Growth hormone (GH), also known as somatotropin, is a member of a family of growth factors that includes prolactin, placental lactogens, proliferins and somatolactin (1, 2). It is synthesized primarily by somatotropes in the anterior pituitary and is released as an endocrine hormone. Other cells and tissues, including lymphoid tissues, can also produce GH (3). GH is a pleiotropic molecule which can act directly or indirectly via IGF-I, to regulate growth and metabolism as well as enhance T cell survival and thymic functions (1, 2, 4). GH exerts its biological actions by binding to the GH receptor (GHR) that is present in many cell types (1, 2). Human GHR cDNA encodes a 638 amino acid (aa) residue type I transmembrane protein with an 18 aa putative signal peptide, a 246 aa extracellular domain, a 24 aa transmembrane domain and a 350 aa cytoplasmic domain (5). At least two alternatively spliced isoforms of human GHR, lacking the sequence encoded by exon 3, or lacking most of the cytoplasmic domain, also exist (6, 7). Soluble GH-binding proteins corresponding to extracellular domain of the transmembrane proteins can be generated from the membrane proteins (8). Ligation of GHR by GH has been shown to result in receptor dimerization and activation of the JAK/STAT signaling cascade (9). The soluble GHBP has been shown to interfere with GH signaling by competing with the transmembrane receptor of GH. Alternatively, the GHBP has also been shown to enhance GH action by slowing GH clearance (8, 10).
Human Growth Hormone R/GHR Biotinylated Antibody
R&D Systems | Catalog # BAF1210

Key Product Details
Species Reactivity
Applications
Label
Antibody Source
Product Specifications
Immunogen
Ala27-Tyr264
Accession # P10912
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human Growth Hormone R/GHR Biotinylated Antibody
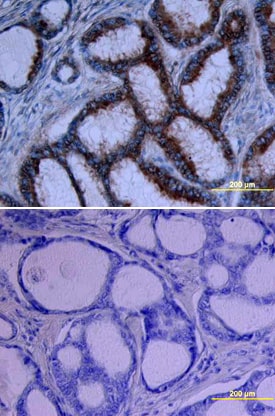
Growth Hormone R/GHR in Human Breast Cancer Tissue.
Growth Hormone R/GHR was detected in immersion fixed paraffin-embedded sections of human breast cancer tissue using Goat Anti-Human Growth Hormone R/GHR Biotinylated Antigen Affinity-purified Polyclonal Antibody (Catalog # BAF1210) at 15 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS008) and counterstained with hematoxylin (blue). Lower panel shows a lack of labeling if primary antibodies are omitted and tissue is stained only with secondary antibody followed by incubation with detection reagents. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Applications for Human Growth Hormone R/GHR Biotinylated Antibody
Flow Cytometry
Sample: Human whole blood CD19+ B cells
Immunohistochemistry
Sample: Immersion fixed paraffin-embedded sections of human breast cancer tissue
Western Blot
Sample: Recombinant Human Growth Hormone R/GHR Fc Chimera (Catalog # 1210-GR)
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS.
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Growth Hormone R/GHR
References
- Goffin, V. et al. (1996) Endocrine Rev. 17:385.
- Le Roith, D. et al. (2001) Endocrine Rev. 22:53.
- Clark, R. (1997) Endocr. Rev. 18:157.
- Welniak, L.A. et al. (2002) J. Leukoc. Biol. 71:381.
- Leung, D.W. et al. (1987) Nature 330:537.
- Stallings-Mann, J.L. et al. (1996) Proc. Nat. Acad. Sci. 93:12394.
- Amit, T. et al. (1997) Endocr. Metab. 82:3813.
- Ross, R.J.M., et al. (1997) Molecular Endocrinology 11:265.
- Carter-Su, C. et al. (1996) Annu. Rev. Physiol. 58:187.
- Postel-Vinay, M.C. and J. Finidori (1995) Eur. J. Endocrinol. 133:654.
Long Name
Alternate Names
Gene Symbol
UniProt
Additional Growth Hormone R/GHR Products
Product Documents for Human Growth Hormone R/GHR Biotinylated Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Growth Hormone R/GHR Biotinylated Antibody
For research use only
Customer Reviews for Human Growth Hormone R/GHR Biotinylated Antibody
There are currently no reviews for this product. Be the first to review Human Growth Hormone R/GHR Biotinylated Antibody and earn rewards!
Have you used Human Growth Hormone R/GHR Biotinylated Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars

