1 alpha and HIF2 alpha are hard to detect endogenously and require some inducement. The most common is hypoxic treatment of the cells. This can be also be chemically induced by CoCl2 treatment as well. Our recommended protocols and lysate preparations can be found in our white paper here
-
Q: Do you have any specific protocol or relative literature for HIF1a FACS staining?
A: This particular antibody has not been tested in house, so unfortunately we do not have a specific protocol for this. We have a collaborator who tested this antibody in FACS and that image is on our datasheet. With the image is a brief of their protocol: 'Flow Cytometry: HIF-1 alpha Antibody [NB100-449] - Hela cells were treated for 15 hrs with 200uM CoCl2, fixed in PFA, and permeabilized in 90% MeOH. 1 X 10^6 cells were stained with 0.125ug anti- HIF-alpha and secondary FITC-conjugated goat anti-rabbit (in a 150ul reaction). Black- treated, anti-KLH control IgG; Red- untreated, anti-HIF1-alpha; Blue- treated, anti-HIF1-alpha.'
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I detect the 60 kDa band for HIF-1 alpha antibody (NB100-449), but no band at the right molecular weight. Do you know what this non-specific band is?
A: Most likely the 60kDa band is a degradation product of HIF1A in your samples. Since HIF-1 alpha degrades so rapidly in the presence of oxygen, our lab often sees bands at lower molecular weights. We have also experienced some non-specific banding around that molecular weight too in some tests.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Could you tell us if these antibodies need to use cell lysate treated with special extraction for WB? Do you have the recommended preparation method of cell lysate?
A:
Usually HIF1 alpha and HIF2 alpha are hard to detect endogenously and require some inducement. The most common is hypoxic treatment of the cells. This can be also be chemically induced by CoCl2 treatment as well. Our recommended protocols and lysate preparations can be found in our white paper here
-
Q: Do you have any specific protocol or relative literature for HIF1a FACS staining?
A: This particular antibody has not been tested in house, so unfortunately we do not have a specific protocol for this. We have a collaborator who tested this antibody in FACS and that image is on our datasheet. With the image is a brief of their protocol: 'Flow Cytometry: HIF-1 alpha Antibody [NB100-449] - Hela cells were treated for 15 hrs with 200uM CoCl2, fixed in PFA, and permeabilized in 90% MeOH. 1 X 10^6 cells were stained with 0.125ug anti- HIF-alpha and secondary FITC-conjugated goat anti-rabbit (in a 150ul reaction). Black- treated, anti-KLH control IgG; Red- untreated, anti-HIF1-alpha; Blue- treated, anti-HIF1-alpha.'
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I detect the 60 kDa band for HIF-1 alpha antibody (NB100-449), but no band at the right molecular weight. Do you know what this non-specific band is?
A: Most likely the 60kDa band is a degradation product of HIF1A in your samples. Since HIF-1 alpha degrades so rapidly in the presence of oxygen, our lab often sees bands at lower molecular weights. We have also experienced some non-specific banding around that molecular weight too in some tests.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Could you tell us if these antibodies need to use cell lysate treated with special extraction for WB? Do you have the recommended preparation method of cell lysate?
A:
Usually HIF1 alpha and HIF2 alpha are hard to detect endogenously and require some inducement. The most common is hypoxic treatment of the cells. This can be also be chemically induced by CoCl2 treatment as well. Our recommended protocols and lysate preparations can be found in our white paper here
-
Q: Do you have any specific protocol or relative literature for HIF1a FACS staining?
A: This particular antibody has not been tested in house, so unfortunately we do not have a specific protocol for this. We have a collaborator who tested this antibody in FACS and that image is on our datasheet. With the image is a brief of their protocol: 'Flow Cytometry: HIF-1 alpha Antibody [NB100-449] - Hela cells were treated for 15 hrs with 200uM CoCl2, fixed in PFA, and permeabilized in 90% MeOH. 1 X 10^6 cells were stained with 0.125ug anti- HIF-alpha and secondary FITC-conjugated goat anti-rabbit (in a 150ul reaction). Black- treated, anti-KLH control IgG; Red- untreated, anti-HIF1-alpha; Blue- treated, anti-HIF1-alpha.'
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I detect the 60 kDa band for HIF-1 alpha antibody (NB100-449), but no band at the right molecular weight. Do you know what this non-specific band is?
A: Most likely the 60kDa band is a degradation product of HIF1A in your samples. Since HIF-1 alpha degrades so rapidly in the presence of oxygen, our lab often sees bands at lower molecular weights. We have also experienced some non-specific banding around that molecular weight too in some tests.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Could you tell us if these antibodies need to use cell lysate treated with special extraction for WB? Do you have the recommended preparation method of cell lysate?
A:
Usually HIF1 alpha and HIF2 alpha are hard to detect endogenously and require some inducement. The most common is hypoxic treatment of the cells. This can be also be chemically induced by CoCl2 treatment as well. Our recommended protocols and lysate preparations can be found in our white paper here
-
Q: Do you have any specific protocol or relative literature for HIF1a FACS staining?
