Recombinant Human Active ERK1 Protein, CF
R&D Systems | Catalog # 1879-KS

Loading...
Key Product Details
- R&D Systems E. coli-derived Recombinant Human Active ERK1 Protein (1879-KS)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
E. coli
Accession Number
Applications
Bioactivity
Loading...
Product Specifications
Source
E. coli-derived human ERK1 protein
Purity
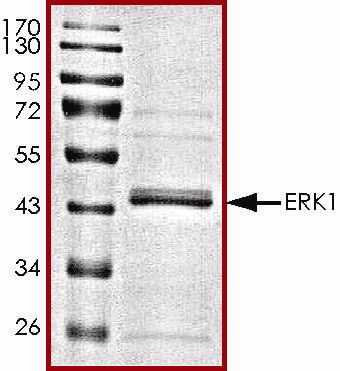
>80%, by SDS-PAGE under reducing conditions and visualized by Colloidal Coomassie® Blue stain at 5 μg per lane.
N-terminal Sequence Analysis
Activated by active MEK1 in vitro
Predicted Molecular Mass
44 kDa
Activity
The specific activity of ERK1 was determined to be 42 nmol/min/mg using a myelin basic protein (MBP) substrate and was equivalent to 382 nmol/min/mg as per radiometric assay.
Scientific Data Images for Recombinant Human Active ERK1 Protein, CF
Recombinant Human Active ERK1 Protein SDS-PAGE.
The approximate molecular weight is 44 kDa and the purity is > 80%.Formulation, Preparation, and Storage
1879-KS
| Formulation | Supplied in 50 mM Tris-HCl, pH 7.5, 150 mM NaCl, 0.25 mM DTT, 0.1 mM PMSF and 25% glycerol. |
| Shipping | The product is shipped with dry ice or equivalent. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | This product is stable at ≤ ‑70°C for up to 1 year from the date of receipt. For optimal storage, aliquot into smaller quantities after centrifugation and store at recommended temperature. Avoid repeated freeze-thaw cycles. |
Background: ERK1
References
- Boulton, T.G. et al. (1991) Biochemistry 30:278.
Long Name
Extracellular Signal-regulated Kinase 1
Alternate Names
MAPK3, P44ERK1, p44mapk, PRKM3
Gene Symbol
MAPK3
UniProt
Additional ERK1 Products
Product Documents for Recombinant Human Active ERK1 Protein, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Human Active ERK1 Protein, CF
For research use only
Citations for Recombinant Human Active ERK1 Protein, CF
Customer Reviews for Recombinant Human Active ERK1 Protein, CF
There are currently no reviews for this product. Be the first to review Recombinant Human Active ERK1 Protein, CF and earn rewards!
Have you used Recombinant Human Active ERK1 Protein, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
View specific protocols for Recombinant Human Active ERK1 Protein, CF (1879-KS):
Materials
- Active Kinase - Active ERK1 (0.1 μg/μL) diluted with Kinase Dilution Buffer and assayed as outlined in Sample Activity Plot. Note: These are suggested working dilutions. Optimal dilutions should be determined by each laboratory for each application.
- Kinase Assay Buffer III - 200 mM Tris-HCl, pH 7.4, 100 mM MgCl2, and 0.5 mg/mL BSA. Add fresh DTT prior to use to a final concentration of 250 μM.
- Kinase Dilution Buffer IX (1x) - Kinase Assay Buffer I diluted at a 1:4 ratio (5-fold dilution) with cold distilled water. Add fresh DTT prior to use to a final concentration of 50 μM.
- ADP-GloTM Kinase Assay Kit - 10 mM ATP Solution, 10 mM ADP Solution, ADP-GloTM Reagent, Kinase Detection Reagent.
- Substrate - Myeline basic protein (MBP) diluted in 100 mM MOPS buffer (pH 6.5) to a final concentration of 0.5 mg/mL.
- Thaw the Active ERK1, Kinase Assay Buffer III (5x), and Substrate on ice. Prepare a 15 μL enzyme dilution using Kinase Dilution Buffer IX (1x), at the desired concentration, in a pre-chilled 96-well plate.
