Human/Mouse PEA‑15 Antibody
R&D Systems | Catalog # AF5588


Key Product Details
Validated by
Biological Validation
Species Reactivity
Validated:
Human, Mouse
Cited:
Human
Applications
Validated:
Immunohistochemistry, Western Blot
Cited:
Immunohistochemistry
Label
Unconjugated
Antibody Source
Polyclonal Sheep IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant human PEA-15
Ala2-Ala130
Accession # Q15121
Ala2-Ala130
Accession # Q15121
Specificity
Detects human and mouse PEA-15 in direct ELISAs and Western blots.
Clonality
Polyclonal
Host
Sheep
Isotype
IgG
Scientific Data Images for Human/Mouse PEA‑15 Antibody
Detection of Human PEA‑15 by Western Blot.
Western blot shows lysates of A172 human glioblastoma cell line and human cortex tissue. PVDF membrane was probed with 1 µg/mL of Sheep Anti-Human/Mouse PEA-15 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF5588) followed by HRP-conjugated Anti-Sheep IgG Secondary Antibody (Catalog # HAF016). A specific band was detected for PEA-15 at approximately 15 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 8.PEA‑15 in Human Brain.
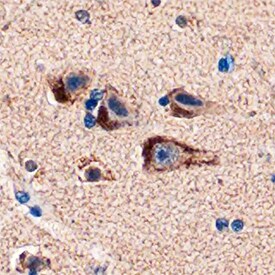
PEA-15 was detected in immersion fixed paraffin-embedded sections of human brain (cortex) using Sheep Anti-Human/Mouse PEA-15 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF5588) at 1 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Sheep HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS019) and counterstained with hematoxylin (blue). Specific staining was localized to neurons. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Detection of Human PEA-15 by Western Blot
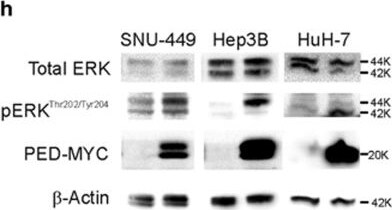
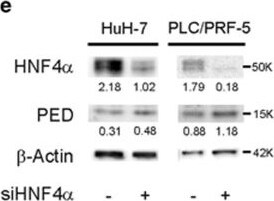
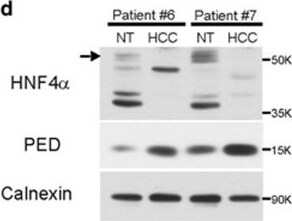
PED is inversely correlated to HNF4 alpha expression. (a) SNU-449 cells were co-transfected with 100 ng of pPED477 PED promoter-luciferase or pGL3 basic construct and treated with siRNA against HNF4 alpha or siRNA control. Luciferase activity was normalized for Renilla activity and is presented as mean±S.D. A representative experiment in triplicate is shown. (b,c) PED expression levels in HCC samples (b; n=59) or corresponding non-tumoral liver tissue (c, n=59) were correlated with HNF4 alpha expression. Correlation was calculated by Spearman test. Data are reported as probe intensity of an mRNA transcriptome array. (d) Western blot analysis for HNF4 alpha and PED in two HCC patient tumor samples and their corresponding non-tumoral (NT) tissues. Calnexin was used as loading control. Arrow: canonical full length HNF4 alpha (52 kDa); other bands are isoforms or truncated forms of the protein. (e,f) HuH-7 and PLC/PRF/5 cell lines were transfected with siRNA against HNF4 alpha (siHNF4 alpha ) or siRNA control. After 72 h the protein expression of HNF4 alpha and PED was measured by western blot (e) and beta -actin served as control. mRNA expression was measured by qPCR (f) using RNA 18 S as internal control at 48 h for HuH-7 and 72 h for PLC/PRF/5. Data are reported as mean±S.D. of two independent experiments performed in triplicate. (g) SNU-449 cells were transfected with siRNA against HNF4 alpha or siRNA against PED alone or in combination, or siRNA control, as indicated. Migration was assessed by CIM plate with xCELLigence apparatus after 12 h and 24 h. Data are reported as mean±S.D. of two independent experiments performed in triplicate. (h) Western blot analysis of pERKThr202/Tyr204 and ERK in SNU-449, Hep3B and HuH-7 cell lines transfected with PED-MYC. beta -Actin was used as loading control. (i) pERKThr202/Tyr204 expression in two HCC patients and their non-tumoral counterpart. Calnexin was used as loading control. *P<0.05, **P<0.01, ****P<0.0001 Image collected and cropped by CiteAb from the following publication (https://www.nature.com/articles/cddis2017512), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human PEA-15 by Western Blot
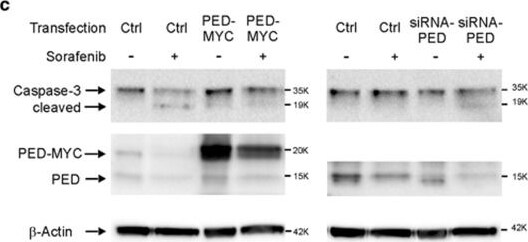
