FGF acidic, also known as FGF1, ECGF, and HBGF-1, is a 17 kDa nonglycosylated member of the FGF family of mitogenic peptides. FGF acidic, which is produced by multiple cell types, stimulates the proliferation of all cells of mesodermal origin and many cells of neuroectodermal, ectodermal, and endodermal origin. It plays a number of roles in development, regeneration, and angiogenesis (1‑3). Bovine FGF acidic shares 53% amino acid sequence identity with FGF basic and 11%‑31% with other bovine FGFs. It shares 92%, 91%, 88%, and 91% aa sequence identity with human, mouse, porcine, and rat FGF acidic, respectively, and exhibits considerable species crossreactivity. During its nonclassical secretion, FGF acidic associates with S100A13, copper ions, and the C2A domain of synaptotagmin 1 (4). It is released extracellularly as a disulfide-linked homodimer and is stored in complex with extracellular heparan sulfate (5). The ability of heparan sulfate to bind FGF acidic is determined by its pattern of sulfation, and alterations in this pattern during embryognesis thereby regulate FGF acidic bioactivity (6). The association of FGF acidic with heparan sulfate is a prerequisite for its subsequent interaction with FGF receptors (7, 8). Ligation triggers receptor dimerization, transphosphorylation, and internalization of receptor/FGF complexes (9). Internalized FGF acidic can translocate to the cytosol with the assistance of Hsp90 and then migrate to the nucleus by means of its two nuclear localization signals (10‑12). The phosphorylation of FGF acidic by nuclear PKC delta triggers its active export to the cytosol where it is dephosphorylated and degraded (13, 14). Intracellular FGF acidic functions as a survival factor by inhibiting p53 activity and proapoptotic signaling (15).
Bovine FGF acidic/FGF1 Antibody
R&D Systems | Catalog # AB-32-NA

Loading...
Key Product Details
Species Reactivity
Validated:
Bovine
Cited:
Rat, Hamster
Applications
Validated:
Western Blot, Neutralization
Cited:
Immunohistochemistry-Paraffin, Western Blot
Label
Unconjugated
Antibody Source
Polyclonal Rabbit IgG
Loading...
Product Specifications
Immunogen
Bovine brain-derived FGF acidic/FGF1
Specificity
Detects bovine FGF acidic/FGF1 in direct ELISAs and Western blots. In direct ELISAs and Western blots, this antibody will recognize both bovine FGF acidic/FGF1 and recombinant human (rh) FGF acidic/FGF1. In direct ELISAs, no cross‑reactivity with rhFGF-basic, rhFGF-4, -5, -6, -7 or -9 is observed. Neutralizes the biological activity of bovine FGF acidic/FGF1 and will also neutralize the biological activity of rhFGF acidic/FGF1, although 5 times the amount of IgG is required.
Clonality
Polyclonal
Host
Rabbit
Isotype
IgG
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Bovine FGF acidic/FGF1 Antibody
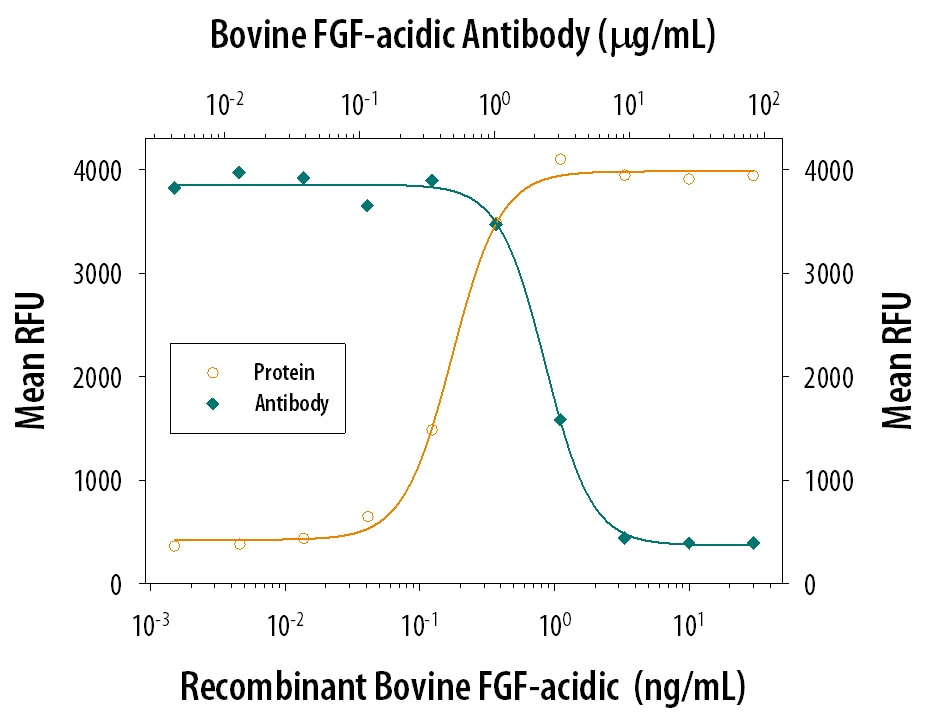
Cell Proliferation Induced by FGF acidic/FGF1 and Neutral-ization by Bovine FGF acidic/FGF1 Antibody.
