Neuropilin-1 (Npn-1, previously neuropilin; also CD304/BDCA4) is a 130-140 kDa type I transmembrane (TM) glycoprotein that regulates axon guidance and angiogenesis (1-4). The full-length 923 amino acid (aa) human Npn-1 contains a 623 aa extracellular domain (ECD) that shows 92-95% aa identity with mouse, rat, bovine, and canine Npn-1 (3, 4). The ECD contains two N-terminal CUB domains (termed a1a2), two domains with homology to coagulation factors V and VIII (b1b2) and a MAM (meprin) domain (c). C-terminally divergent splice variants with 704, 644, 609, and 551 aa lack the MAM and TM domains and are demonstrated or presumed to be soluble antagonists (1, 5-7). A 906 aa form lacks a TM segment, but secretion has not been found (8). The sema domains of Class III secreted semaphorins such as Sema3A bind Npn-1 a1a2 (9). Heparin, the heparin-binding forms of VEGF (VEGF165, VEGF-B, and VEGF-E), PlGF (PlGF‑2), and the C-terminus of Sema3 bind the b1b2 region (9, 10). Npn-1 and Npn-2 share 48% aa identity within the ECD and can form homo- and hetero-oligomers via interaction of their MAM domains (1). Neuropilins show partially overlapping expression in neuronal and endothelial cells during development (1, 2). Both neuropilins act as co-receptors with plexins, mainly plexin A3 and A4, to bind class III semaphorins that mediate axon repulsion (11). However, only Npn-1 binds Sema3A, and only Npn-2 binds Sema3F (1). Both are co-receptors with VEGF R2 (also called KDR or Flk-1) for VEGF165 binding (1). Sema3A signaling can be blocked by VEGF165, which has higher affinity for Npn-1 (12). Npn-1 is preferentially expressed in arteries during development or those undergoing remodeling (1, 2). Npn-1 is also expressed on dendritic cells and mediates DC-induced T cell proliferation (13).

Key Product Details
Validated by
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Phe22-Lys644
Accession # NP_001019799
Specificity
Clonality
Host
Isotype
Endotoxin Level
Scientific Data Images for Human Neuropilin‑1 Antibody
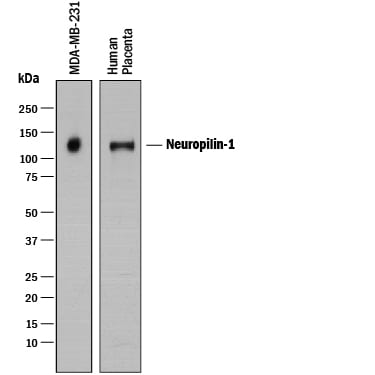
Detection of Human Neuropilin‑1 by Western Blot.
Western blot shows lysates of MDA‑MB‑231 human breast cancer cell line and human placenta tissue. PVDF membrane was probed with 1 µg/mL of Sheep Anti-Human Neuropilin‑1 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3870) followed by HRP-conjugated Anti-Sheep IgG Secondary Antibody (Catalog # HAF016). A specific band was detected for Neuropilin‑1 at approximately 130 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Detection of Neuropilin‑1 in HUVEC Human Cells by Flow Cytometry.
HUVEC human umbilical vein endothelial cells were stained with Sheep Anti-Human Neuropilin-1 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3870, filled histogram) or control antibody (Catalog # 5-001-A, open histogram), followed by NorthernLights™ 637-conjugated Anti-Sheep IgG Secondary Antibody (Catalog # NL011).Neuropilin‑1 in PC-3 and HLDM-2 Human Cell Lines.
