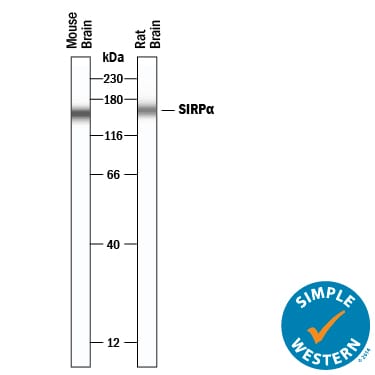
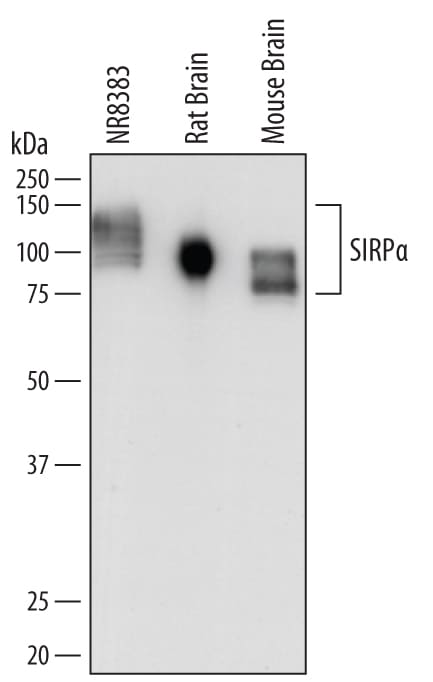
SIRP alpha (Signal regulatory protein alpha; also CD172a, Shps1 and Bit) is a variably glycosylated, 85-120 kDa member of the SIRP ‘family' of proteins. It is expressed on neurons, macrophages, monocytes, granulocytes and dendritic cells. SIRP alpha is phosphorylated/activated in response to cell adhesion. This may, or may not, involve binding to known ligands CD47, SP-A and SP-D. Following phosphorylation, SIRP alpha binds to SHP-1 and -2, resulting in the negative regulation of immune system activity. Mature rat SIRP alpha is a 478 amino acid (aa) type I transmembrane glycoprotein. It contains a 342 aa extracellular region (aa 32-373) that possesses one V-type and two C1-type Ig-like domains. Its cytoplasmic domain possesses two ITIMs that interact with protein phosphatases. There is one potential splice variant that shows a four aa insertion after Gln424. Over aa 32-373, rat SIRP alpha shares 63% and 73% aa sequence identity with human and mouse SIRP alpha, respectively.