A: This particular antibody has not been tested in house, so unfortunately we do not have a specific protocol for this. We have a collaborator who tested this antibody in FACS and that image is on our datasheet. With the image is a brief of their protocol: 'Flow Cytometry: HIF-1 alpha Antibody [NB100-449] - Hela cells were treated for 15 hrs with 200uM CoCl2, fixed in PFA, and permeabilized in 90% MeOH. 1 X 10^6 cells were stained with 0.125ug anti- HIF-alpha and secondary FITC-conjugated goat anti-rabbit (in a 150ul reaction). Black- treated, anti-KLH control IgG; Red- untreated, anti-HIF1-alpha; Blue- treated, anti-HIF1-alpha.'
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I detect the 60 kDa band for HIF-1 alpha antibody (NB100-449), but no band at the right molecular weight. Do you know what this non-specific band is?
A: Most likely the 60kDa band is a degradation product of HIF1A in your samples. Since HIF-1 alpha degrades so rapidly in the presence of oxygen, our lab often sees bands at lower molecular weights. We have also experienced some non-specific banding around that molecular weight too in some tests.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Could you tell us if these antibodies need to use cell lysate treated with special extraction for WB? Do you have the recommended preparation method of cell lysate?
A:
Usually HIF1 alpha and HIF2 alpha are hard to detect endogenously and require some inducement. The most common is hypoxic treatment of the cells. This can be also be chemically induced by CoCl2 treatment as well. Our recommended protocols and lysate preparations can be found in our white paper here
-
Q: Do you have any specific protocol or relative literature for HIF1a FACS staining?
A: This particular antibody has not been tested in house, so unfortunately we do not have a specific protocol for this. We have a collaborator who tested this antibody in FACS and that image is on our datasheet. With the image is a brief of their protocol: 'Flow Cytometry: HIF-1 alpha Antibody [NB100-449] - Hela cells were treated for 15 hrs with 200uM CoCl2, fixed in PFA, and permeabilized in 90% MeOH. 1 X 10^6 cells were stained with 0.125ug anti- HIF-alpha and secondary FITC-conjugated goat anti-rabbit (in a 150ul reaction). Black- treated, anti-KLH control IgG; Red- untreated, anti-HIF1-alpha; Blue- treated, anti-HIF1-alpha.'
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I detect the 60 kDa band for HIF-1 alpha antibody (NB100-449), but no band at the right molecular weight. Do you know what this non-specific band is?
A: Most likely the 60kDa band is a degradation product of HIF1A in your samples. Since HIF-1 alpha degrades so rapidly in the presence of oxygen, our lab often sees bands at lower molecular weights. We have also experienced some non-specific banding around that molecular weight too in some tests.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Could you tell us if these antibodies need to use cell lysate treated with special extraction for WB? Do you have the recommended preparation method of cell lysate?
A:
Usually HIF1 alpha and HIF2 alpha are hard to detect endogenously and require some inducement. The most common is hypoxic treatment of the cells. This can be also be chemically induced by CoCl2 treatment as well. Our recommended protocols and lysate preparations can be found in our white paper here
-
Q: Do you have any specific protocol or relative literature for HIF1a FACS staining?
A: This particular antibody has not been tested in house, so unfortunately we do not have a specific protocol for this. We have a collaborator who tested this antibody in FACS and that image is on our datasheet. With the image is a brief of their protocol: 'Flow Cytometry: HIF-1 alpha Antibody [NB100-449] - Hela cells were treated for 15 hrs with 200uM CoCl2, fixed in PFA, and permeabilized in 90% MeOH. 1 X 10^6 cells were stained with 0.125ug anti- HIF-alpha and secondary FITC-conjugated goat anti-rabbit (in a 150ul reaction). Black- treated, anti-KLH control IgG; Red- untreated, anti-HIF1-alpha; Blue- treated, anti-HIF1-alpha.'
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I detect the 60 kDa band for HIF-1 alpha antibody (NB100-449), but no band at the right molecular weight. Do you know what this non-specific band is?
A: Most likely the 60kDa band is a degradation product of HIF1A in your samples. Since HIF-1 alpha degrades so rapidly in the presence of oxygen, our lab often sees bands at lower molecular weights. We have also experienced some non-specific banding around that molecular weight too in some tests.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Could you tell us if these antibodies need to use cell lysate treated with special extraction for WB? Do you have the recommended preparation method of cell lysate?
A:
Usually HIF1 alpha and HIF2 alpha are hard to detect endogenously and require some inducement. The most common is hypoxic treatment of the cells. This can be also be chemically induced by CoCl2 treatment as well. Our recommended protocols and lysate preparations can be found in our white paper here
-
Q: Do you have any specific protocol or relative literature for HIF1a FACS staining?
