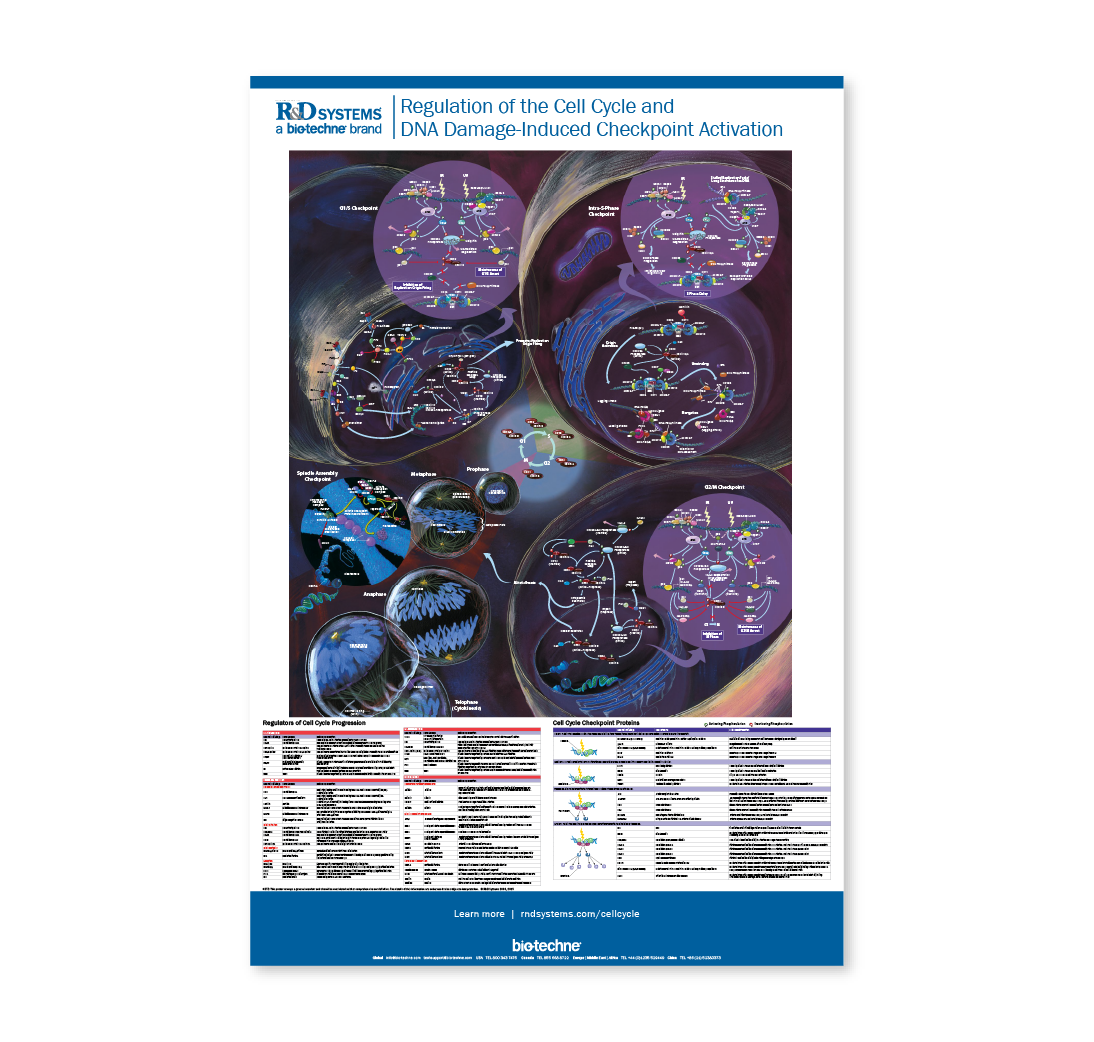
Regulation of the Cell Cycle & DNA Damage-Induced Checkpoint Activation
Life Science PostersThe human genome is exposed to potentially deleterious events during every cell division cycle. Cellular metabolism or routine errors in DNA replication and recombination represent endogenous sources of potential DNA damage. In addition, exposure to exogenous genotoxic agents such as ionizing radiation (IR), ultraviolet light (UV), oxidative stress, and chemical mutagens can lead to nucleotide modifications and DNA single- and double-strand breaks. In order to protect the integrity of the genome, a series of proteins are activated when DNA damage is present to either induce cell cycle arrest and initiate the appropriate DNA repair pathway, or promote apoptosis. The poster shows the critical proteins and phosphorylation events involved in regulating progression of the mammalian cell cycle and the mechanisms by which cell cycle progression can be inhibited when DNA damage is present. Specific functions of the proteins depicted in the poster are described in the tables at the bottom. Proteins listed in table on the bottom left are involved in normal cell cycle progression and are separated based on whether they function in G1, G2, S phase, or mitosis. Proteins listed in the table on the bottom right are required for checkpoint activation. Each checkpoint protein is categorized as a checkpoint sensor, mediator, transducer, or effector protein based on the role that it plays in checkpoint activation. Critical activating (green P) and inactivating (red P) phosphorylation events mediated by cyclin-dependent kinases (CDKs), cyclin-activating kinase (CAK), CDC7/DBF4, Myt1, Plk1, Wee1, ATM, ATR, Chk1, and Chk2 are indicated throughout.
Related Information
- Cell Cycle
- DNA Damage and Repair
- DNA Damage and Repair Kits & Reagents
- Intracellular Kinases
- Mini-review: DNA Damage Response