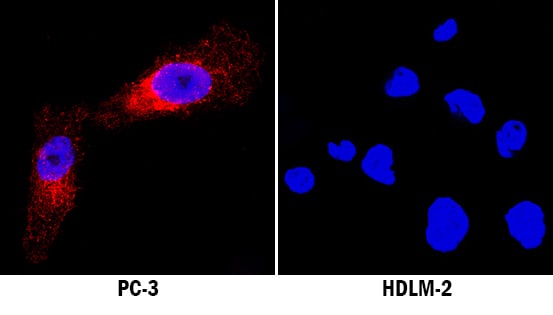
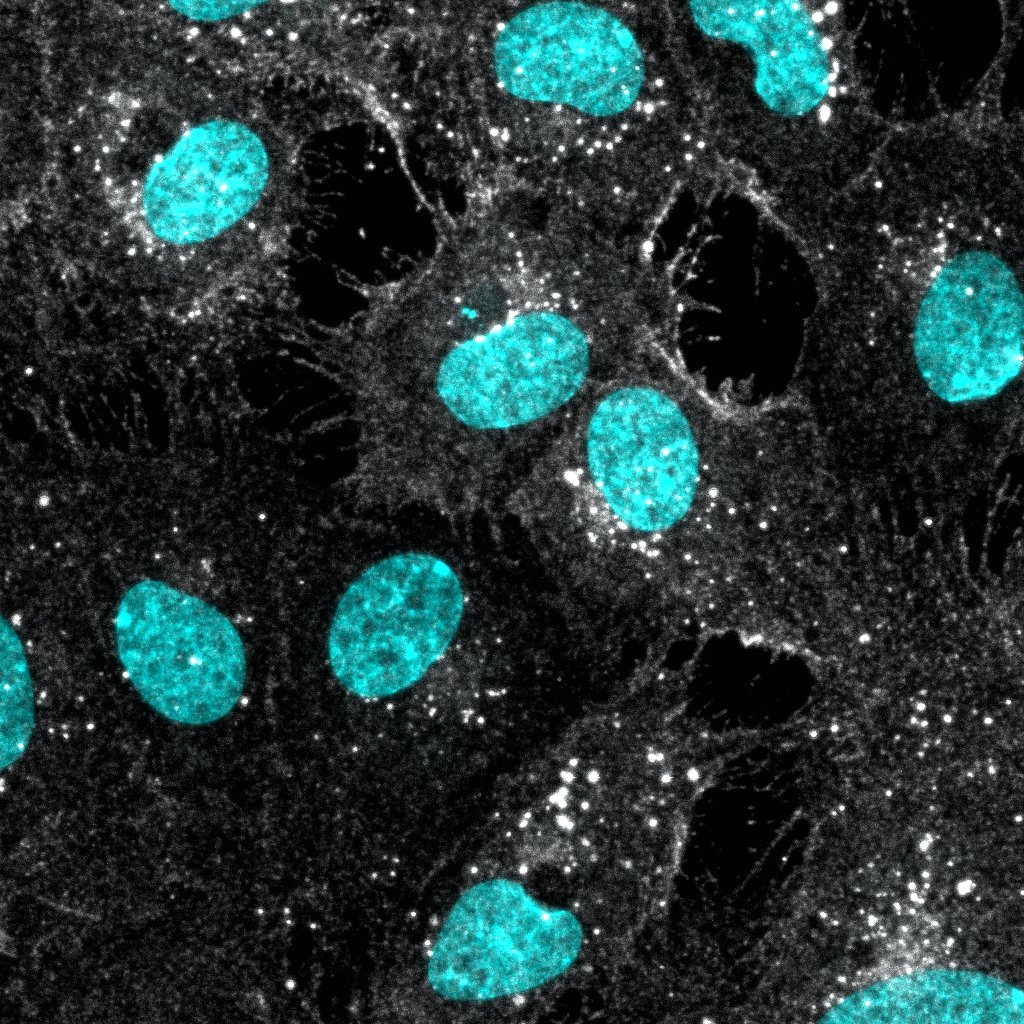
Neuropilin-1 was detected in immersion fixed PC-3 human prostate cancer cell line (positive staining) and HLDM-2 human Hodgkin's lymphoma cell line (negative staining) using Sheep Anti-Human Neuropilin-1 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3870) at 5 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Sheep IgG Secondary Antibody (red; Catalog # NL010) and counterstained with DAPI (blue). Specific staining was localized to cytoplasm. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Neuropilin-1 in Human Pancreatic Cancer Tissue.
