Recombinant Mouse EMMPRIN/CD147 His-tag Protein, CF
R&D Systems | Catalog # 772-EM

Loading...
Key Product Details
- R&D Systems NS0-derived Recombinant Mouse EMMPRIN/CD147 His-tag Protein (772-EM)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
NS0
Accession Number
Applications
Bioactivity
Loading...
Product Specifications
Source
Mouse myeloma cell line, NS0-derived mouse EMMPRIN/CD147 protein
Ala22-Arg325, with a C-terminal 6-His tag
Ala22-Arg325, with a C-terminal 6-His tag
Purity
>95%, by SDS-PAGE visualized with Silver Staining and quantitative densitometry by Coomassie® Blue Staining.
Endotoxin Level
<0.10 EU per 1 μg of the protein by the LAL method.
N-terminal Sequence Analysis
Ala22
Predicted Molecular Mass
34 kDa
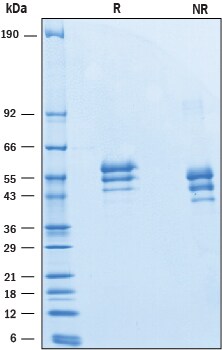
SDS-PAGE
43-66 kDa, reducing conditions
Activity
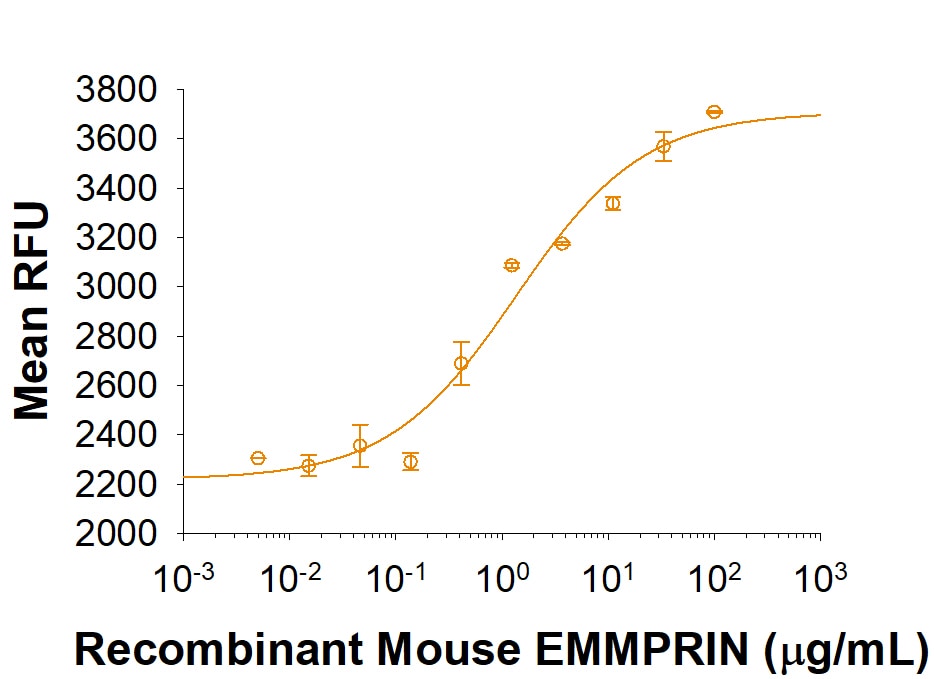
Measured by the ability of the immobilized protein to induce active MMP-1 secretion by NHLF human normal lung fibroblasts.
The ED50 for this effect is 0.6-4.8 μg/mL
The ED50 for this effect is 0.6-4.8 μg/mL
Scientific Data Images for Recombinant Mouse EMMPRIN/CD147 His-tag Protein, CF
Recombinant Mouse EMMPRIN/CD147 His-tag Protein Bioactivity
Recombinant Mouse EMMPRIN/CD147 induces NHLF human normal lung fibroblasts active MMP-1 secretion. The ED50 for this effect is 0.6-4.8 μg/mL.Recombinant Mouse EMMPRIN/CD147 His-tag Protein SDS-PAGE
2 μg/lane of Recombinant Mouse EMMPRIN/CD147 was resolved with SDS-PAGE under reducing (R) and non-reducing (NR) conditions and visualized by Coomassie® Blue staining, showing bands at 43-66 kDa.Formulation, Preparation, and Storage
772-EM
| Formulation | Lyophilized from a 0.2 μm filtered solution in PBS. |
| Reconstitution | Reconstitute at 500 μg/mL in PBS. |
| Shipping | The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage |
|
Calculators
Background: EMMPRIN/CD147
References
- Gabison, E. E. et al. (2005) Biochimie 87:361.
- Yurchenko, V. et al. (2006) Immunology 117:301.
- Kasinrerk, W. et al. (1992) J. Immunol. 149:847.
- Iacono, K.T. et al. (2007) Exp. Mol. Pathol. 83:283.
- Riethdorf, S. et al. (2006) Int. J. Cancer 119:1800.
- Quemener, C. et al. (2007) Cancer Res. 67:9.
- Wilson, M. C. et al. (2005) J. Biol. Chem. 280:27213.
- Xu, D. and M. E. Hemler (2005) Mol. Cell. Proteomics 4:1061.
- Tang, W. et al. (2004) Mol. Biol. Cell 15:4043.
- Zhao, P. et al. (2010) Cancer Sci. 101:387.
- Dai, J. et al. (2009) BMC Cancer 9:337.
- Li, Y. et al. (2012) J. Biol. Chem. 287:4759.
- Hibino, T. et al. (2013) Cancer Res. 73:172.
- Arora, K. et al. (2005) J. Immunol. 175:517.
- Pushkarsky, T. et al. (2005) J. Biol. Chem. 280:27866.
- Egawa, N. et al. (2006) J. Biol. Chem. 281:37576.
- Sidhu, S. S. et al. (2004) Oncogene 23:956.
- Heller, M. et al. (2003) J. Neurochem. 84:557.
- Mannowetz N. et al. (2012) J. cell Physiol. 227:2154.
Long Name
Extracellular Matrix Metalloproteinase Inducer
Alternate Names
Basigin, BSG, CD147
Gene Symbol
BSG
UniProt
Additional EMMPRIN/CD147 Products
Product Documents for Recombinant Mouse EMMPRIN/CD147 His-tag Protein, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Mouse EMMPRIN/CD147 His-tag Protein, CF
For research use only
Citations for Recombinant Mouse EMMPRIN/CD147 His-tag Protein, CF
Customer Reviews for Recombinant Mouse EMMPRIN/CD147 His-tag Protein, CF
There are currently no reviews for this product. Be the first to review Recombinant Mouse EMMPRIN/CD147 His-tag Protein, CF and earn rewards!
Have you used Recombinant Mouse EMMPRIN/CD147 His-tag Protein, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Loading...