A: This particular antibody has not been tested in house, so unfortunately we do not have a specific protocol for this. We have a collaborator who tested this antibody in FACS and that image is on our datasheet. With the image is a brief of their protocol: 'Flow Cytometry: HIF-1 alpha Antibody [NB100-449] - Hela cells were treated for 15 hrs with 200uM CoCl2, fixed in PFA, and permeabilized in 90% MeOH. 1 X 10^6 cells were stained with 0.125ug anti- HIF-alpha and secondary FITC-conjugated goat anti-rabbit (in a 150ul reaction). Black- treated, anti-KLH control IgG; Red- untreated, anti-HIF1-alpha; Blue- treated, anti-HIF1-alpha.'
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I detect the 60 kDa band for HIF-1 alpha antibody (NB100-449), but no band at the right molecular weight. Do you know what this non-specific band is?
A: Most likely the 60kDa band is a degradation product of HIF1A in your samples. Since HIF-1 alpha degrades so rapidly in the presence of oxygen, our lab often sees bands at lower molecular weights. We have also experienced some non-specific banding around that molecular weight too in some tests.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Could you tell us if these antibodies need to use cell lysate treated with special extraction for WB? Do you have the recommended preparation method of cell lysate?
A:
Usually HIF1 alpha and HIF2 alpha are hard to detect endogenously and require some inducement. The most common is hypoxic treatment of the cells. This can be also be chemically induced by CoCl2 treatment as well. Our recommended protocols and lysate preparations can be found in our white paper here
-
Q: Do you have any specific protocol or relative literature for HIF1a FACS staining?
A: This particular antibody has not been tested in house, so unfortunately we do not have a specific protocol for this. We have a collaborator who tested this antibody in FACS and that image is on our datasheet. With the image is a brief of their protocol: 'Flow Cytometry: HIF-1 alpha Antibody [NB100-449] - Hela cells were treated for 15 hrs with 200uM CoCl2, fixed in PFA, and permeabilized in 90% MeOH. 1 X 10^6 cells were stained with 0.125ug anti- HIF-alpha and secondary FITC-conjugated goat anti-rabbit (in a 150ul reaction). Black- treated, anti-KLH control IgG; Red- untreated, anti-HIF1-alpha; Blue- treated, anti-HIF1-alpha.'
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I detect the 60 kDa band for HIF-1 alpha antibody (NB100-449), but no band at the right molecular weight. Do you know what this non-specific band is?
A: Most likely the 60kDa band is a degradation product of HIF1A in your samples. Since HIF-1 alpha degrades so rapidly in the presence of oxygen, our lab often sees bands at lower molecular weights. We have also experienced some non-specific banding around that molecular weight too in some tests.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Could you tell us if these antibodies need to use cell lysate treated with special extraction for WB? Do you have the recommended preparation method of cell lysate?
A:
Usually HIF1 alpha and HIF2 alpha are hard to detect endogenously and require some inducement. The most common is hypoxic treatment of the cells. This can be also be chemically induced by CoCl2 treatment as well. Our recommended protocols and lysate preparations can be found in our white paper here
-
Q: Do you have any specific protocol or relative literature for HIF1a FACS staining?
A: This particular antibody has not been tested in house, so unfortunately we do not have a specific protocol for this. We have a collaborator who tested this antibody in FACS and that image is on our datasheet. With the image is a brief of their protocol: 'Flow Cytometry: HIF-1 alpha Antibody [NB100-449] - Hela cells were treated for 15 hrs with 200uM CoCl2, fixed in PFA, and permeabilized in 90% MeOH. 1 X 10^6 cells were stained with 0.125ug anti- HIF-alpha and secondary FITC-conjugated goat anti-rabbit (in a 150ul reaction). Black- treated, anti-KLH control IgG; Red- untreated, anti-HIF1-alpha; Blue- treated, anti-HIF1-alpha.'
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I detect the 60 kDa band for HIF-1 alpha antibody (NB100-449), but no band at the right molecular weight. Do you know what this non-specific band is?
A: Most likely the 60kDa band is a degradation product of HIF1A in your samples. Since HIF-1 alpha degrades so rapidly in the presence of oxygen, our lab often sees bands at lower molecular weights. We have also experienced some non-specific banding around that molecular weight too in some tests.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Could you tell us if these antibodies need to use cell lysate treated with special extraction for WB? Do you have the recommended preparation method of cell lysate?
A:
Usually HIF1 alpha and HIF2 alpha are hard to detect endogenously and require some inducement. The most common is hypoxic treatment of the cells. This can be also be chemically induced by CoCl2 treatment as well. Our recommended protocols and lysate preparations can be found in our white paper here
-
Q: Do you have any specific protocol or relative literature for HIF1a FACS staining?
