Differentiation of Hematopoietic Progenitors from hPSCs
In Brief
Nafria et al. describe a 3-stage procedure for the differentiation of human pluripotent stem cells to multipotent hematopoietic progenitors in 14+ days using a cocktail of small molecules and growth factors.
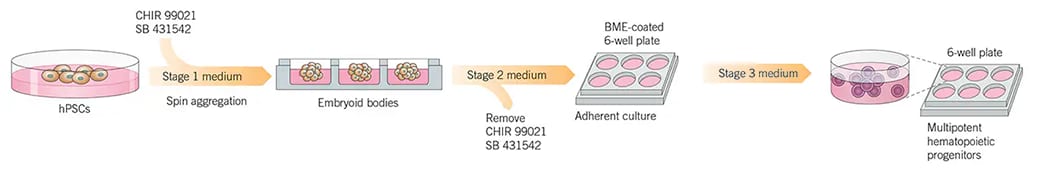
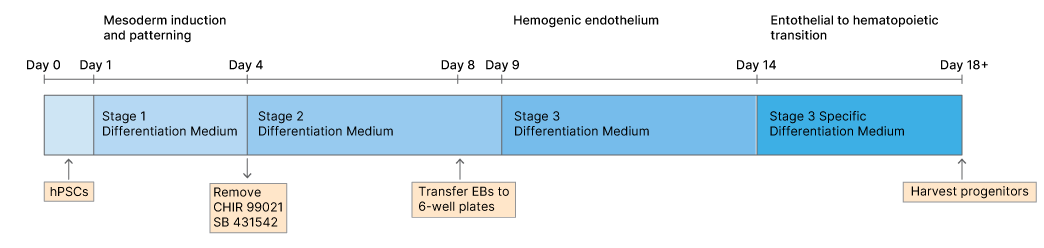
Briefly, human pluripotent stem cells (hPSCs) are co-cultured with feeder cells for 1 week, after which the hPSCs are harvested, dissociated and seeded in non-adherent round-bottom 96 well plates in stage 1 differentiation medium. On day 0 plates are centrifuged and cells aggregate forming embryoid bodies (EB; 1 per well). On day 1, SB 431542 and CHIR 99021 are added to the Stage 1 differentiation medium to direct the mesoderm toward the definitive hematopoietic program. On day 4 the medium is exchanged for Stage 2 medium without small molecule inhibitors. At day 8 of differentiation EBs are transferred to Basement Membrane Extract (BME, e.g. CultrexTM) coated 6-well plates containing stage 2 medium. The next day (Day 9) the medium is removed and replaced with stage 3 medium. Then around day 13-15 Stage 3 medium is replaced with Stage 3 Specific medium to promote proliferation and differentiation of hematopoietic lineage cells.
Vascular structures and hemogenic endothelium expressing the SOX17 marker are generated from day 8 to 14 and from day 14 onward, SOX17- CD34+ RUNX1C-/+ hematopoietic progenitors are generated within the SOX17+ endothelium displaying similarities to human aorta-gonad mesonephros (AGM) and adult hematopoietic stem cells.
Cocktails
| Stage1 Differentiation Medium | Stage 2 Differentiation Medium | Stage 3 Differentiation Medium | Stage 3 Specific Medium | ||||
|---|---|---|---|---|---|---|---|
| STAPEL medium | STAPEL medium | STAPEL medium | STAPEL medium | ||||
| CHIR 99021 (Cat.No. 4423) | 0.5 μM | BMP-4 (314-BP/CF) | 20 ng/mL | BMP-4 (314-BP/CF) | 10 ng/mL | SCF (7466-SC/CF) | 100 ng/mL |
| BMP-4 (314-BP/CF) | 20 ng/mL | VEGF (293-VE/CF) | 50 ng/mL | SCF (7466-SC/CF) | 100 ng/mL | Flt3 (308-FK/CF) | 100 ng/mL |
| VEGF (293-VE/CF) | 25 ng/mL | SCF (7466-SC/CF) | 50 ng/mL | Flt3 (308-FK/CF) | 100 ng/mL | TPO (288-TP/CF) | 50 ng/mL |
| SCF (7466-SC/CF) | 25 ng/mL | IGF-2 (292-G2) | 10 ng/mL | TPO (288-TP/CF) | 50 ng/mL | IL-6 (7270-IL/CF) | 25 ng/mL |
| Activin A (388-AC/CF) | 7.5 ng/mL | FGF-2 (3718-FB) | 10 ng/mL | VEGF (293-VE/CF) | 50 ng/mL | IL-3 (203-IL/CF) | 25 ng/mL |
| FGF-2 (3718-FB) | 10 ng/mL | IL-6 (7270-IL/CF) | 25 ng/mL | Penicillin/Streptomycin (B21210) | Optional | ||
| From Day 1 | IL-3 (203-IL/CF) | 25 ng/mL | |||||
| CHIR 99021 (Cat.No. 4423) | 3 μM | IGF-2 (292-G2) | 10 ng/mL | ||||
| SB 431542 (Cat.No. 1614) | 3.8 μM | FGF-2 (3718-FB) | 10 ng/mL | ||||
| Penicillin/Streptomycin (B21210) | Optional | ||||||
Reference
Nafria et al. (2020) Protocol for the generation of definitive hematopoietic progenitors from human pluripotent stem cells.1 100130 PMID: 33377024
