Recombinant Human DPPIV/CD26 (High Purity Dimer) Protein, CF
R&D Systems | Catalog # 9168-SE

Key Product Details
Source
Accession Number
Structure / Form
Applications
Product Specifications
Source
Asn29-Pro766, with a C-terminal Asp-Ile and 6-His tag
Purity
Endotoxin Level
N-terminal Sequence Analysis
Predicted Molecular Mass
SDS-PAGE
Activity
The specific activity is >8500 pmol/min/µg as measured under the described conditions.
Scientific Data Images for Recombinant Human DPPIV/CD26 (High Purity Dimer) Protein, CF
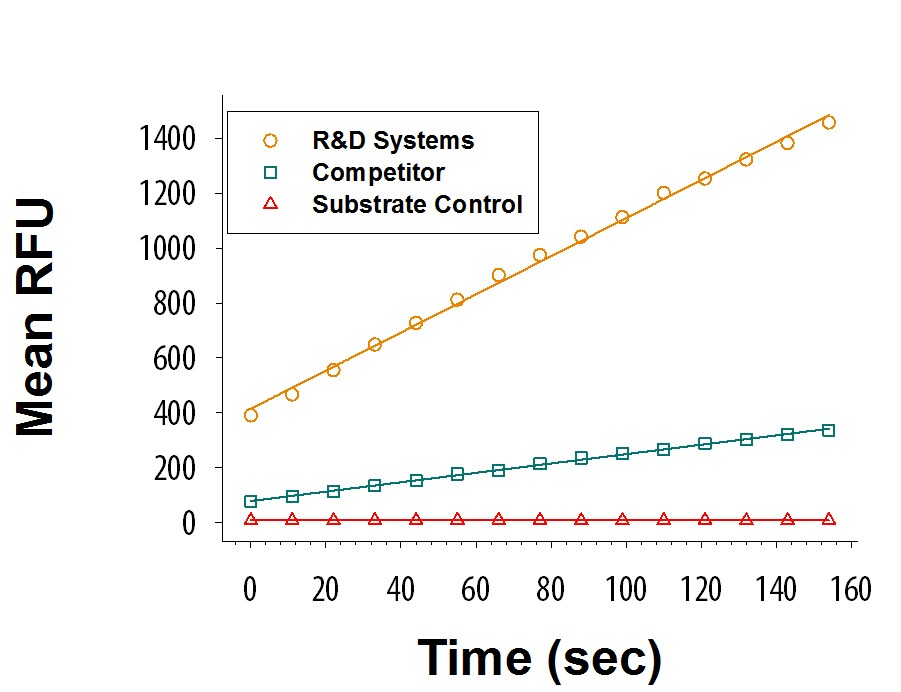
Recombinant Human DPPIV/CD26 (High Purity Dimer) Protein Enzyme Activity
Recombinant Human DPPIV/CD26 (Catalog # 9168-SE) is measured by its ability to cleave the fluorogenic peptide substrate, Gly-Pro-7-amido-4-methylcoumarin (GP-AMC). The activity (orange) is approximately 4-fold greater than the competitor's DPPIV/CD26 (green).Formulation, Preparation, and Storage
9168-SE
| Formulation | Supplied as a 0.2 μm filtered solution in MES and NaCl. |
| Shipping | The product is shipped with dry ice or equivalent. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Background: DPPIV/CD26
References
- Klemann, C. et al. (2016) Clin. Exp. Immunol. Epub PMID 26919392.
- Mortier, A. et al. (2016) J. Leukoc. Biol. Epub PMID 26744452.
- Tanaka, T. et al. (1992) J. Immunol. 149:481.
- Rohrborn, D. et al. (2014) FEBS Lett. 588:3870.
- Waumans, Y. et al. (2015) Front. Immunol. 6:387.
- Lamers, D. et al. (2011) Diabetes 60:1917.
- Proost, P. et al. (1998) J. Biol. Chem. 273:7222.
- Proost, P. et al. (2001) Blood 98:3554.
- Ohtsuki, T. et al. (1998) FEBS Lett. 431:236.
- Barreira da Silva, R. et al. (2015) Nat. Immunol. 16:850.
- Guan, E. et al. (2002) J. Biol. Chem. 277:32348.
- Callebaut, C. et al. (1993) Science 262:2045.
- Raj, V.S. et al. (2013) Nature 495:251.
- Broxmeyer, H.E. et al. (2012) Nat. Med. 18:1786.
- Christopherson II, K.W. et al. (2004) Science 305:1000.
- Kameoka, J. et al. (1993) Science 261:466.
- Ohnuma, K. et al. (2007) J. Biol. Chem. 282:10117.
- Hatano, R. et al. (2013) Immunology 138:165.
Long Name
Alternate Names
Gene Symbol
UniProt
Additional DPPIV/CD26 Products
Product Documents for Recombinant Human DPPIV/CD26 (High Purity Dimer) Protein, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Human DPPIV/CD26 (High Purity Dimer) Protein, CF
For research use only
Citations for Recombinant Human DPPIV/CD26 (High Purity Dimer) Protein, CF
Customer Reviews for Recombinant Human DPPIV/CD26 (High Purity Dimer) Protein, CF
There are currently no reviews for this product. Be the first to review Recombinant Human DPPIV/CD26 (High Purity Dimer) Protein, CF and earn rewards!
Have you used Recombinant Human DPPIV/CD26 (High Purity Dimer) Protein, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
View specific protocols for Recombinant Human DPPIV/CD26 (High Purity Dimer) Protein, CF (9168-SE):
- Assay Buffer: 25 mM Tris, pH 8.0
- Recombinant Human DPPIV/CD26 (rhDPPIV) (Catalog # 9168-SE)
- Substrate: H-Gly-Pro-AMC (Bachem, Catalog # I-1225), 10 mM stock in DMSO
- F16 Black Maxisorp Plate (Nunc, Catalog # 475515)
- Fluorescent Plate Reader (Model: SpectraMax Gemini EM by Molecular Devices) or equivalent
- Dilute rhDPPIV to 0.1 ng/μL in Assay Buffer.
- Dilute Substrate to 200 μM in Assay Buffer.
- Load into a black plate 50 μL of 0.1 ng/μL rhDPPIV, and start the reaction by adding 50 μL of 200 μM Substrate. As a Substrate Blank combine 50 μL of Assay Buffer and 50 μL of 200 μM Substrate.
- Read at excitation and emission wavelengths of 380 nm and 460 nm, respectively, in kinetic mode for 5 minutes.
- Calculate specific activity:
Specific Activity (pmol/min/µg) = | Adjusted Vmax* (RFU/min) x Conversion Factor** (pmol/RFU) |
| amount of enzyme (µg) |
*Adjusted for Substrate Blank.
**Derived using calibration standard 7-amino, 4-Methyl Coumarin (Sigma, Catalog # A9891).
- rhDPPIV: 0.005 μg
- Substrate: 100 μM