Neuropilin-1 was detected in immersion fixed paraffin-embedded sections of human pancreatic cancer tissue using Sheep Anti-Human Neuropilin-1 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3870) at 10 µg/mL overnight at 4 °C. Before incubation with the primary antibody tissue was subjected to heat-induced epitope retrieval using Antigen Retrieval Reagent-Basic (Catalog # CTS013). Tissue was stained using the Anti-Sheep HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS019) and counterstained with hematoxylin (blue). Specific labeling was localized to the cytoplasm and plasma membrane of cancer cells. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Detection of Human Neuropilin-1 by Knockdown Validated
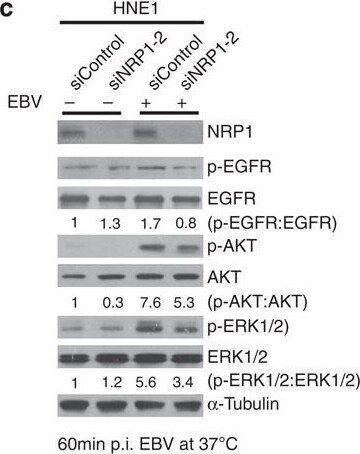
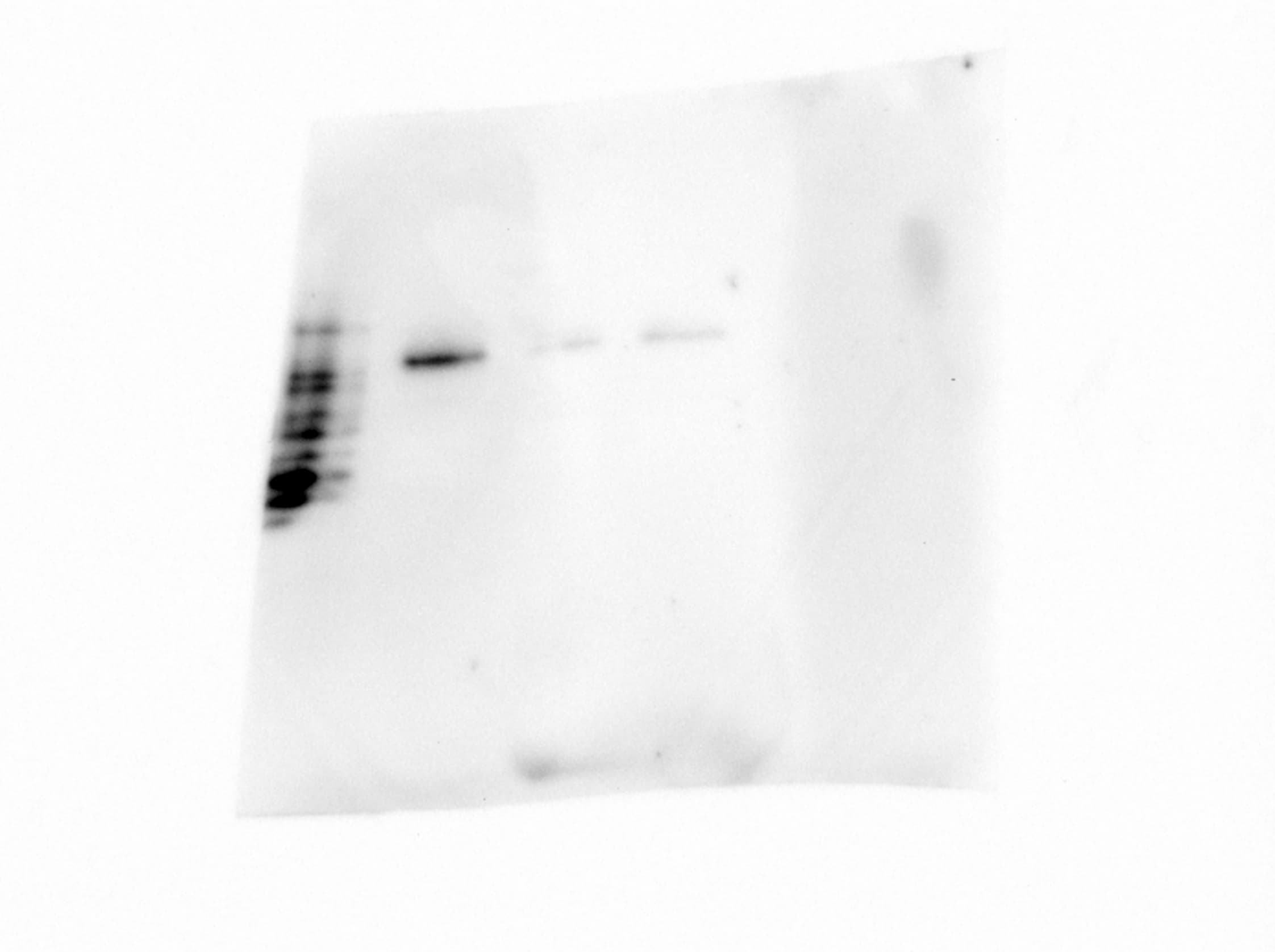
NRP1-dependent EGFR/ERK signalling pathways are activated by EBV and are also associated with the promotion of EBV infection.(a,b) EBV infection activated EGFR/AKT and EGFR/ERK pathways. Serum-starved HNE1 cells were infected with EBV (a) or purified EBV (b) at an MOI of 5 × 103 for the indicated times. (c) EBV-activated EGFR/AKT or EGFR/ERK pathways were partially mediated by NRP1. After transfected with the indicated siRNA duplexes for 48 h, HNE1 cells were serum-starved and infected with EBV at an MOI of 5 × 103 for 1 h, followed by western blotting. For a–c, the intensity of the bands, determined with the ImageJ software, was shown as indicated. alpha -Tubulin, internal control. (d) Schematic diagram of RTKs (EGFR and VEGFR2)/NRP1/Ras/ERK and RTKs/NRP1/PI3K/AKT signalling cascade inhibitors targeting the indicated members of the pathways are shown. (e) EGFR/Ras/ERK signalling pathway was required for EBV infection. EGF-treated HNE1 and NPEC1-Bmi1 cells pre-incubated with Gefitinib (20 μM), Sorafenib (HNE1, 20 μM; NPEC1-Bmi1, 5 μM), U0126 (50 μM) and LY294002 (50 μM) for 30 min were infected with EBV for 2 h, and then cultured in the fresh medium for 48 h until FACS analysis for the infection efficiency. (f,g) Downregulation of EGFR and c-Met impaired EBV infection. HNE1 cells were transfected with siRNA duplexes targeting EGFR or c-Met for 48 