Mouse periostin, also known as OSF-2 (osteoblast-specific factor 2) is a 170 kDa, secreted, homodimeric protein that belongs to the periostin family of the FAS1 superfamily of molecules (1-4). It is a TGF-beta inducible molecule that serves as both an adhesion molecule and tumor suppressor (2, 5, 6, 7). It is synthesized as a 838 amino acid (aa) precursor that contains a 23 aa signal sequence and an 815 aa mature region (2, 8). It is unknown if the molecule has any significant glycosylation (2). Based on human OSF-2, the homodimer is not disulfide-linked (3). The molecule consists of two distinct regions. The N-terminus contains an 55 aa EMI domain, while theC-terminus contains four, 130 aa fasciculin type 1 (or FAS1) domains. The EMI domain is cysteine-rich and shows a highly basic alpha -helix (9). Each FAS1 repeat exhibits a novel 7-stranded beta -wedge with a multiple alpha -helix fold (1, 8). Multiple alternate splice forms are known to exist C-terminal (aa 672-812) to the four-fold FAS1 repeats. These mature molecules are 760, 761, 787, and 788 aa in length and show block deletions of 54 aa, 27 aa and/or 28 aa (10). The significance of the alternate splice forms is not clear. They do, however, appear to be temporally regulated (6). OSF-2 is known to bind to alpha v beta 3 and alpha v beta 5 integrins (3). It is synthesized by smooth muscle cells, fibroblasts, and osteoblasts (2, 5, 7). Mature mouse OSF-2 is 98%, 92%, and 91% aa identical to rat, canine, and human OSF-2, respectively.
Human/Mouse Periostin/OSF‑2 Antibody
R&D Systems | Catalog # MAB3548

Key Product Details
Species Reactivity
Validated:
Human, Mouse
Cited:
Human, Mouse
Applications
Validated:
Western Blot, Immunocytochemistry
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Western Blot
Label
Unconjugated
Antibody Source
Monoclonal Rat IgG2A Clone # 345613
Loading...
Product Specifications
Immunogen
S. frugiperda insect ovarian cell line Sf 21-derived recombinant mouse Periostin isoform 2
Asn24-Gln811
Accession # NP_056599
Asn24-Gln811
Accession # NP_056599
Specificity
Detects mouse Periostin/OSF-2 in direct ELISAs and Western blots.
Clonality
Monoclonal
Host
Rat
Isotype
IgG2A
Scientific Data Images for Human/Mouse Periostin/OSF‑2 Antibody
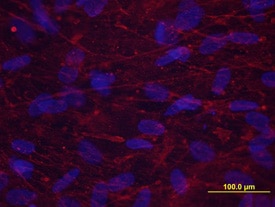
Periostin/OSF‑2 in Human Osteocytes.
Periostin/OSF-2 was detected in immersion fixed human osteocytes using Rat Anti-Human/Mouse Periostin/OSF-2 Monoclonal Antibody (Catalog # MAB3548) at 10 µg/mL for 3 hours at room temperature. Cells were stained red and counterstained with DAPI (blue).Applications for Human/Mouse Periostin/OSF‑2 Antibody
Application
Recommended Usage
Immunocytochemistry
8-25 µg/mL
Sample: Immersion fixed ST-2 mouse bone marrow-derived stromal cell line differentiated to osteocytes using Human/Mouse StemXVivo Osteogenic/Adipogenic Base Media (Catalog # CCM007) and Mouse StemXVivo Osteogenic supplement (Catalog # CCM009), and immersion fixed human osteocytes
Sample: Immersion fixed ST-2 mouse bone marrow-derived stromal cell line differentiated to osteocytes using Human/Mouse StemXVivo Osteogenic/Adipogenic Base Media (Catalog # CCM007) and Mouse StemXVivo Osteogenic supplement (Catalog # CCM009), and immersion fixed human osteocytes
Western Blot
1 µg/mL
Sample: Recombinant Mouse Periostin/OSF‑2 (Catalog # 2955-F2)
Sample: Recombinant Mouse Periostin/OSF‑2 (Catalog # 2955-F2)
Reviewed Applications
Read 1 review rated 5 using MAB3548 in the following applications:
Formulation, Preparation, and Storage
Purification
Protein A or G purified from hybridoma culture supernatant
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Periostin/OSF-2
References
- Clout, N.J. and D. Tisi (2003) Structure 11:197.
- Horiuchi, K. et al. (1999) J. Bone Miner. Res. 14:1239.
- Gillan, L. et al. (2002) Cancer Res. 62:5358.
- Litvin, J. et al. (2005) Anat. Rec. A Discov. Mol. Cell. Evol. Biol. 287A:1205.
- Lindner, V. et al. (2005) Arterioscler. Thromb. Vasc. Biol. 25:77.
- Kruzynska-Frejtag, A. et al. (2004) Dev. Dyn. 229:857.
- Yoshioka, N. et al. (2002) Exp. Cell Res. 279:91.
- Takeshita, S. et al. (1993) Biochem. J. 294:271.
- Callebaut, I. et al. (2003) Biochem. Biophys. Res. Commun. 300:619.
- Swiss-Prot Accession #:Q62009.
Long Name
Osteoblast Specific Factor 2
Alternate Names
Fasciclin I-like, OSF-2, OSF2, POSTN, TRIF52
Gene Symbol
POSTN
UniProt
Additional Periostin/OSF-2 Products
Product Documents for Human/Mouse Periostin/OSF‑2 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human/Mouse Periostin/OSF‑2 Antibody
For research use only
Related Research Areas
Citations for Human/Mouse Periostin/OSF‑2 Antibody
Customer Reviews for Human/Mouse Periostin/OSF‑2 Antibody (1)
5 out of 5
1 Customer Rating
Have you used Human/Mouse Periostin/OSF‑2 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
Application: Western BlotSample Tested: 4T1 mouse breast cancer cell lineSpecies: MouseVerified Customer | Posted 03/07/2020
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...