- Prepare a substrate/ATP mixture as follows (25 μM ATP example)
a. 10 mM ATP Solution: 1 μL
b. Kinase Assay Buffer III (5x): 79 μL
c. Substrate at 0.5 mg/mL: 80 μL - Transfer the following reaction components prepared in Step 1 and 2 to a 384-well opaque plate bringing the reaction volume up to 5 μL:
a. 3 μL of diluted Active ERK1
b. 2 μL of Substrate/ATP mix as prepared in the Step 2. This initiates the reaction. - Set up the blank control as outlined in step 2, excluding the addition of the kinase. Replace the kinase with an equal volume of Kinase Dilution Buffer IX (1x).
- Incubate at ambient temperature for 40 minutes.
- After the 40 minute incubation period, terminate the reaction and deplete the remaining ATP by adding 5 μL of ADP-Glo Reagent. Spin down and shake the 384-well plate. Then incubate the reaction mixture for another 40 minutes at ambient temperature.
- Add 10 μL of the Kinase Detection Reagent to the 384-well plate and incubate the reaction mixture for another 30 minutes at ambient temperature
- Read the 384-well reaction plate using the Luminescence Module Protocol on a GloMax®-Multi Microplate Multimode Reader.
- Determine the corrected activity (RLU) by removing the blank control value (see step 4) for each sample and calculate the kinase specific activity as outlined below.
Calculation of Specific Activity of ADP (RLU/pmol)
From ATP-ADP conversion curve, determine RLU/pmol of ADP
Kinase Specific Activity (SA) (pmol/minutes/μg or nmol/minutes/mg)
Corrected RLU from reaction / [(SA of ADP in RLU/pmol) x (Reaction time in minutes) x (Enzyme amount in μg or mg)]
FAQs for Recombinant Human Active ERK1 Protein, CF
Showing
1
-
1 of
1 FAQ
Showing All
-
Q: What is the activation protocol for this enzyme?
A: This enzyme is supplied in its activated form, so no activation step is required.
Loading...
Associated Pathways
 Common Cytokine Receptor Gamma-Chain Family Signaling Pathways
Common Cytokine Receptor Gamma-Chain Family Signaling Pathways
 HIF Enhancer Pathways
HIF Enhancer Pathways
 IL-1 Family Signaling Pathways
IL-1 Family Signaling Pathways
 IL-2 Signaling Pathways
IL-2 Signaling Pathways
IL-4 Signaling Pathways
 IL-7 Signaling Pathways
IL-7 Signaling Pathways
 IL-9 Signaling Pathways
IL-9 Signaling Pathways
 IL-15 Signaling Pathways
IL-15 Signaling Pathways
 IL-17 Family Signaling Pathways
IL-17 Family Signaling Pathways
 IL-21 Signaling Pathways
IL-21 Signaling Pathways
 MAPK Signaling: Oxidative Stress Pathway
MAPK Signaling: Oxidative Stress Pathway
 MAPK Signaling Pathway: Mitogen Stimulation Pathway
MAPK Signaling Pathway: Mitogen Stimulation Pathway
 mTOR Signaling Pathway
mTOR Signaling Pathway
 NOD-like Receptor Signaling Pathways
NOD-like Receptor Signaling Pathways
 Pathogen or Damage-activated C-Type Lectin Receptor Signaling Pathways
Pathogen or Damage-activated C-Type Lectin Receptor Signaling Pathways
 TGF-beta Signaling Pathways
TGF-beta Signaling Pathways
 VEGF - VEGF R2 Signaling Pathways
VEGF - VEGF R2 Signaling Pathways

 IL-7 Signaling Pathways
IL-7 Signaling Pathways
 IL-9 Signaling Pathways
IL-9 Signaling Pathways
 IL-15 Signaling Pathways
IL-15 Signaling Pathways
 IL-17 Family Signaling Pathways
IL-17 Family Signaling Pathways
 IL-21 Signaling Pathways
IL-21 Signaling Pathways
 MAPK Signaling: Oxidative Stress Pathway
MAPK Signaling: Oxidative Stress Pathway
 MAPK Signaling Pathway: Mitogen Stimulation Pathway
MAPK Signaling Pathway: Mitogen Stimulation Pathway
 mTOR Signaling Pathway
mTOR Signaling Pathway
 NOD-like Receptor Signaling Pathways
NOD-like Receptor Signaling Pathways
 Pathogen or Damage-activated C-Type Lectin Receptor Signaling Pathways
Pathogen or Damage-activated C-Type Lectin Receptor Signaling Pathways
 TGF-beta Signaling Pathways
TGF-beta Signaling Pathways
 VEGF - VEGF R2 Signaling Pathways
VEGF - VEGF R2 Signaling Pathways

