M-Cadherin (M-CAD or Cadherin-15) is a 124 kDa type I transmembrane glycoprotein of the Cadherin superfamily of calcium-dependent homotypic adhesion molecules (1-4). Like other classical Cadherins, the 814 amino acid (aa) human M-CAD contains a signal sequence (21 aa), a propeptide (29 aa), an extracellular domain with five Cadherin domain repeats (ECD, 556 aa), a transmembrane segment (20 aa) and a cytoplasmic domain (188 aa) (1). A splice variant that diverges within the third Cadherin repeat has been sequenced. The Cadherin repeats are responsible for cell-cell adhesion by homophilic binding on opposing cells (1-4). Intracellularly, M-CAD binds beta -catenin or plakoglobin ( gamma -catenin), which in turn bind alpha -catenin (4). M-CAD also binds p120 catenin (5). Connection of Cadherin/catenin complexes to the actin cytoskeleton is in question, but is possible through a linker (6). Connection to microtubules has been shown (7). M-CAD is present during early stages of skeletal muscle development and is thought to align myoblasts for fusion (8). It is also present in muscle satellite cells and participates in muscle regeneration (9). M-CAD is also expressed in the granule cell layer of the cerebellar glomerulus (10). Deletion of mouse M-CAD has little effect in vivo, most likely due to compensation by N-Cadherin (11). However, M-CAD upregulation and adhesion between myoblasts during induction of differentiation in vitro is required for their fusion (8, 12, 13). M-CAD activity is later downregulated by sequestering to caveoli, p120 catenin/RhoA-induced ubiquitination and/or cleavage by calpain-3. This terminates fusion and allows sarcomere formation (5, 12, 13). Human M-CAD ECD shows 88% aa identity with mouse, rat, or bovine and 85% aa identity with canine M-CAD ECD. M-CAD is an outlier among classical Cadherins, with 40% aa identity or less in the ECD (4).
Human M‑Cadherin/Cadherin‑15 Antibody
R&D Systems | Catalog # AF4096

Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Val22-Ala606
Accession # P55291
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human M‑Cadherin/Cadherin‑15 Antibody
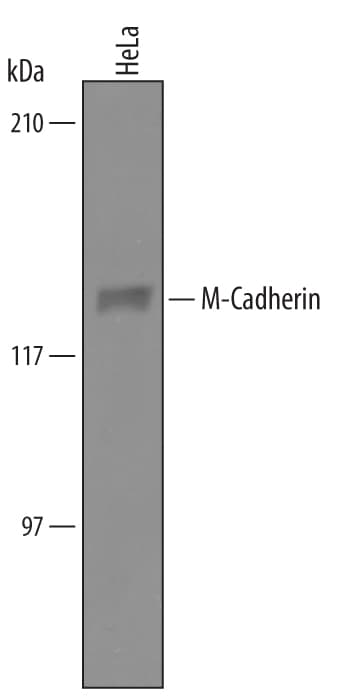
Detection of Human M-Cadherin/Cadherin-15 by Western Blot.
Western blot shows lysates of HeLa human cervical epithelial carcinoma cell line. PVDF Membrane was probed with 1 µg/mL of Sheep Anti-Human M-Cadherin/Cadherin-15 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF4096) followed by HRP-conjugated Anti-Sheep IgG Secondary Antibody (Catalog # HAF016). A specific band was detected for M-Cadherin/Cadherin-15 at approximately 130 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 8.Detection of M‑Cadherin/ Cadherin‑15 in C2C12 Mouse Cell Line by Flow Cytometry.
C2C12 mouse myoblast cell line was stained with Sheep Anti-Human M-Cadherin/ Cadherin-15 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF4096, filled histogram) or control antibody (Catalog # 5-001-A, open histogram), followed by NorthernLights™ 557-conjugated Anti-Sheep IgG Secondary Antibody (Catalog # NL010).M‑Cadherin/Cadherin-15 in C2C12 Mouse Cell Line.
M-Cadherin/Cadherin-15 was detected in immersion fixed C2C12 mouse myoblast cell line using Sheep Anti-Human M-Cadherin/Cadherin-15 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF4096) at 10 µg/mL for 3 hours at room temperature. Cells were stained using the Northern-Lights™ 557-conjugated Anti-Mouse IgG Secondary Antibody (red; Catalog # NL007) and counter-stained with DAPI (blue). Specific staining was localized to cell surfaces and cytoplasm. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Applications for Human M‑Cadherin/Cadherin‑15 Antibody
CyTOF-ready
Flow Cytometry
Sample: C2C12 mouse myoblast cell line
Immunocytochemistry
Sample: Immersion fixed C2C12 mouse myoblast cell line
Western Blot
Sample: HeLa human cervical epithelial carcinoma cell line
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: M-Cadherin/Cadherin-15
References
- Kaufmann, U. et al. (1999) Cell Tissue Res. 296:191.
- Angst, B.D. et al. (2001) J. Cell Sci. 114:629.
- Shimoyama, Y. et al. (1998) J. Biol. Chem. 273:10011.
- Kuch, C. et al. (1997) Exp. Cell Res. 232:331.
- Charasse, S. et al. (2006) Mol. Biol. Cell 17:749.
- Weis, W.I. and W. J. Nelson (2006) J. Biol. Chem. 281:35593.
- Kaufmann, U. et al. (1999) J. Cell Sci. 112:55.
- Zeschnigk, M. et al. (1995) J. Cell Sci. 108:2973.
- Wrobel, E. et al. (2007) Eur. J. Cell Biol. Jan 10; [Epub ahead of print]
- Rose, O. et al. (1995) Proc. Natl. Acad. Sci. USA 92:6022.
- Hollnagel, A. et al. (2002) Mol. Cell. Biol. 22:4760.
- Volonte, D. et al. (2003) Mol. Biol. Cell 14:4075.
- Kramerova, I. et al. (2006) Mol. Cell. Biol. 26:8437.
Long Name
Alternate Names
Gene Symbol
UniProt
Additional M-Cadherin/Cadherin-15 Products
Product Documents for Human M‑Cadherin/Cadherin‑15 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human M‑Cadherin/Cadherin‑15 Antibody
For research use only
Related Research Areas
Citations for Human M‑Cadherin/Cadherin‑15 Antibody
Customer Reviews for Human M‑Cadherin/Cadherin‑15 Antibody
There are currently no reviews for this product. Be the first to review Human M‑Cadherin/Cadherin‑15 Antibody and earn rewards!
Have you used Human M‑Cadherin/Cadherin‑15 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Associated Pathways