A: This particular antibody has not been tested in house, so unfortunately we do not have a specific protocol for this. We have a collaborator who tested this antibody in FACS and that image is on our datasheet. With the image is a brief of their protocol: 'Flow Cytometry: HIF-1 alpha Antibody [NB100-449] - Hela cells were treated for 15 hrs with 200uM CoCl2, fixed in PFA, and permeabilized in 90% MeOH. 1 X 10^6 cells were stained with 0.125ug anti- HIF-alpha and secondary FITC-conjugated goat anti-rabbit (in a 150ul reaction). Black- treated, anti-KLH control IgG; Red- untreated, anti-HIF1-alpha; Blue- treated, anti-HIF1-alpha.'
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I detect the 60 kDa band for HIF-1 alpha antibody (NB100-449), but no band at the right molecular weight. Do you know what this non-specific band is?
A: Most likely the 60kDa band is a degradation product of HIF1A in your samples. Since HIF-1 alpha degrades so rapidly in the presence of oxygen, our lab often sees bands at lower molecular weights. We have also experienced some non-specific banding around that molecular weight too in some tests.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Could you tell us if these antibodies need to use cell lysate treated with special extraction for WB? Do you have the recommended preparation method of cell lysate?
A:
Usually HIF1 alpha and HIF2 alpha are hard to detect endogenously and require some inducement. The most common is hypoxic treatment of the cells. This can be also be chemically induced by CoCl2 treatment as well. Our recommended protocols and lysate preparations can be found in our white paper here
-
Q: Do you have any specific protocol or relative literature for HIF1a FACS staining?
A: This particular antibody has not been tested in house, so unfortunately we do not have a specific protocol for this. We have a collaborator who tested this antibody in FACS and that image is on our datasheet. With the image is a brief of their protocol: 'Flow Cytometry: HIF-1 alpha Antibody [NB100-449] - Hela cells were treated for 15 hrs with 200uM CoCl2, fixed in PFA, and permeabilized in 90% MeOH. 1 X 10^6 cells were stained with 0.125ug anti- HIF-alpha and secondary FITC-conjugated goat anti-rabbit (in a 150ul reaction). Black- treated, anti-KLH control IgG; Red- untreated, anti-HIF1-alpha; Blue- treated, anti-HIF1-alpha.'
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I detect the 60 kDa band for HIF-1 alpha antibody (NB100-449), but no band at the right molecular weight. Do you know what this non-specific band is?
A: Most likely the 60kDa band is a degradation product of HIF1A in your samples. Since HIF-1 alpha degrades so rapidly in the presence of oxygen, our lab often sees bands at lower molecular weights. We have also experienced some non-specific banding around that molecular weight too in some tests.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Could you tell us if these antibodies need to use cell lysate treated with special extraction for WB? Do you have the recommended preparation method of cell lysate?
A:
Usually HIF1 alpha and HIF2 alpha are hard to detect endogenously and require some inducement. The most common is hypoxic treatment of the cells. This can be also be chemically induced by CoCl2 treatment as well. Our recommended protocols and lysate preparations can be found in our white paper here
-
Q: Do you have any specific protocol or relative literature for HIF1a FACS staining?
A: This particular antibody has not been tested in house, so unfortunately we do not have a specific protocol for this. We have a collaborator who tested this antibody in FACS and that image is on our datasheet. With the image is a brief of their protocol: 'Flow Cytometry: HIF-1 alpha Antibody [NB100-449] - Hela cells were treated for 15 hrs with 200uM CoCl2, fixed in PFA, and permeabilized in 90% MeOH. 1 X 10^6 cells were stained with 0.125ug anti- HIF-alpha and secondary FITC-conjugated goat anti-rabbit (in a 150ul reaction). Black- treated, anti-KLH control IgG; Red- untreated, anti-HIF1-alpha; Blue- treated, anti-HIF1-alpha.'
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I detect the 60 kDa band for HIF-1 alpha antibody (NB100-449), but no band at the right molecular weight. Do you know what this non-specific band is?
A: Most likely the 60kDa band is a degradation product of HIF1A in your samples. Since HIF-1 alpha degrades so rapidly in the presence of oxygen, our lab often sees bands at lower molecular weights. We have also experienced some non-specific banding around that molecular weight too in some tests.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Could you tell us if these antibodies need to use cell lysate treated with special extraction for WB? Do you have the recommended preparation method of cell lysate?
A:
Usually HIF1 alpha and HIF2 alpha are hard to detect endogenously and require some inducement. The most common is hypoxic treatment of the cells. This can be also be chemically induced by CoCl2 treatment as well. Our recommended protocols and lysate preparations can be found in our white paper here
-
Q: Do you have any specific protocol or relative literature for HIF1a FACS staining?
A: This particular antibody has not been tested in house, so unfortunately we do not have a specific protocol for this. We have a collaborator who tested this antibody in FACS and that image is on our datasheet. With the image is a brief of their protocol: 'Flow Cytometry: HIF-1 alpha Antibody [NB100-449] - Hela cells were treated for 15 hrs with 200uM CoCl2, fixed in PFA, and permeabilized in 90% MeOH. 1 X 10^6 cells were stained with 0.125ug anti- HIF-alpha and secondary FITC-conjugated goat anti-rabbit (in a 150ul reaction). Black- treated, anti-KLH control IgG; Red- untreated, anti-HIF1-alpha; Blue- treated, anti-HIF1-alpha.'
