Mouse Neuropilin‑1 Antibody
R&D Systems | Catalog # MAB59941

Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Phe21-Pro856
Accession # P97333
Specificity
Clonality
Host
Isotype
Scientific Data Images for Mouse Neuropilin‑1 Antibody
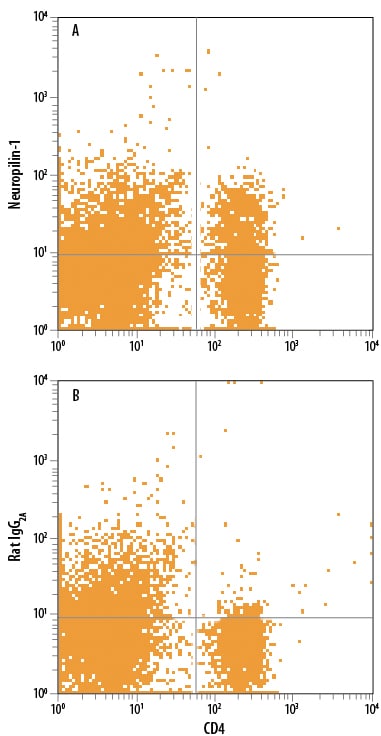
Detection of Neuropilin‑1 in Mouse Splenocytes by Flow Cytometry.
Mouse splenocytes were stained with Rat Anti-Mouse CD4 APC-conjugated Monoclonal Antibody (Catalog # FAB554A) and either (A) Rat Anti-Mouse Neuropilin-1 Monoclonal Antibody (Catalog # MAB59941) or (B) Rat IgG2AIsotype Control (Catalog # MAB006) followed by Phycoerythrin-conjugated Anti-Rat IgG Secondary Antibody (Catalog # F0105B).Applications for Mouse Neuropilin‑1 Antibody
CyTOF-ready
Flow Cytometry
Sample: Mouse splenocytes
Neutralization
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Reconstitution
Sterile PBS to a final concentration of 0.5 mg/mL. For liquid material, refer to CoA for concentration.
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Neuropilin-1
References
- Bielenberg, D.R. et al. (2006) Exp. Cell Res. 312:584.
- Gu, C. et al. (2003) Dev. Cell 5:45.
- He, Z. and M. Tessier-Lavigne (1997) Cell 90:739.
- Soker, S. et al. (1998) Cell 92:735.
- Entrez accession #EDL11827
- Gagnon, M.L. et al. (2000) Proc. Natl. Acad. Sci. USA 97:2573.
- Cackowski, F.C. et al. (2004) Genomics 84:82.
- Rossignol, M. et al. (2000) Genomics 70:211.
- Gu, C. et al. (2002) J. Biol. Chem. 277:18069.
- Mamluk, R. et al. (2002) J. Biol. Chem. 277:24818.
- Yaron, A. et al. (2005) Neuron 45:513.
- Narazaki, M. and G. Tosato (2006) Blood 107:3892.
- Tordjman, R. et al. (2002) Nat. Immunol. 3477.
Alternate Names
Gene Symbol
UniProt
Additional Neuropilin-1 Products
Product Documents for Mouse Neuropilin‑1 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse Neuropilin‑1 Antibody
For research use only
Related Research Areas
Citations for Mouse Neuropilin‑1 Antibody
Customer Reviews for Mouse Neuropilin‑1 Antibody
There are currently no reviews for this product. Be the first to review Mouse Neuropilin‑1 Antibody and earn rewards!
Have you used Mouse Neuropilin‑1 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Associated Pathways
 VEGF - VEGF R2 Signaling Pathways
VEGF - VEGF R2 Signaling Pathways