Bovine FGF acidic/FGF1 (Catalog # 132-FA) stimulates proliferation in the NR6R-3T3 mouse fibroblast cell line in a dose-dependent manner (orange line). Proliferation elicited by Bovine FGF acidic/FGF1 (0.75 ng/mL) is neutralized (green line) by increasing concentrations of Rabbit Anti-Bovine FGF acidic/FGF1 Polyclonal Antibody (Catalog # AB-32-NA). The ND50 is typically 1-3 µg/mL.Applications for Bovine FGF acidic/FGF1 Antibody
Application
Recommended Usage
Western Blot
1 µg/mL
Sample: Bovine FGF acidic/FGF1 (Catalog # 132-FA)
Sample: Bovine FGF acidic/FGF1 (Catalog # 132-FA)
Neutralization
Measured by its ability to neutralize FGF acidic/FGF1-induced proliferation in the NR6R‑3T3 mouse fibroblast cell line. The Neutralization Dose (ND50) is typically 1-3 µg/mL in the presence of 0.75 ng/mL Bovine FGF acidic/FGF1.
Formulation, Preparation, and Storage
Purification
Protein A or G purified
Reconstitution
Reconstitute at 1 mg/mL in sterile PBS.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose.
Shipping
The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: FGF acidic/FGF1
References
- Gimenez-Gallego, G. et al. (1985) Science 230:1385.
- Galzie, Z. et al. (1997) Biochem. Cell Biol. 75:669.
- Presta, M. et al. (2005) Cytokine Growth Factor Rev. 16:159.
- Rajalingam, D. et al. (2007) Biochemistry 46:9225.
- Guerrini, M. et al. (2007) Curr. Pharm. Des. 13:2045.
- Allen, B.L. and A.C. Rapraeger (2003) J. Cell Biol. 163:637.
- Robinson, C.J. et al. (2005) J. Biol. Chem. 280:42274.
- Mohammadi, M. et al. (2005) Cytokine Growth Factor Rev. 16:107.
- Wiedlocha, A. and V. Sorensen (2004) Curr. Top. Microbiol. Immunol. 286:45.
- Wesche, J. et al. (2006) J. Biol. Chem. 281:11405.
- Imamura, T. et al. (1990) Science 249:1567.
- Wesche, J. et al. (2005) Biochemistry 44:6071.
- Wiedlocha, A. et al. (2005) Mol. Biol. Cell 16:794.
- Nilsen, T. et al. (2007) J. Biol. Chem. 282:26245.
- Bouleau, S. et al. (2005) Oncogene 24:7839.
Long Name
Fibroblast Growth Factor acidic
Alternate Names
alpha-ECGF, beta-ECGF, FGF acidic, FGF-1, HBGF-1
Gene Symbol
FGF1
Additional FGF acidic/FGF1 Products
Product Documents for Bovine FGF acidic/FGF1 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Bovine FGF acidic/FGF1 Antibody
For research use only
Related Research Areas
Citations for Bovine FGF acidic/FGF1 Antibody
Customer Reviews for Bovine FGF acidic/FGF1 Antibody
There are currently no reviews for this product. Be the first to review Bovine FGF acidic/FGF1 Antibody and earn rewards!
Have you used Bovine FGF acidic/FGF1 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