PED confers resistance to sorafenib therapy. (a) HuH-7 and SNU-449 cells were transfected with siRNA against PED or siRNA control. Afterwards, HuH-7 and SNU-449 cells were treated with 10 μM and 20 μm respectively of sorafenib or left untreated. Cell growth was evaluated by using the xCELLigence instrument at the indicated time. Data are reported as mean±S.D. of two independent experiments performed in triplicate. (b) HuH-7 and Hep3B cells were transfected with PED-MYC vector for 24 h and then seeded in a 96-well plate. 10 μm of sorafenib was added and 24 h or 48 h later, cell viability was measured by a MTT assay. Data are reported as mean±S.D. of two independent experiments perfomed in triplicate. (c) HuH-7 cells were transfected with PED-MYC or empty vector (Ctrl) and siRNA against PED or siRNA control (Ctrl), as indicated. After 24 h, cells were treated with 10 μM or 7 μM sorafenib, respectively, for 48 h and caspase-3 activation was measured by western blot. (d) HuH-7 cells were transfected with siRNA against PED or control siRNA. Afterwards, cells were treated with 10 μM sorafenib and 48 h later caspase-3/7 assay activation was measured. Data are reported as mean±SD of one experiment performed in triplicate. *P<0.1, **P<0.01, ***P<0.001, ****P<0.0001 Image collected and cropped by CiteAb from the following publication (https://www.nature.com/articles/cddis2017512), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human PEA-15 by Western Blot
PED is inversely correlated to HNF4 alpha expression. (a) SNU-449 cells were co-transfected with 100 ng of pPED477 PED promoter-luciferase or pGL3 basic construct and treated with siRNA against HNF4 alpha or siRNA control. Luciferase activity was normalized for Renilla activity and is presented as mean±S.D. A representative experiment in triplicate is shown. (b,c) PED expression levels in HCC samples (b; n=59) or corresponding non-tumoral liver tissue (c, n=59) were correlated with HNF4 alpha expression. Correlation was calculated by Spearman test. Data are reported as probe intensity of an mRNA transcriptome array. (d) Western blot analysis for HNF4 alpha and PED in two HCC patient tumor samples and their corresponding non-tumoral (NT) tissues. Calnexin was used as loading control. Arrow: canonical full length HNF4 alpha (52 kDa); other bands are isoforms or truncated forms of the protein. (e,f) HuH-7 and PLC/PRF/5 cell lines were transfected with siRNA against HNF4 alpha (siHNF4 alpha ) or siRNA control. After 72 h the protein expression of HNF4 alpha and PED was measured by western blot (e) and beta -actin served as control. mRNA expression was measured by qPCR (f) using RNA 18 S as internal control at 48 h for HuH-7 and 72 h for PLC/PRF/5. Data are reported as mean±S.D. of two independent experiments performed in triplicate. (g) SNU-449 cells were transfected with siRNA against HNF4 alpha or siRNA against PED alone or in combination, or siRNA control, as indicated. Migration was assessed by CIM plate with xCELLigence apparatus after 12 h and 24 h. Data are reported as mean±S.D. of two independent experiments performed in triplicate. (h) Western blot analysis of pERKThr202/Tyr204 and ERK in SNU-449, Hep3B and HuH-7 cell lines transfected with PED-MYC. beta -Actin was used as loading control. (i) pERKThr202/Tyr204 expression in two HCC patients and their non-tumoral counterpart. Calnexin was used as loading control. *P<0.05, **P<0.01, ****P<0.0001 Image collected and cropped by CiteAb from the following publication (https://www.nature.com/articles/cddis2017512), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human PEA-15 by Western Blot
PED is inversely correlated to HNF4 alpha expression. (a) SNU-449 cells were co-transfected with 100 ng of pPED477 PED promoter-luciferase or pGL3 basic construct and treated with siRNA against HNF4 alpha or siRNA control. Luciferase activity was normalized for Renilla activity and is presented as mean±S.D. A representative experiment in triplicate is shown. (b,c) PED expression levels in HCC samples (b; n=59) or corresponding non-tumoral liver tissue (c, n=59) were correlated with HNF4 alpha expression. Correlation was calculated by Spearman test. Data are reported as probe intensity of an mRNA transcriptome array. (d) Western blot analysis for HNF4 alpha and PED in two HCC patient tumor samples and their corresponding non-tumoral (NT) tissues. Calnexin was used as loading control. Arrow: canonical full length HNF4 alpha (52 kDa); other bands are isoforms or truncated forms of the protein. (e,f) HuH-7 and PLC/PRF/5 cell lines were transfected with siRNA against HNF4 alpha (siHNF4 alpha ) or siRNA control. After 72 h the protein expression of HNF4 alpha and PED was measured by western blot (e) and beta -actin served as control. mRNA expression was measured by qPCR (f) using RNA 18 S as internal control at 48 h for HuH-7 and 72 h for PLC/PRF/5. Data are reported as mean±S.D. of two independent experiments performed in triplicate. (g) SNU-449 