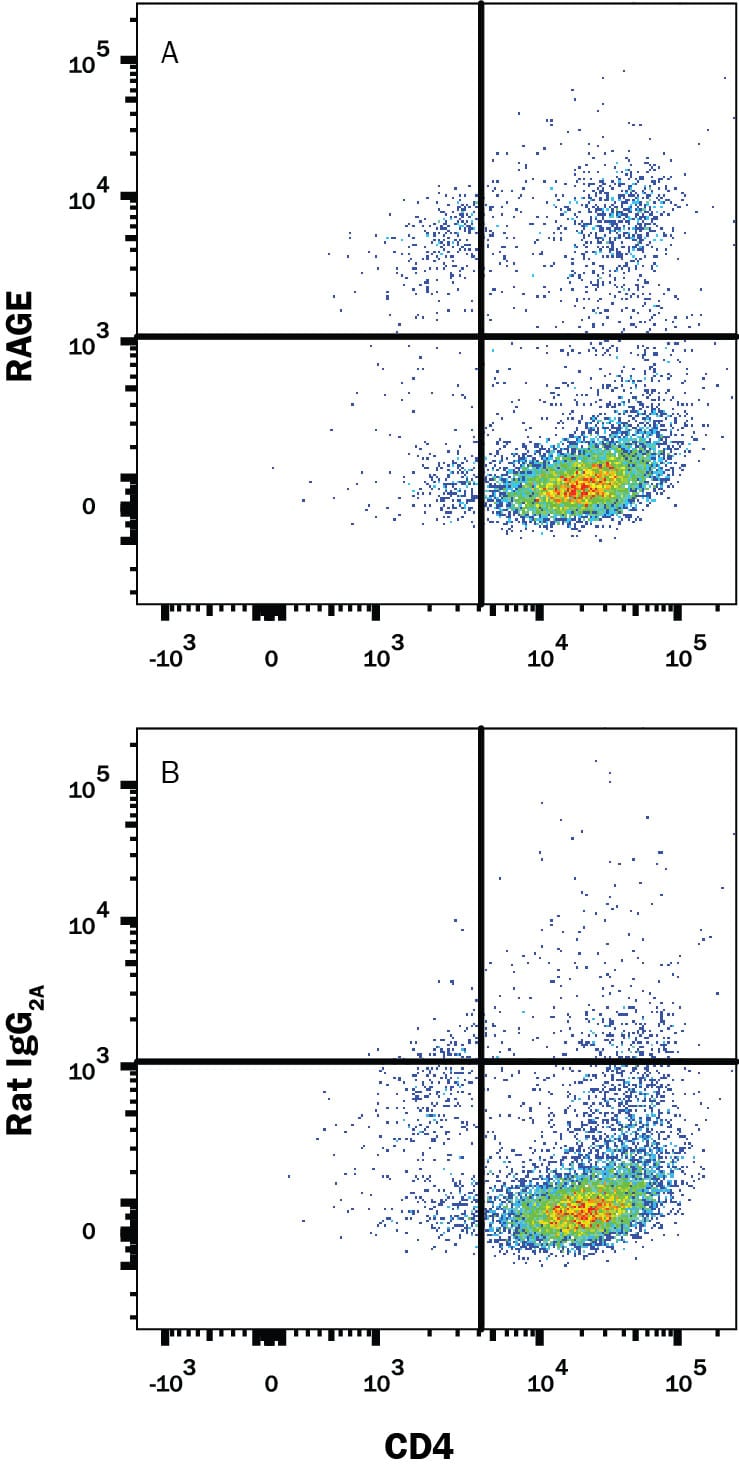
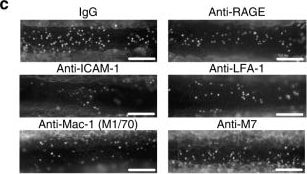
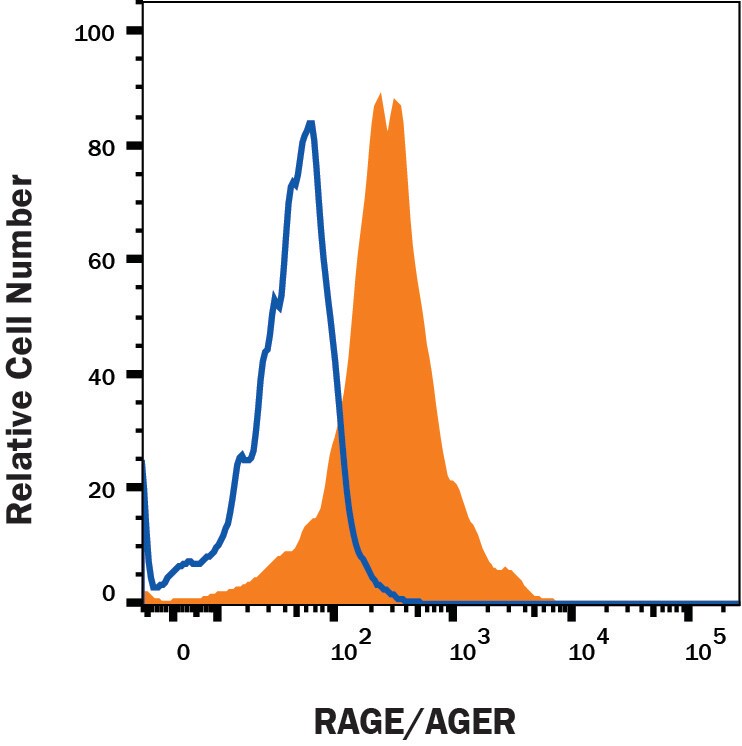
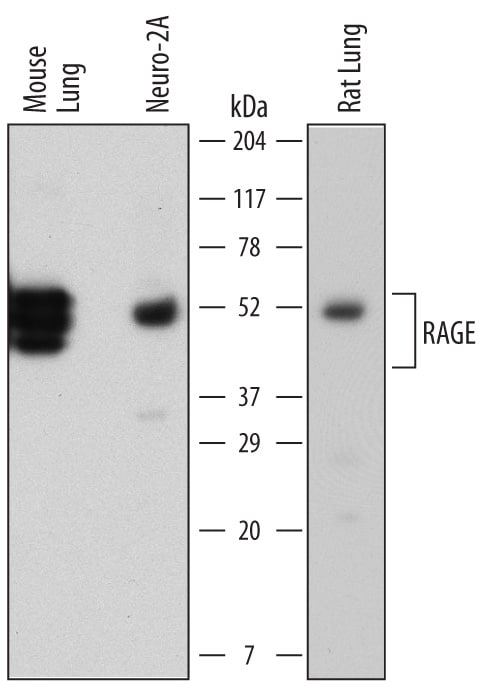
Advanced glycation endproducts (AGE) are adducts formed by the non-enzymatic glycation or oxidation of macromolecules (1). AGE forms during aging and its formation is accelerated under pathophysiologic states such as diabetes, Alzheimer’s disease, renal failure and immune/inflammatory disorders. Receptor for Advanced Glycation Endoproducts (RAGE), named for its ability to bind AGE, is a multiligand receptor belonging the immunoglobulin (Ig) superfamily. Besides AGE, RAGE binds amyloid beta -peptide, S100/calgranulin family proteins, high mobility group B1 (HMGB1, also know as amphoterin) and leukocyte integrins (1, 2).
The mouse RAGE gene encodes a 403 amino acid (aa) residue type I transmembrane glycoprotein with a 22 aa signal peptide, a 319 aa extracellular domain containing a Ig-like V-type domain and two Ig-like Ce-type domains, a 21 aa transmembrane domain and a 41 aa cytoplasmic domain (3). The V-type domain and the cytoplasmic domain are important for ligand binding and for intracellular signaling, respectively. Two alternative splice variants, lacking the V-type domain or the cytoplasmic tail, are known (1, 4). RAGE is highly expressed in the embryonic central nervous system (5). In adult tissues, RAGE is expressed at low levels in multiple tissues including endothelial and smooth muscle cells, mononuclear phagocytes, pericytes, microglia, neurons, cardiac myocytes and hepatocytes (6). The expression of RAGE is upregulated upon ligand interaction. Depending on the cellular context and interacting ligand, RAGE activation can trigger differential signaling pathways that affect divergent pathways of gene expression (1, 7). RAGE activation modulates varied essential cellular responses (including inflammation, immunity, proliferation, cellular adhesion and migration) that contribute to cellular dysfunction associated with chronic diseases such as diabetes, cancer, amyloidoses and immune or inflammatory disorders (1).