h, followed by real-time PCR analysis for the expressions of EGFR and c-Met (f) or FACS analysis for EBV infection efficiency (g). (h) HRas partially rescued the inhibitory effect of Gefitinib on EBV infection. HNE1 cells transfected with pMSCV-HRAS-V12 plasmid for 24 h were pre-incubated with 20 μM Gefitinib for 30 min, followed by exposure to EBV. (i) HRas failed to rescue the suppressive effect of silencing NRP1 on EBV infection. After transfected with siNRP1 or siControl for 36 h, HNE1 cells were reseeded, and then transfected with the plasmid for HRAS-V12 or the empty vector for 24 h, followed by exposure to EBV. The infected cells were analysed by FACS, with controls set to 100%. For (e–i), data represent three times independent experiments. Graphs show mean±s.e.m. ***P<0.001; **P<0.01; *P<0.05; Student’s t-test. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/25670642), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Neuropilin-1 by Knockdown Validated
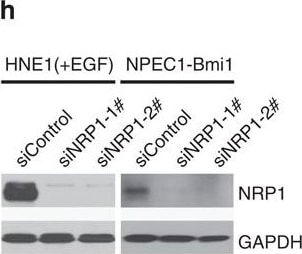
NRP1 enhances EBV infection, while NRP2 suppresses EBV infection.(a,b) Downregulation of NRP1 impaired, whereas knockdown of NRP2 promoted EBV infection. HNE1 cells were transfected with siRNA duplexes targeting NRP1 or NRP2 for 48 h, followed by NRP expression analysis by real-time PCR (a) or analysis for the efficiency of EBV infection (b); n=3. (c) EBV infection was blocked by soluble NRP1ABC, but enhanced by antibody against NRP2. For NRP1ABC protein-blocking experiment, HNE1 cells were infected with EBV, which was pre-incubated with purified NRP1ABC for 1 h. For antibody against NRP2-blocking experiment, HNE1 cells were pre-incubated with an anti-NRP2 antibody (100 μg ml−1) or goat IgG (control) at 4 °C for 1 h and then were exposed to EBV at an MOI of 5 × 103 for 3 h at 4 °C. (d,e) Overexpression of NRP1 enhanced EBV infection, while NRP2 suppressed EBV infection. HNE1 cells were transiently transfected with the expression plasmid for NRP1, NRP2 or the empty vector (pMSCV) for 24 h, followed by analysis for the expression of NRP1 and NRP2 by western blotting (d) or were exposed to EBV (e). (f,g) EGF upregulated NRP1 expression and enhanced