Beta IG-H3, also known as TGFBI and RGD-CAP, is a matricellular adaptor protein that is induced in most cell types in response to TGF-beta stimulation (1‑4). The mouse beta IG-H3 cDNA encodes a 683 amino acid (aa) precursor that includes a 23 aa signal sequence, one EMI domain, four FAS1 domains, and one RGD motif (2). Mouse beta IG-H3 shares 91% aa sequence identity with human and porcine beta IG-H3. beta IG-H3 is expressed as a 75 kDa protein with no post-translational additions (5). Following secretion, cleavages at multiple positions near the C-terminal end liberate peptides with pro-apoptotic activity (5, 6). Peptides that encompass the RGD motif contribute to the pro-apoptotic effects of TGF-beta (6). FAS1 domains contain YH motifs that are characterized by conserved Tyr and His residues (7). The YH motifs in each of the FAS1 domains enable beta IG-H3 to bind to matrix fibronectin, collagen I, collagen VI, biglycan, and decorin (3, 8‑11), in addition to cell expressed integrins alpha V/ beta 3, alpha V beta 5, and alpha 3 beta 1 (7, 8, 12, 13). The expression of beta IG-H3 is modulated at particular developmental stages in some cell types. It is upregulated in keratinocytes and immature dendritic cells but downregulated in osteoblasts (8, 12, 14). It promotes keratinocyte differentiation but blocks osteoblast differentiation (8, 12). beta IG-H3 stimulates macrophage endocytosis and vascular endothelial cell proliferation and migration (13, 14). High glucose levels induce beta IG-H3 in renal proximal tubule cells which is predictive of diabetic nephropathy (3). Several point mutations (clustered in the fourth FAS1 domain) of beta IG-H3 are linked to different corneal dystrophies (15). beta IG-H3 is downregulated in many cancers (4, 16) and functions as a suppressor of tumorigenicity when overexpressed (2, 4, 16).

Key Product Details
Species Reactivity
Mouse
Applications
Immunohistochemistry, Western Blot, ELISA Capture (Matched Antibody Pair)
Label
Unconjugated
Antibody Source
Polyclonal Sheep IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant mouse beta IG-H3
Gly24-His683
Accession # P82198
Gly24-His683
Accession # P82198
Specificity
Detects mouse beta IG-H3 in ELISAs and Western blots. In sandwich immunoassays, less than 6% cross-reactivity with recombinant human (rh) beta IG-H3 is observed.
Clonality
Polyclonal
Host
Sheep
Isotype
IgG
Scientific Data Images for Mouse beta IG‑H3 Antibody
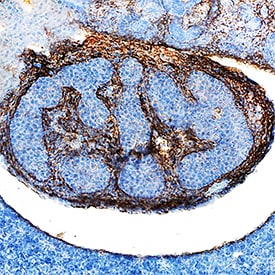
beta IG‑H3 in Mouse Embryo.
beta IG-H3 was detected in immersion fixed frozen sections of mouse embryo (13 d.p.c.) using Sheep Anti-Mouse beta IG-H3 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2559) at 5 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS008) and counterstained with hematoxylin (blue). Specific staining was localized to developing duodenum. View our protocol for Chromogenic IHC Staining of Frozen Tissue Sections.Applications for Mouse beta IG‑H3 Antibody
Application
Recommended Usage
Immunohistochemistry
5-15 µg/mL
Sample: Immersion fixed frozen sections of mouse embryo (13 d.p.c.)
Sample: Immersion fixed frozen sections of mouse embryo (13 d.p.c.)
Western Blot
0.1 µg/mL
Sample: Recombinant Mouse beta IG-H3 (Catalog # 2559-BG)
Sample: Recombinant Mouse beta IG-H3 (Catalog # 2559-BG)
Mouse beta IG-H3 Sandwich Immunoassay
Please Note: Optimal dilutions of this antibody should be experimentally determined.
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: beta IG-H3
References
- Skonier, J. et al. (1992) DNA Cell Biol. 11:511.
- Skonier, J. et al. (1994) DNA Cell Biol. 13:571.
- Lee, S-H. et al. (2003) Kidney Int. 64:1012.
- Zhao, Y.L. et al. (2002) Oncogene 21:7471.
- Andersen, R.B. et al. (2004) Biochemistry 43:16374.
- Kim, J-E. et al. (2003) Oncogene 22:2045.
- Kim, J-E. et al. (2002) J. Biol. Chem. 277:46159.
- Thapa, N. et al. (2005) Bone 36:232.
- Hanssen, E. et al. (2003) J. Biol. Chem. 278:24334.
- Billings, P.C. et al. (2002) J. Biol. Chem. 277:28003.
- Reinboth, B. et al. (2006) J. Biol. Chem. 281:7816.
- Oh, J-E. et al. (2005) J. Biol. Chem. 280:21629.
- Nam, J-O. et al. (2003) J. Biol. Chem. 278:25902.
- Cao, W. et al. (2006) Blood 107:2777.
- Stewart, H.S. et al. (1999) Hum. Mutat. 14:126.
- Zhao, Y. et al. (2006) Mol. Carcinog. 45:84.
Long Name
TGF-beta Induced Gene H3
Alternate Names
beta IGH3, Kerato-epithelin, RGD-CAP, TGFBI, TGFBIP
Gene Symbol
TGFBI
UniProt
Additional beta IG-H3 Products
Product Documents for Mouse beta IG‑H3 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse beta IG‑H3 Antibody
For research use only
Related Research Areas
Customer Reviews for Mouse beta IG‑H3 Antibody
There are currently no reviews for this product. Be the first to review Mouse beta IG‑H3 Antibody and earn rewards!
Have you used Mouse beta IG‑H3 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