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I detect the 60 kDa band for HIF-1 alpha antibody (NB100-449), but no band at the right molecular weight. Do you know what this non-specific band is?
A: Most likely the 60kDa band is a degradation product of HIF1A in your samples. Since HIF-1 alpha degrades so rapidly in the presence of oxygen, our lab often sees bands at lower molecular weights. We have also experienced some non-specific banding around that molecular weight too in some tests.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Could you tell us if these antibodies need to use cell lysate treated with special extraction for WB? Do you have the recommended preparation method of cell lysate?
A:
Usually HIF1 alpha and HIF2 alpha are hard to detect endogenously and require some inducement. The most common is hypoxic treatment of the cells. This can be also be chemically induced by CoCl2 treatment as well. Our recommended protocols and lysate preparations can be found in our white paper here
-
Q: Do you have any specific protocol or relative literature for HIF1a FACS staining?
A: This particular antibody has not been tested in house, so unfortunately we do not have a specific protocol for this. We have a collaborator who tested this antibody in FACS and that image is on our datasheet. With the image is a brief of their protocol: 'Flow Cytometry: HIF-1 alpha Antibody [NB100-449] - Hela cells were treated for 15 hrs with 200uM CoCl2, fixed in PFA, and permeabilized in 90% MeOH. 1 X 10^6 cells were stained with 0.125ug anti- HIF-alpha and secondary FITC-conjugated goat anti-rabbit (in a 150ul reaction). Black- treated, anti-KLH control IgG; Red- untreated, anti-HIF1-alpha; Blue- treated, anti-HIF1-alpha.'
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I detect the 60 kDa band for HIF-1 alpha antibody (NB100-449), but no band at the right molecular weight. Do you know what this non-specific band is?
A: Most likely the 60kDa band is a degradation product of HIF1A in your samples. Since HIF-1 alpha degrades so rapidly in the presence of oxygen, our lab often sees bands at lower molecular weights. We have also experienced some non-specific banding around that molecular weight too in some tests.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Could you tell us if these antibodies need to use cell lysate treated with special extraction for WB? Do you have the recommended preparation method of cell lysate?
A:
Usually HIF1 alpha and HIF2 alpha are hard to detect endogenously and require some inducement. The most common is hypoxic treatment of the cells. This can be also be chemically induced by CoCl2 treatment as well. Our recommended protocols and lysate preparations can be found in our white paper here
-
Q: Do you have any specific protocol or relative literature for HIF1a FACS staining?
A: This particular antibody has not been tested in house, so unfortunately we do not have a specific protocol for this. We have a collaborator who tested this antibody in FACS and that image is on our datasheet. With the image is a brief of their protocol: 'Flow Cytometry: HIF-1 alpha Antibody [NB100-449] - Hela cells were treated for 15 hrs with 200uM CoCl2, fixed in PFA, and permeabilized in 90% MeOH. 1 X 10^6 cells were stained with 0.125ug anti- HIF-alpha and secondary FITC-conjugated goat anti-rabbit (in a 150ul reaction). Black- treated, anti-KLH control IgG; Red- untreated, anti-HIF1-alpha; Blue- treated, anti-HIF1-alpha.'
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I detect the 60 kDa band for HIF-1 alpha antibody (NB100-449), but no band at the right molecular weight. Do you know what this non-specific band is?
A: Most likely the 60kDa band is a degradation product of HIF1A in your samples. Since HIF-1 alpha degrades so rapidly in the presence of oxygen, our lab often sees bands at lower molecular weights. We have also experienced some non-specific banding around that molecular weight too in some tests.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Could you tell us if these antibodies need to use cell lysate treated with special extraction for WB? Do you have the recommended preparation method of cell lysate?
A:
Usually HIF1 alpha and HIF2 alpha are hard to detect endogenously and require some inducement. The most common is hypoxic treatment of the cells. This can be also be chemically induced by CoCl2 treatment as well. Our recommended protocols and lysate preparations can be found in our white paper here
-
Q: Do you have any specific protocol or relative literature for HIF1a FACS staining?
A: This particular antibody has not been tested in house, so unfortunately we do not have a specific protocol for this. We have a collaborator who tested this antibody in FACS and that image is on our datasheet. With the image is a brief of their protocol: 'Flow Cytometry: HIF-1 alpha Antibody [NB100-449] - Hela cells were treated for 15 hrs with 200uM CoCl2, fixed in PFA, and permeabilized in 90% MeOH. 1 X 10^6 cells were stained with 0.125ug anti- HIF-alpha and secondary FITC-conjugated goat anti-rabbit (in a 150ul reaction). Black- treated, anti-KLH control IgG; Red- untreated, anti-HIF1-alpha; Blue- treated, anti-HIF1-alpha.'
