Endoglin (CD105) is a 90 kDa type I transmembrane glycoprotein of the zona pellucida (ZP) family of proteins (1‑3). Endoglin and betaglycan/T beta RIII are type III receptors for TGF beta superfamily ligands, sharing 71% aa identity in the transmembrane (TM) and cytoplasmic domains. Endoglin is highly expressed on proliferating vascular endothelial cells, chondrocytes, and syncytiotrophoblasts of term placenta, with lower amounts on hematopoietic, mesenchymal and neural crest stem cells, activated monocytes, and lymphoid and myeloid leukemic cells (2-5). Human Endoglin cDNA encodes 658 amino acids (aa) including a 25 aa signal sequence, a 561 aa extracellular domain (ECD) with an orphan domain and a two-part ZP domain, a TM domain and a 47 aa cytoplasmic domain (1-3). An isoform with a 14 aa cytoplasmic domain (S-endoglin) can oppose effects of long (L) Endoglin (6, 7). The human Endoglin ECD shares 65-72% aa identity with mouse, rat, bovine, porcine and canine Endoglin. Endoglin homodimers interact with TGF-beta 1 and TGF-beta 3 (but not TGF-beta 2), but only after binding T beta RII (8). Similarly, they interact with activin-A and BMP-7 via activin type IIA or B receptors, and with BMP-2 via BMPR-1A/ALK-3 or BMPR-1B/ALK-6 (9). BMP-9, however, is reported to bind Endoglin directly (10). Endoglin modifies ligand-induced signaling in multiple ways. For example, expression of Endoglin can inhibit TGF-beta 1 signals but enhance BMP7 signals in the same myoblast cell line (11). In endothelial cells, Endoglin inhibits T beta RI/ALK5, but enhances ALK1-mediated activation (12). Deletion of mouse Endoglin causes lethal vascular and cardiovascular defects, and human Endoglin haploinsufficiency can a cause the vascular disorder, hereditary hemorrhagic telangiectasia type I (13, 14). These abnormalities confirm the essential function of Endoglin in differentiation of smooth muscle, angiogenesis, and neovascularization (2‑4, 12‑14). In preeclampsia of pregnancy, high levels of proteolytically generated soluble Endoglin and VEGF R1 (sFLT1), along with low placental growth factor (PlGF), are pathogenic due to antiangiogenic activity (15).
Human Endoglin/CD105 PE‑conjugated Antibody
R&D Systems | Catalog # FAB10971P

Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Glu26-Gly586
Accession # Q5T9B9
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human Endoglin/CD105 PE‑conjugated Antibody
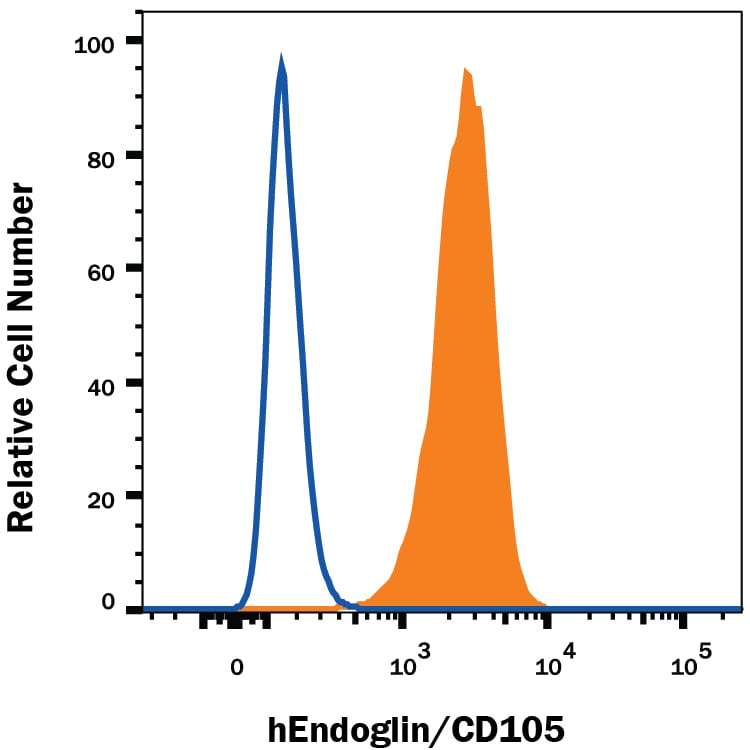
Detection of Endoglin/CD105 in U937 Human Cell Line by Flow Cytometry.