EBV infection. HNE1 cells cultured with 10 ng ml−1 EGF for 24 h were analysed for the expression of NRP1 and NRP2 by western blotting (f) or were exposed to EBV (g). (h,i) EGF-enhanced EBV infection was partially dependent on NRP1. After transfected with siRNA against NRP1 for 48 h, EGF-treated HNE1 and NPEC-Bmi1 cells maintained in KSF medium supplemented with EGF were analysed for NRP1 expression by western blotting (h) or were exposed to EBV (i). For (b), (c), (e), (g) and (i), HNE1 or NPEC1-Bmi1 cells were exposed to EBV at an MOI of 2.5 × 103 for 2 h at 37 °C, unless otherwise indicated. The percentage of GFP-positive infected cells was analysed by FACS 48 h post infection, with controls (empty vector-transfected cells or vehicle treated-cells) set to 100%. Data represent three to five independent experiments. Values in all graphs are means±s.e.m. ***P<0.001; **P<0.01; *P<0.05; Student’s t-test. For (d), (f) and (h), GAPDH served as an internal control. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/25670642), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Neuropilin-1 by Western Blot
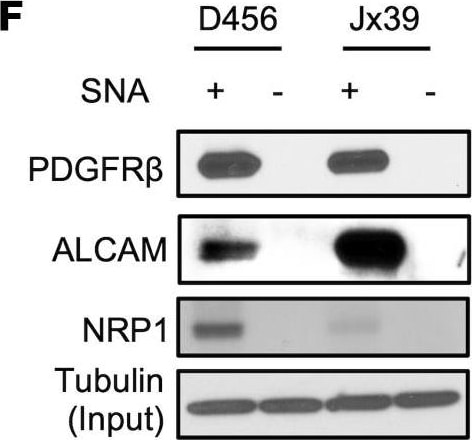
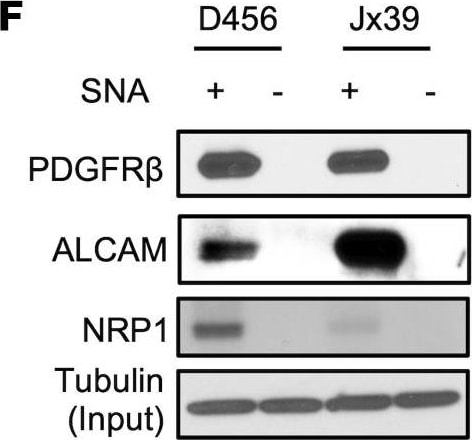
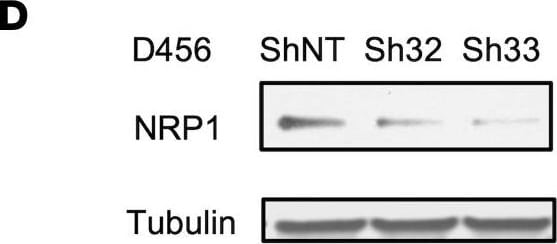
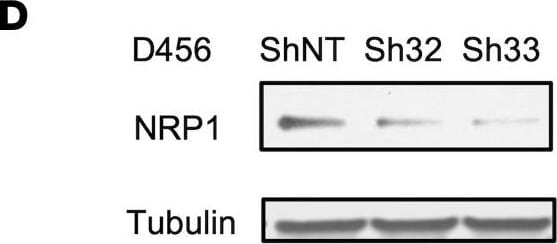
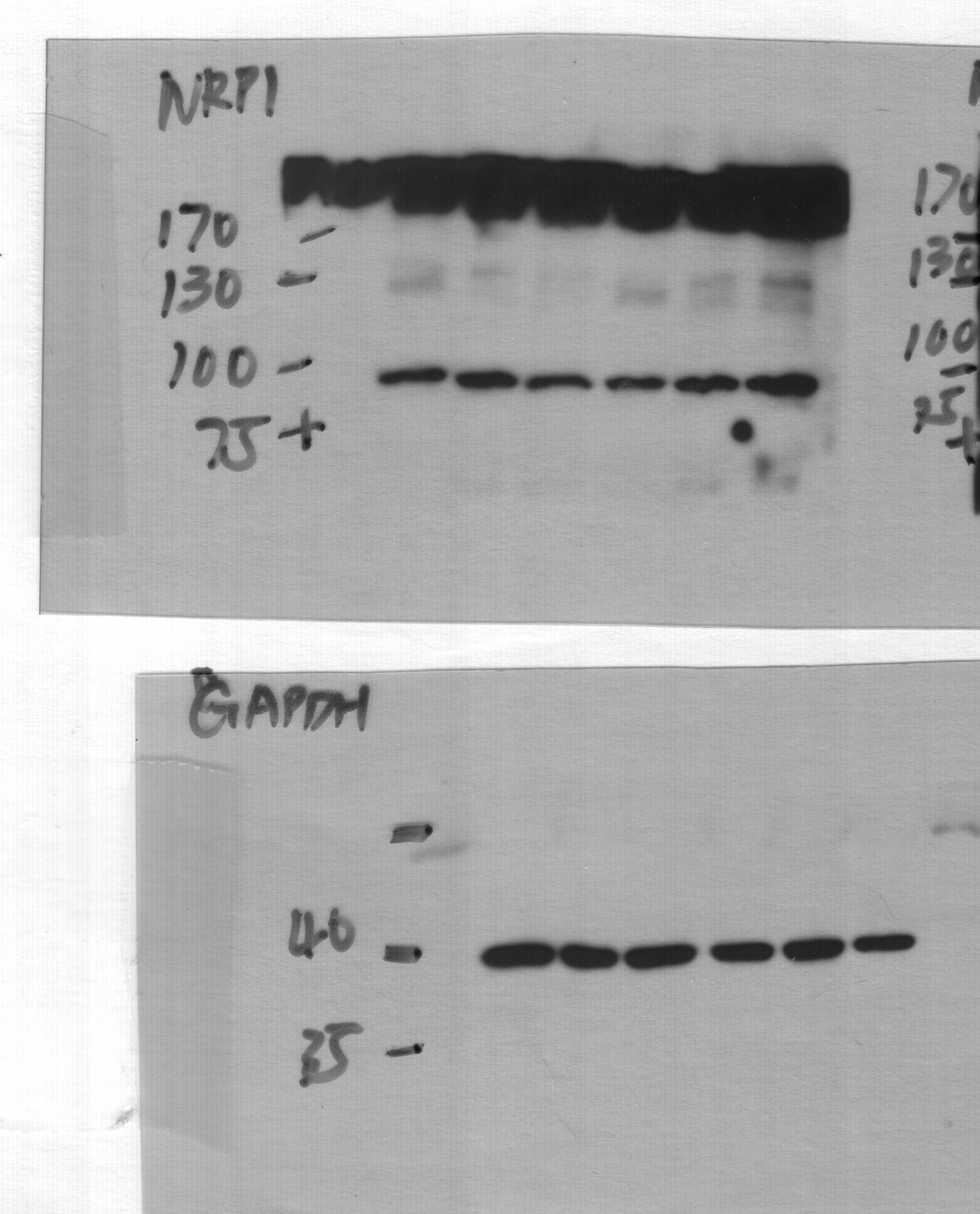
ST6GAL1 targeting decreases levels of a subset of N-glycoproteins that are known BTIC regulators.(A) Schematic of proteomic analysis of D456 BTICs with and without ST6GAL1 KD (n = 4 for each group of shNT, sh32, and sh33). IB with samples independent of the proteomic analysis verified that successful targeting ST6GAL1 resulted in decreased (B) PDGFRB, (C) ALCAM, and (D) NRP1 protein. (E) Schematic of pulldown using SNA-bound Agarose beads. (F) SNA pulldown and protein A/G bound agarose beads as a control demonstrated that PDGFRB, ALCAM, and NRP1 were targets for alpha 2,6 sialylation. (G) SNA pulldown of D456 PDX cells with ST6GAL1 KD compared with NT, illustrating differential pulldown of PDGFRB. (H) PDGF-BB–induced (10 minutes) activation of PDGFRB in D456 GBM PDX cells with ST6GAL1 KD compared with NT; IB for p-PDGFRB and total PDGFRB. The experiments were repeated in at least 3 independent biological replicates. Data from 1 representative experiment are shown. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36345944), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Neuropilin-1 by Western Blot