-
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I detect the 60 kDa band for HIF-1 alpha antibody (NB100-449), but no band at the right molecular weight. Do you know what this non-specific band is?
A: Most likely the 60kDa band is a degradation product of HIF1A in your samples. Since HIF-1 alpha degrades so rapidly in the presence of oxygen, our lab often sees bands at lower molecular weights. We have also experienced some non-specific banding around that molecular weight too in some tests.
-
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
Associated Pathways
 HIF Repressor Pathways
HIF Repressor Pathways
 Notch Signaling Pathways
Notch Signaling Pathways


![Western Blot: HIF-1 alpha Antibody [NB100-449] Western Blot: HIF-1 alpha Antibody [NB100-449]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-Western-Blot-NB100-449-img0015.jpg)
![Simple Western: HIF-1 alpha Antibody [NB100-449] Simple Western: HIF-1 alpha Antibody [NB100-449]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-Simple-Western-NB100-449-img0032.jpg)
![Immunohistochemistry: HIF-1 alpha Antibody [NB100-449] Immunohistochemistry: HIF-1 alpha Antibody [NB100-449]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-Immunohistochemistry-NB100-449-img0034.jpg)
![Immunohistochemistry: HIF-1 alpha Antibody [NB100-449] Immunohistochemistry: HIF-1 alpha Antibody [NB100-449]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-Immunohistochemistry-NB100-449-img0047.jpg)
![Western Blot: HIF-1 alpha Antibody [NB100-449] Western Blot: HIF-1 alpha Antibody [NB100-449]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-Western-Blot-NB100-449-img0038.jpg)
![Western Blot: HIF-1 alpha Antibody [NB100-449] Western Blot: HIF-1 alpha Antibody [NB100-449]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-Western-Blot-NB100-449-img0053.jpg)
![Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody [NB100-449] Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody [NB100-449]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-Immunocytochemistry-Immunofluorescence-NB100-449-img0039.jpg)
![Immunohistochemistry-Paraffin: HIF-1 alpha Antibody [NB100-449] Immunohistochemistry-Paraffin: HIF-1 alpha Antibody [NB100-449]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-Immunohistochemistry-Paraffin-NB100-449-img0052.jpg)
![Flow Cytometry: HIF-1 alpha Antibody [NB100-449] Flow Cytometry: HIF-1 alpha Antibody [NB100-449]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-Flow-Cytometry-NB100-449-img0035.jpg)
![Western Blot: HIF-1 alpha Antibody [NB100-449] Western Blot: HIF-1 alpha Antibody [NB100-449]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-Western-Blot-NB100-449-img0033.jpg)
