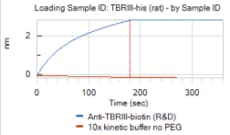
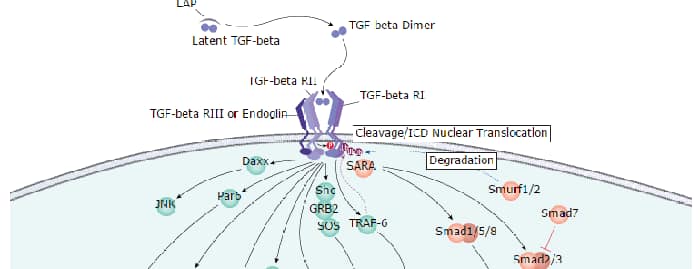
Most cell types express three sizes of receptors for TGF-beta. These are designated Type I (53 kDa), Type II (70-85 kDa), and Type III (250-350 kDa). The Type I receptor is a membrane-bound serine/threonine kinase that apparently requires the presence of the Type II receptor to bind TGF-beta. The Type II receptor is also a membrane-bound serine/threonine kinase that binds TGF-beta 1 and TGF-beta 3 with high affinity and TGF-beta 2 with a much lower affinity. The Type I and Type II receptors together form a heterodimeric signaling complex that is essential for the transduction of the anti-proliferative signals of TGF-beta.
The Type III receptor is a transmembrane proteoglycan with a large extracellular domain and a 43 amino acid residue cytoplasmic domain. The cytoplasmic domain of the Type III receptor lacks an obvious signaling motif and the receptor may not be involved directly in signal transduction. The Type III receptor binds TGF-beta 2 with the highest affinity. Other TGF-beta isoforms also bind the Type III receptor, but with lower affinities. Cellular responsiveness to TGF-beta 2 appears to be dependent on the presence of the Type III receptor which can interact with the signaling receptor complex. In addition to the transmembrane Type III receptor, a soluble form of the receptor is secreted by some cell types. The physiological role of this soluble receptor remains to be determined. The recombinant TGF-beta soluble receptor Type III binds the TGF-beta isoforms differentially in solution and exhibits TGF-beta antagonistic activities in vitro.