ST6GAL1 targeting decreases levels of a subset of N-glycoproteins that are known BTIC regulators.(A) Schematic of proteomic analysis of D456 BTICs with and without ST6GAL1 KD (n = 4 for each group of shNT, sh32, and sh33). IB with samples independent of the proteomic analysis verified that successful targeting ST6GAL1 resulted in decreased (B) PDGFRB, (C) ALCAM, and (D) NRP1 protein. (E) Schematic of pulldown using SNA-bound Agarose beads. (F) SNA pulldown and protein A/G bound agarose beads as a control demonstrated that PDGFRB, ALCAM, and NRP1 were targets for alpha 2,6 sialylation. (G) SNA pulldown of D456 PDX cells with ST6GAL1 KD compared with NT, illustrating differential pulldown of PDGFRB. (H) PDGF-BB–induced (10 minutes) activation of PDGFRB in D456 GBM PDX cells with ST6GAL1 KD compared with NT; IB for p-PDGFRB and total PDGFRB. The experiments were repeated in at least 3 independent biological replicates. Data from 1 representative experiment are shown. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36345944), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Neuropilin-1 by Western Blot
ST6GAL1 targeting decreases levels of a subset of N-glycoproteins that are known BTIC regulators.(A) Schematic of proteomic analysis of D456 BTICs with and without ST6GAL1 KD (n = 4 for each group of shNT, sh32, and sh33). IB with samples independent of the proteomic analysis verified that successful targeting ST6GAL1 resulted in decreased (B) PDGFRB, (C) ALCAM, and (D) NRP1 protein. (E) Schematic of pulldown using SNA-bound Agarose beads. (F) SNA pulldown and protein A/G bound agarose beads as a control demonstrated that PDGFRB, ALCAM, and NRP1 were targets for alpha 2,6 sialylation. (G) SNA pulldown of D456 PDX cells with ST6GAL1 KD compared with NT, illustrating differential pulldown of PDGFRB. (H) PDGF-BB–induced (10 minutes) activation of PDGFRB in D456 GBM PDX cells with ST6GAL1 KD compared with NT; IB for p-PDGFRB and total PDGFRB. The experiments were repeated in at least 3 independent biological replicates. Data from 1 representative experiment are shown. