Mouse Relaxin-1 (RLN1 or M1 relaxin) is the counterpart of human relaxin-2 (H2 relaxin) within the structurally related insulin/relaxin superfamily, while orthologs of the human relaxin-1 are found only in higher primates (1, 2). As with other insulin/relaxin superfamily members, RLN1 is synthesized as a preprohormone (1‑3, 5). Processing of the 21 kDa preprorelaxin-1 and 16-17 kDa prorelaxin-1 includes removal of the signal sequence, formation of two disulfide bonds between A and B chains and removal of the intervening C‑chain by a prohormone convertase. The resulting mature protein is an unglycosylated 6 kDa dimer of disulfide-linked A and B chains that binds the leucine-rich G-protein coupled receptor RXFP1, previously called LGR7 (1‑4). Mouse RLN1 shares only 67%, 39%, 36% and 42% amino acid (aa) identity with rat, equine and feline RLN1 and human Relaxin-2, respectively, and its activity shows partial species specificity. For example, a unique amino acid near the end of the A chain in mice, Tyr184, lowers its affinity for RXFP1 compared to other species (5). Mouse RLN1 is prominently expressed in the prostate and ovary, with lower levels in the brain, heart and other organs (1‑3). In the prostate, RLN1 is anti-apoptotic and contributes to development and maintenance of male fertility (6). In the female mouse, circulating RLN1 produced by the corpus luteum during pregnancy is essential for growth and softening of the cervix and vagina in preparation for delivery. It also promotes development of the mouse mammary apparatus, regulates plasma osmolality, and increases cardiac output and glomerular filtration rate in pregnancy (1, 2). Many RLN1 effects on reproductive tissues are augmented by estrogen (1‑3, 7). In non‑reproductive tissues, RLN1 mediates collagen turnover (7). RLN1-deficient mice develop age‑related fibrosis and smooth muscle hypertrophy in organs such as lung, heart, kidney and liver (7‑10).
Mouse Relaxin‑1 Antibody
R&D Systems | Catalog # AF6637


Key Product Details
Species Reactivity
Mouse
Applications
Immunohistochemistry, Western Blot
Label
Unconjugated
Antibody Source
Polyclonal Sheep IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant mouse Relaxin-1
Arg23-Arg54 with an N-terminal Met (B chain) & Glu161-Cys185 (A chain)
Accession # NP_035402
Arg23-Arg54 with an N-terminal Met (B chain) & Glu161-Cys185 (A chain)
Accession # NP_035402
Specificity
Detects mouse Relaxin-1 in direct ELISAs and Western blots. In direct ELISAs, less than 1% cross-reactivity with recombinant human Relaxin-1 is observed.
Clonality
Polyclonal
Host
Sheep
Isotype
IgG
Scientific Data Images for Mouse Relaxin‑1 Antibody
Detection of Mouse Relaxin‑1 by Western Blot.
Western blot shows lysates of mouse testis tissue. PVDF membrane was probed with 1 µg/mL of Sheep Anti-Mouse Relaxin-1 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF6637) followed by HRP-conjugated Anti-Sheep IgG Secondary Antibody (Catalog # HAF016). A specific band was detected for Prorelaxin-1 at approximately 16-17 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Relaxin‑1 in Mouse Testis.
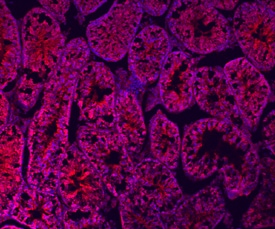
Relaxin-1 was detected in immersion fixed frozen sections of adult mouse testis using Sheep Anti-Mouse Relaxin-1 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF6637) at 10 µg/mL overnight at 4 °C. Tissue was stained using the NorthernLights™ 557-conjugated Anti-Sheep IgG Secondary Antibody (red, upper panel; Catalog # NL010) and counterstained with DAPI (blue, lower panel). Specific staining was localized to the cytoplasm of spermatocytes. View our protocol for Fluorescent IHC Staining of Frozen Tissue Sections.Applications for Mouse Relaxin‑1 Antibody
Application
Recommended Usage
Immunohistochemistry
5-15 µg/mL
Sample: Immersion fixed frozen sections of adult mouse testis
Sample: Immersion fixed frozen sections of adult mouse testis
Western Blot
1 µg/mL
Sample: Mouse testis tissue
Sample: Mouse testis tissue
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Sterile PBS to a final concentration of 0.2 mg/mL. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Relaxin-1
References
- Kong, R.C.K. et al. (2010) Mol. Cell. Endocrinol. 320:1.
- Sherwood, O.D. (2004) Endocr. Rev. 25:205.
- Callander, G.E. and R.A.D. Bathgate (2010) Cell. Mol. Life Sci. 67:2327.
- Novak, J. et al. (2006) FASEB J. 20:2352.
- Schwabe, C. and E. E. Bullesbach (1994) FASEB J. 8:1152.
- Samuel, C.S. et al. (2003) Lab. Invest. 83:1055.
- Lekgabe, E.D. et al. (2006) Endocrinology 147:5575.
- Samuel, C.S. et al. (2003) FASEB J. 17:121.
- Bennett, R.G. (2009) Transl. Res. 154:1.
- Hewitson, T.D. et al. (2007) Endocrinology 148:660.
Alternate Names
H1, Relaxin1, RLN1, RLXH1
Gene Symbol
RLN1
UniProt
Additional Relaxin-1 Products
Product Documents for Mouse Relaxin‑1 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse Relaxin‑1 Antibody
For research use only
Related Research Areas
Citations for Mouse Relaxin‑1 Antibody
Customer Reviews for Mouse Relaxin‑1 Antibody
There are currently no reviews for this product. Be the first to review Mouse Relaxin‑1 Antibody and earn rewards!
Have you used Mouse Relaxin‑1 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...