Human IL-17 Quantikine ELISA Kit
R&D Systems | Catalog # D1700


Key Product Details
Assay Length
Sample Type & Volume Required Per Well
Sensitivity
Assay Range
Product Summary for Human IL-17 Quantikine ELISA Kit
Product Specifications
Assay Type
Format
Measurement
Detection Method
Conjugate
Species
Specificity
Cross-reactivity
Interference
Precision
Intra-Assay Precision (Precision within an assay) Three samples of known concentration were tested on one plate to assess intra-assay precision.
Inter-Assay Precision (Precision between assays) Three samples of known concentration were tested in separate assays to assess inter-assay precision.
Cell Culture Supernates
| Intra-Assay Precision | Inter-Assay Precision | |||||
|---|---|---|---|---|---|---|
| Sample | 1 | 2 | 3 | 1 | 2 | 3 |
| n | 20 | 20 | 20 | 20 | 20 | 20 |
| Mean (pg/mL) | 107 | 604 | 1217 | 76.6 | 272 | 549 |
| Standard Deviation | 7.8 | 25.0 | 63.3 | 6.5 | 23.4 | 43.9 |
| CV% | 7.3 | 4.1 | 5.2 | 8.5 | 8.6 | 8.0 |
Citrate Plasma, EDTA Plasma, Heparin Plasma, Serum
| Intra-Assay Precision | Inter-Assay Precision | |||||
|---|---|---|---|---|---|---|
| Sample | 1 | 2 | 3 | 1 | 2 | 3 |
| n | 20 | 20 | 20 | 20 | 20 | 20 |
| Mean (pg/mL) | 131 | 736 | 1480 | 125 | 408 | 794 |
| Standard Deviation | 5.8 | 34.6 | 61.2 | 9.8 | 34.3 | 55.6 |
| CV% | 4.4 | 4.7 | 4.1 | 7.8 | 8.4 | 7.0 |
Recovery for Human IL-17 Quantikine ELISA Kit
The recovery of human IL-17 spiked to three different levels throughout the range of the assay in various matrices was evaluated.
| Sample Type | Average % Recovery | Range % |
|---|---|---|
| Cell Culture Media (n=5) | 97 | 88-112 |
| Citrate Plasma (n=5) | 105 | 92-116 |
| EDTA Plasma (n=5) | 101 | 90-110 |
| Heparin Plasma (n=5) | 101 | 93-109 |
| Serum (n=5) | 98 | 90-105 |
Linearity
To assess the linearity of the assay, five samples spiked with high concentrations of IL-17 were diluted with the appropriate Calibrator Diluent to produce samples with values within the dynamic range of the assay.

Scientific Data Images for Human IL-17 Quantikine ELISA Kit
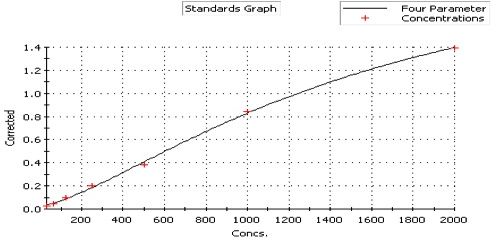
Human IL-17/IL-17A ELISA Cell Culture Supernate Standard Curve
Human IL-17/IL-17A ELISA Serum/Plasma Standard Curve
Preparation and Storage
Shipping
Stability & Storage
Background: IL-17/IL-17A
Long Name
Alternate Names
Entrez Gene IDs
Gene Symbol
Additional IL-17/IL-17A Products
Product Documents for Human IL-17 Quantikine ELISA Kit
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human IL-17 Quantikine ELISA Kit
For research use only
⚠ WARNING: This product can expose you to chemicals including N,N-Dimethylforamide, which is known to the State of California to cause cancer. For more information, go to www.P65Warnings.ca.gov.Citations for Human IL-17 Quantikine ELISA Kit
Customer Reviews for Human IL-17 Quantikine ELISA Kit (1)
Have you used Human IL-17 Quantikine ELISA Kit?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Sample Tested: T cellsSpecies: HumanVerified Customer | Posted 10/26/2015Standard curve is shown. <br />Buffer: kit dilution buffer<br />Dilution: 1/100
There are no reviews that match your criteria.
Protocols
View specific protocols for Human IL-17 Quantikine ELISA Kit (D1700):
- Prepare all reagents, standard dilutions, and samples as directed in the product insert.
- Remove excess microplate strips from the plate frame, return them to the foil pouch containing the desiccant pack, and reseal.
- Add 100 µL of Assay Diluent to each well.
- Add 100 µL of Standard, control, or sample to each well. Cover with a plate sealer, and incubate at room temperature for 3 hours.
- Aspirate each well and wash, repeating the process twice for a total of 3 washes.
- Add 200 µL of Conjugate to each well. Cover with a new plate sealer, and incubate at room temperature for 1 hour.
- Aspirate and wash 3 times.
- Add 200 µL Substrate Solution to each well. Incubate at room temperature for 30 minutes. PROTECT FROM LIGHT.
- Add 50 µL of Stop Solution to each well. Read at 450 nm within 30 minutes. Set wavelength correction to 540 nm or 570 nm.




Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- ELISA Sample Preparation & Collection Guide
- ELISA Troubleshooting Guide
- How to Run an R&D Systems DuoSet ELISA
- How to Run an R&D Systems Quantikine ELISA
- How to Run an R&D Systems Quantikine™ QuicKit™ ELISA
- Quantikine HS ELISA Kit Assay Principle, Alkaline Phosphatase
- Quantikine HS ELISA Kit Principle, Streptavidin-HRP Polymer
- Sandwich ELISA (Colorimetric) – Biotin/Streptavidin Detection Protocol
- Sandwich ELISA (Colorimetric) – Direct Detection Protocol
- Troubleshooting Guide: ELISA
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Associated Pathways
 Innate Lymphoid Cell Differentiation Pathways
Innate Lymphoid Cell Differentiation Pathways
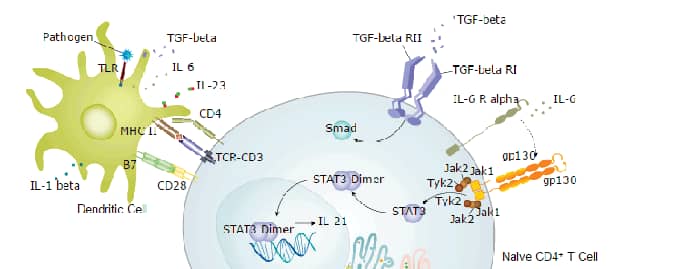
 Th17 Differentiation Pathway
Th17 Differentiation Pathway