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36345944), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Neuropilin-1 by Western Blot
ST6GAL1 targeting decreases levels of a subset of N-glycoproteins that are known BTIC regulators.(A) Schematic of proteomic analysis of D456 BTICs with and without ST6GAL1 KD (n = 4 for each group of shNT, sh32, and sh33). IB with samples independent of the proteomic analysis verified that successful targeting ST6GAL1 resulted in decreased (B) PDGFRB, (C) ALCAM, and (D) NRP1 protein. (E) Schematic of pulldown using SNA-bound Agarose beads. (F) SNA pulldown and protein A/G bound agarose beads as a control demonstrated that PDGFRB, ALCAM, and NRP1 were targets for alpha 2,6 sialylation. (G) SNA pulldown of D456 PDX cells with ST6GAL1 KD compared with NT, illustrating differential pulldown of PDGFRB. (H) PDGF-BB–induced (10 minutes) activation of PDGFRB in D456 GBM PDX cells with ST6GAL1 KD compared with NT; IB for p-PDGFRB and total PDGFRB. The experiments were repeated in at least 3 independent biological replicates. Data from 1 representative experiment are shown. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36345944), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human Neuropilin‑1 Antibody
Blockade of Receptor-ligand Interaction
CyTOF-ready
Flow Cytometry
Sample: HUVEC human umbilical vein endothelial cells
Immunocytochemistry
Sample: Immersion fixed PC-3 human prostate cancer cell line and HLDM-2 human Hodgkin's lymphoma cell line
Immunohistochemistry
Sample: Immersion fixed paraffin-embedded sections of human ovarian and pancreatic cancer tissue
Western Blot
Sample: MDA‑MB‑231 human breast cancer cell line and human placenta tissue
Reviewed Applications
Read 5 reviews rated 3.8 using AF3870 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Neuropilin-1
References
- Bielenberg, D.R. et al. (2006) Exp. Cell Res. 312:584.
- Gu, C. et al. (2003) Dev. Cell 5:45.
- He, Z. and M. Tessier-Lavigne (1997) Cell 90:739.