![Western Blot: HIF-1 alpha Antibody [NB100-449] Western Blot: HIF-1 alpha Antibody [NB100-449]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-Western-Blot-NB100-449-img0036.jpg)
![Western Blot: HIF-1 alpha Antibody [NB100-449] Western Blot: HIF-1 alpha Antibody [NB100-449]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-Western-Blot-NB100-449-img0037.jpg)
![Western Blot: HIF-1 alpha Antibody [NB100-449] Western Blot: HIF-1 alpha Antibody [NB100-449]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-Western-Blot-NB100-449-img0042.jpg)
![Western Blot: HIF-1 alpha Antibody [NB100-449] Western Blot: HIF-1 alpha Antibody [NB100-449]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-Western-Blot-NB100-449-img0043.jpg)
![Western Blot: HIF-1 alpha Antibody [NB100-449] Western Blot: HIF-1 alpha Antibody [NB100-449]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-Western-Blot-NB100-449-img0044.jpg)
![Western Blot: HIF-1 alpha Antibody [NB100-449] Western Blot: HIF-1 alpha Antibody [NB100-449]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-Western-Blot-NB100-449-img0045.jpg)
![Western Blot: HIF-1 alpha Antibody [NB100-449] Western Blot: HIF-1 alpha Antibody [NB100-449]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-Western-Blot-NB100-449-img0046.jpg)
![Western Blot: HIF-1 alpha Antibody [NB100-449] Western Blot: HIF-1 alpha Antibody [NB100-449]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-Western-Blot-NB100-449-img0051.jpg)
![Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody [NB100-449] Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody [NB100-449]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-Immunocytochemistry-Immunofluorescence-NB100-449-img0006.jpg)
![Immunoprecipitation: HIF-1 alpha Antibody [NB100-449] Immunoprecipitation: HIF-1 alpha Antibody [NB100-449]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-Immunoprecipitation-NB100-449-img0005.jpg)
![Immunoprecipitation: HIF-1 alpha Antibody [NB100-449] Immunoprecipitation: HIF-1 alpha Antibody [NB100-449]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-Immunoprecipitation-NB100-449-img0048.jpg)
![Western Blot: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-271220231253349.jpg)
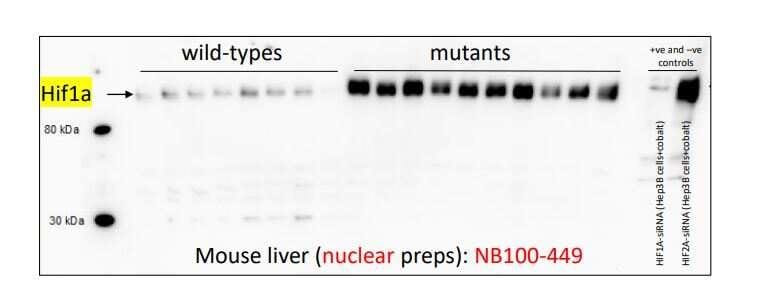
![Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-210202423452449.jpg)
![Immunohistochemistry: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-310202415284545.jpg)
![Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-310202415304212.jpg)
![Immunohistochemistry-Paraffin: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-310202415363719.jpg)
![Immunohistochemistry-Paraffin: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-31020241534344.jpg)
![Immunohistochemistry: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-310202415384135.jpg)