cells were transfected with siRNA against HNF4 alpha or siRNA against PED alone or in combination, or siRNA control, as indicated. Migration was assessed by CIM plate with xCELLigence apparatus after 12 h and 24 h. Data are reported as mean±S.D. of two independent experiments performed in triplicate. (h) Western blot analysis of pERKThr202/Tyr204 and ERK in SNU-449, Hep3B and HuH-7 cell lines transfected with PED-MYC. beta -Actin was used as loading control. (i) pERKThr202/Tyr204 expression in two HCC patients and their non-tumoral counterpart. Calnexin was used as loading control. *P<0.05, **P<0.01, ****P<0.0001 Image collected and cropped by CiteAb from the following publication (https://www.nature.com/articles/cddis2017512), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human PEA-15 by Western Blot
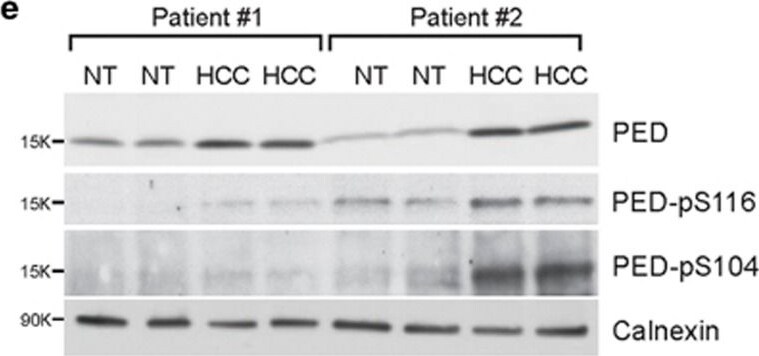
PED is overexpressed in HCC samples. (a) PED expression levels in HCC samples and their matched non-tumoral (NT) counterpart measured by an mRNA gene expression microarray. Data are reported as probe intensity. (b) PED mRNA was measured by qRT-PCR in a separate cohort of 14 HCC patients and compared with the 10 available non-tumoral counterpart. 18 S was used as internal control and 2− delta delta Ct formula was applied to determine relative expression levels. Statistical analysis (a,b) with paired Student t-test. (c) Representative immunohistochemical staining from an HCC tumor (left) with positive (3+) PED staining and non-tumoral liver tissue (NT) showing negative PED staining (right). Scale bar=20 μm. (d) Percentage and h-score (staining intensity × percentage of positive tumor cells) of PED positivity in HCC samples and non-tumoral (NT) liver tissues by immunohistochemistry. (e) Western blot analysis of total PED and phosphorylated PED (PED S116 and PED S104) in two HCC patient samples and their corresponding NT control tissues. Calnexin was used as internal control. *P<0.05, ***P<0.001 Image collected and cropped by CiteAb from the following publication (https://www.nature.com/articles/cddis2017512), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human PEA-15 by Western Blot
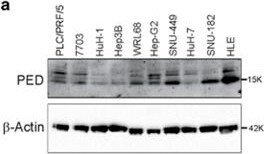
PED modulates cell migration. (a) Western blot analysis of PED protein expression in 10 different HCC cell lines. beta -Actin was used as loading control. (b) HuH-7 and SNU-449 cells were transfected with PED-MYC or an empty control vector as wells as with siRNA against PED (siRNA PED) or control siRNA. Cell growth properties were evaluated by using xCELLigence instrument at the time indicated. Data are reported as mean±S.D. of two independent experiments performed at least in triplicate. Difference was evaluated between PED overexpressing (PED-MYC), PED silenced (siRNA PED), empty vector transfected and a siRNA control transfected cells (two-way ANOVA test). (c) HLE, SNU-449 and HuH-7 cell lines were transfected with a vector overexpressing PED (PED-MYC) or empty control vector, siRNA against PED (siRNA PED) or siRNA control. Migration was assessed using a transwell assay after 24 h. One representative image of crystal violet stained cells at 100 × is shown above and quantification by colorimetry below. ***P<0.001, ****P<0.0001 Image collected and cropped by CiteAb from the following publication (https://www.nature.com/articles/cddis2017512), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human/Mouse PEA‑15 Antibody
Application
Recommended Usage
Immunohistochemistry
5-15 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human brain (cortex)
Sample: Immersion fixed paraffin-embedded sections of human brain (cortex)
Western Blot
1 µg/mL
Sample: A172 human glioblastoma cell line and human cortex tissue
Sample: A172 human glioblastoma cell line and human cortex tissue
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: PEA-15
Long Name
Phosphoprotein Enriched in Astrocytes 15
Alternate Names
HMAT1, HUMMAT1H, MAT1H, PEA15, PED
Gene Symbol
PEA15
UniProt
Additional PEA-15 Products
Product Documents for Human/Mouse PEA‑15 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human/Mouse PEA‑15 Antibody
For research use only
Related Research Areas
Citations for Human/Mouse PEA‑15 Antibody
Customer Reviews for Human/Mouse PEA‑15 Antibody
There are currently no reviews for this product. Be the first to review Human/Mouse PEA‑15 Antibody and earn rewards!
Have you used Human/Mouse PEA‑15 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...