U937 human histiocytic lymphoma cell line was stained with Mouse Anti-Human Endoglin/CD105 PE-conjugated Mono-clonal Antibody (Catalog # FAB10971P, filled histogram) or isotype control antibody (Catalog # IC002P, open histogram). View our protocol for Staining Membrane-associated Proteins.Detection of Human Endoglin/CD105 by Flow Cytometry
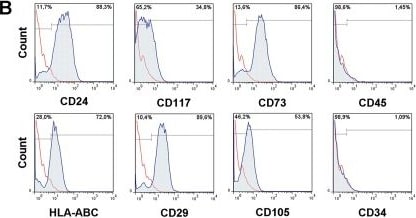
Isolation and characterization of amniotic derived mesenchymal stem cells with renal marker expression(A) Light microscopic image of mesenchymal stem cells isolated from amniotic fluid. A subpopulation of these cells (with renal markers) displays a trumpet shaped phenotype (scale bar = 200 μm). (B) hAFSCs were characterized by flow cytometry (positive for CD24, CD117, CD73, HLA-ABC, CD29, and CD105 but not for CD45, or CD34). (C) Confocal imaging of hAFSCs for renal markers SIX2 and PAX2. (D) Expression of Six2 and Pax2 (left panel) in hAFSCs compared to human embryonic kidney (hEK). Agarose gel analysis of PCR fragments (SIX2: 336 bp; PAX2: 65 bp; KSP: 152bp; GAPDH: 189 bp) (E). hAFSCs were differentiated along adipogenic, osteogenic and chondrogenic lineages (left to right). In the adipogenic differentiation the cells formed lipid vesicles stained with oil red O (left) (magnification 100X). Calcium deposits stained with Alizarin confirmed osteogenesis (center). Micromasses stained with alcian blue confirmed chondrogenesis (right) (scale bar = 200 μm). Quantification of chondrogenic and osteogenic differentiation using dye extraction and quantification with spectrophotometry before and after differentiation (left panel). qPCR analysis of chondrogenic markers (Sox9, Col II) and osteogenic markers (ALP, Runx2 and OST) before and after differentiation (right panel). Image collected and cropped by CiteAb from the following publication (https://dx.plos.org/10.1371/journal.pone.0136145), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human Endoglin/CD105 PE‑conjugated Antibody
Flow Cytometry
Sample: U937 human histiocytic lymphoma cell line
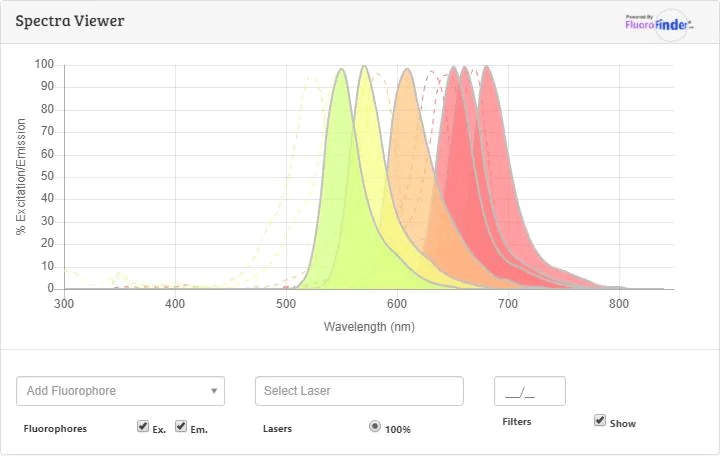
Spectra Viewer
Plan Your Experiments
Use our spectra viewer to interactively plan your experiments, assessing multiplexing options. View the excitation and emission spectra for our fluorescent dye range and other commonly used dyes.
Spectra ViewerFlow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, 2 to 8 °C as supplied.
Background: Endoglin/CD105
References
- Gougos, A. and Letarte, M. (1990) J. Biol. Chem. 265:8361.
- ten Dijke, P. et al. (2008) Angiogenesis 11:79.
- Bernabeu, C. et al. (2007) J. Cell. Biochem. 102:1375.
- Mancini, M.L. et al. (2007) Dev. Biol. 308:520.
- Moody, J.L. et al. (2007) Stem Cells 25:2809.
- Velasco, S. et al. (2008) J. Cell Sci. 121:913.
- Perez-Gomez, E. et al. (2005) Oncogene 24:4450.
- Cheifetz, S, et al. (1992) J. Biol. Chem. 267:19027.
- Barbara, N.P. et al. (1999) J. Biol. Chem. 274:584.
- Scharpfenecker, M. et al. (2007) J. Cell Sci. 120:964.
- Scherner, O. et al. (2007) J. Biol. Chem. 282:13934.
- Pece-Barbara, N. et al. (2005) J. Biol. Chem. 280:27800.
- Arthur, H.M. et al. (2000) Dev. Biol. 217:42.
- Lebrin, F. and C.L. Mummery (2008) Trends Cardiovasc. Med. 18:25.
- Venkatesha, S. et al. (2006) Nat. Med. 12:642.
Alternate Names
Gene Symbol
UniProt
Additional Endoglin/CD105 Products
Product Documents for Human Endoglin/CD105 PE‑conjugated Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Endoglin/CD105 PE‑conjugated Antibody
For research use only
Citations for Human Endoglin/CD105 PE‑conjugated Antibody
Customer Reviews for Human Endoglin/CD105 PE‑conjugated Antibody
There are currently no reviews for this product. Be the first to review Human Endoglin/CD105 PE‑conjugated Antibody and earn rewards!
Have you used Human Endoglin/CD105 PE‑conjugated Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- View all Protocols, Troubleshooting, Illustrated assays and Webinars