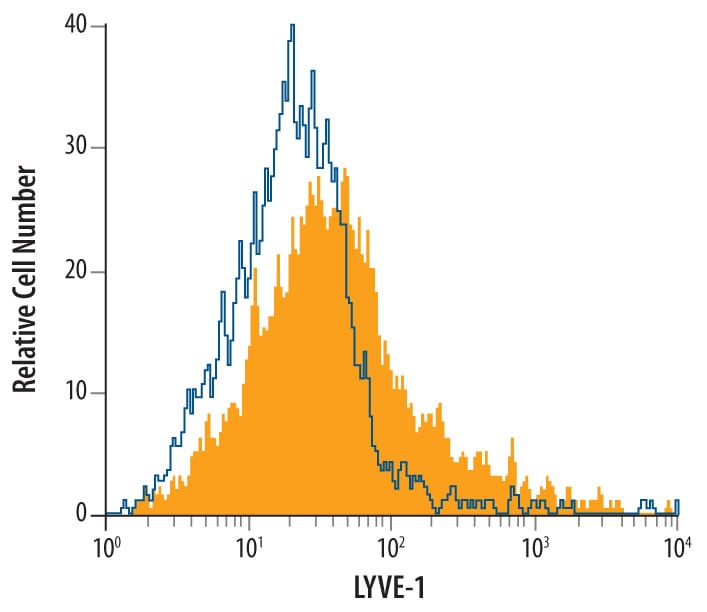
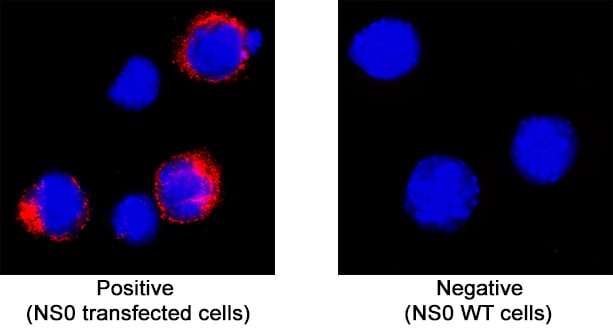
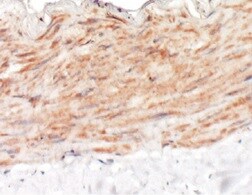
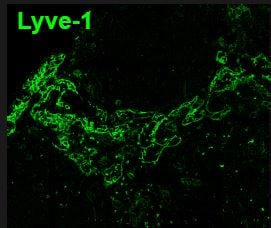
Lymphatic vessel endothelial hyaluronan (HA) receptor-1 (LYVE-1) is a recently identified receptor of HA, a linear high molecular weight polymer composed of alternating units of D-glucuronic acid and N-acetyl-D-glucosamine. HA is found in the extracellular matrix of most animal tissues and in body fluids. It modulates cell behavior and functions during tissue remodeling, development, homeostasis, and disease (1). The turnover of HA (several grams/day in humans) occurs primarily in the lymphatics and liver, the two major clearance systems that catabolize approximately 85% and 15% of HA, respectively (1-3). LYVE-1 shares 41% homology with the other known HA receptor, CD44 (4). The homology between the two proteins increases to 61% within the HA binding domain. The HA binding domain, known as the link module, is a common structural motif found in other HA binding proteins such as link protein, aggrecan and versican (1, 5). Human and mouse LYVE-1 share 69% amino acid sequence identity.
LYVE-1 is primarily expressed on both the luminal and abluminal surfaces of lymphatic vessels (4, 5). In addition, LYVE-1 is also present in normal hepatic blood sinusoidal endothelial cells (6). LYVE-1 mediates the endocytosis of HA and may transport HA from tissue to lymph by transcytosis, delivering HA to lymphatic capillaries for removal and degradation in the regional lymph nodes (5, 7, 8). Because of its restricted expression patterns, LYVE-1, along with other lymphatic proteins such as VEGF R3, podoplanin and the homeobox protein propero-related (Prox-1), constitute a set of markers useful for distinguishing between lymphatic and blood microvasculature (4, 5, 9-11).