- Soker, S. et al. (1998) Cell 92:735.
- Gagnon, M.L. et al. (2000) Proc. Natl. Acad. Sci. USA 97:2573.
- Cackowski, F.C. et al. (2004) Genomics 84:82.
- Rossignol, M. et al. (2000) Genomics 70:211.
- Tao, Q. et al. (2003) Angiogenesis 6:39.
- Gu, C. et al. (2002) J. Biol. Chem. 277:18069.
- Mamluk, R. et al. (2002) J. Biol. Chem. 277:24818.
- Yaron, A. et al. (2005) Neuron 45:513.
- Narazaki, M. and G. Tosato (2006) Blood 107:3892.
- Tordjman, R. et al. (2002) Nat. Immunol. 3477.
Alternate Names
Gene Symbol
UniProt
Additional Neuropilin-1 Products
Product Documents for Human Neuropilin‑1 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Neuropilin‑1 Antibody
This product or the use of this product is covered by U.S. Patents owned by The Regents of the University of California. This product is for research use only and is not to be used for commercial purposes. Use of this product to produce products for sale or for diagnostic, therapeutic or drug discovery purposes is prohibited. In order to obtain a license to use this product for such purposes, contact The Regents of the University of California.
U.S. Patent # 6,054,293, 6,623,738, and other U.S. and international patents pending.
For research use only
Related Research Areas
Citations for Human Neuropilin‑1 Antibody
Customer Reviews for Human Neuropilin‑1 Antibody (5)
Have you used Human Neuropilin‑1 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Application: Western BlotSample Tested: HEK293T human embryonic kidney cell lineSpecies: HumanVerified Customer | Posted 04/08/2022
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: HUVEC human umbilical vein endothelial cellsSpecies: HumanVerified Customer | Posted 07/16/2021
-
Application: Western BlotSample Tested: HUVEC human umbilical vein endothelial cellsSpecies: HumanVerified Customer | Posted 03/24/2021
-
Application: Western BlotSample Tested: HEK293T human embryonic kidney cell lineSpecies: HumanVerified Customer | Posted 12/18/2018
-
Verified Customer | Posted 04/21/2017
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Associated Pathways
 VEGF - VEGF R2 Signaling Pathways
VEGF - VEGF R2 Signaling Pathways