![Immunohistochemistry: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-310202415293325.jpg)
![Western Blot: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-310202415394514.jpg)
![Immunohistochemistry: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-310202415291950.jpg)
![Western Blot: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-310202415304256.jpg)
![Immunohistochemistry-Frozen: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-31020241553383.jpg)
![Western Blot: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-310202415532021.jpg)
![Western Blot: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-310202415525284.jpg)
![Western Blot: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-31020241612685.jpg)
![Western Blot: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-31020241552391.jpg)
![Western Blot: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-31020241554543.jpg)
![Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-31020241554544.jpg)
![Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-31020241612613.jpg)
![Western Blot: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-31020241612635.jpg)
![Western Blot: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-3102024155453.jpg)
![Western Blot: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-31020241554521.jpg)
![Western Blot: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-31020241552522.jpg)
![Western Blot: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-31020241682187.jpg)
![Western Blot: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-31020241683944.jpg)
![Western Blot: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-31020241612654.jpg)
![Western Blot: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-310202415533837.jpg)
![Immunohistochemistry: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-31020241612628.jpg)
![Western Blot: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-310202415533827.jpg)
![Western Blot: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-31020241682146.jpg)
![Western Blot: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-31020241682155.jpg)
![Western Blot: HIF-1 alpha Antibody [NB100-449] - HIF-1 alpha Antibody](https://resources.rndsystems.com/images/products/nb100-449_rabbit-polyclonal-hif-1-alpha-antibody-310202415541992.jpg)

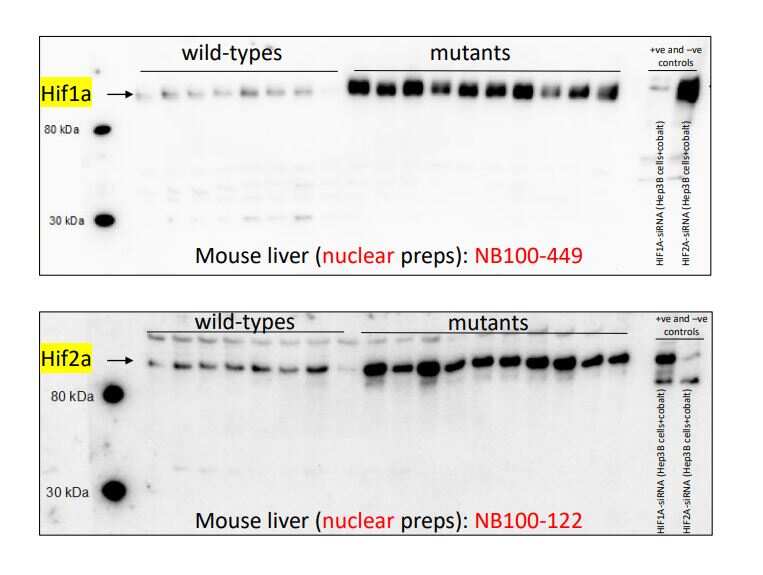
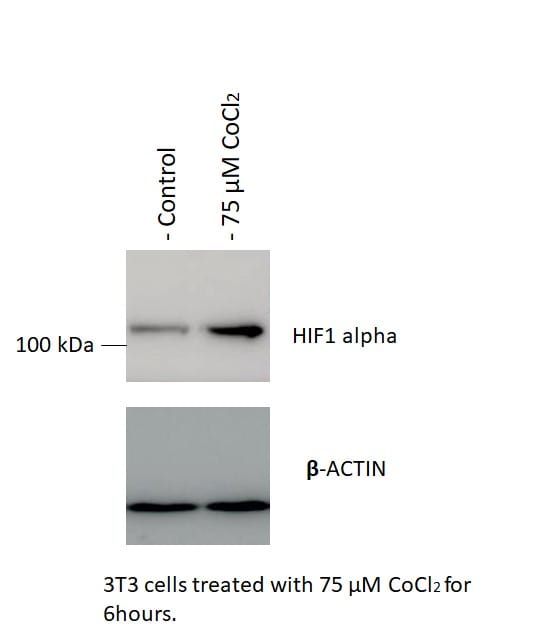
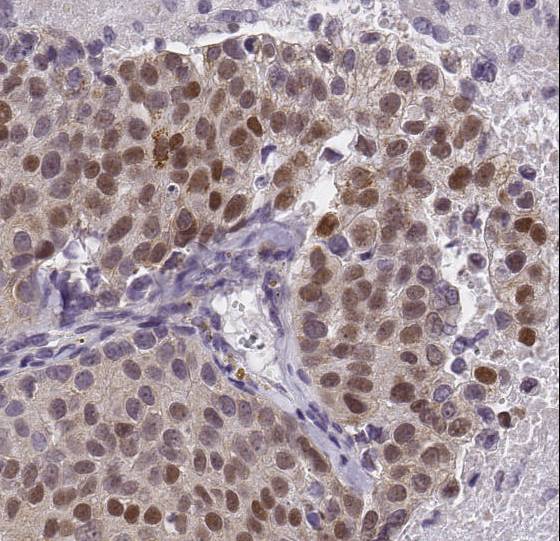
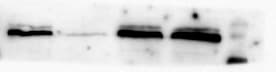
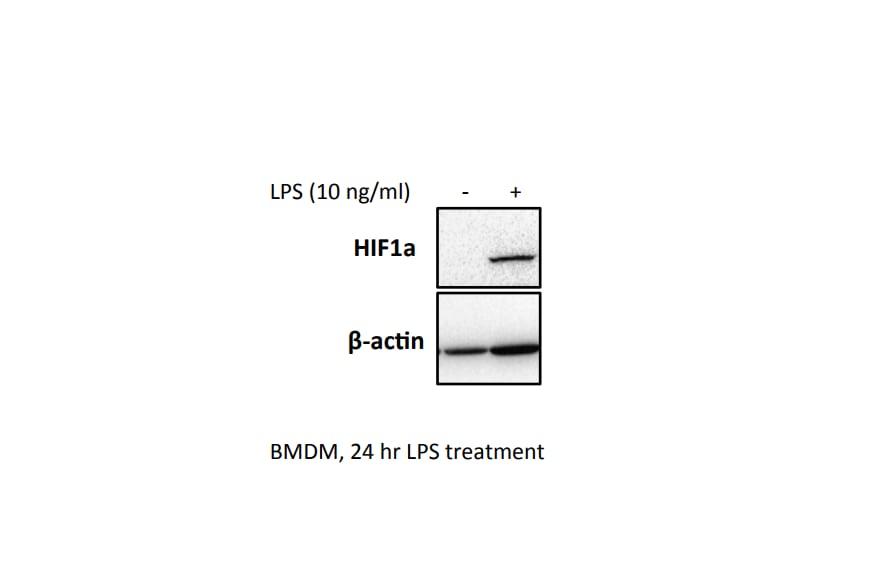
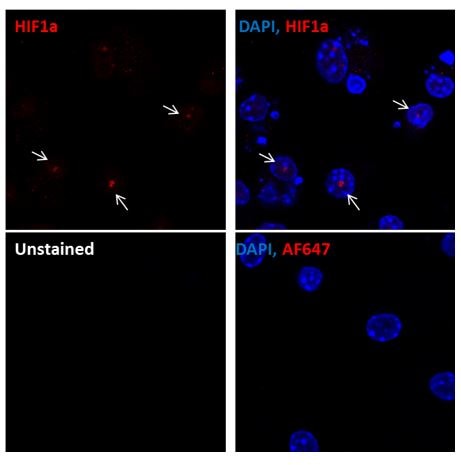


-(01-ml)_NB100-449_9376.jpg)
-(01-ml)_NB100-449_8916.gif)
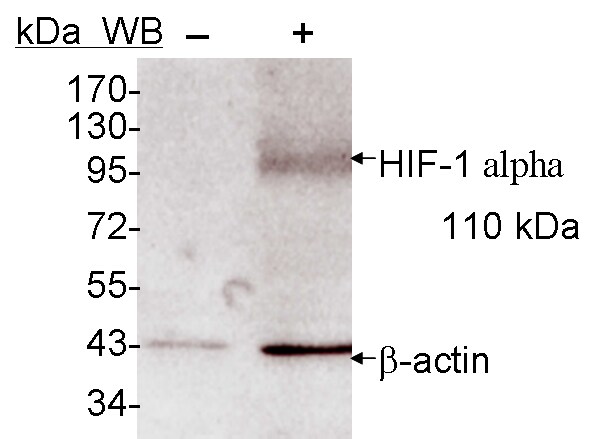
-(01-ml)_NB100-449_7166.jpg)
-(01-ml)_NB100-449_6456.gif)