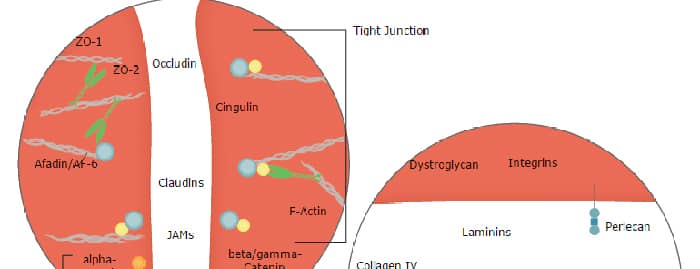
Dystroglycan, also DAG-1 (Dystrophin-associated glycoprotein 1) is a 180-200 kDa heterodimeric adhesion molecule that links the cell cytoskeleton to the extracellular matrix. It is found on skeletal muscle, cardiac muscle, fibroblasts, smooth muscle and keratinocytes. DAG-1 binds multiple matrix molecules, including laminin-1 and -2, agrin, and perlecan. Intracellularly, the cytoplasmic tail of DAG-1 contributes to a large 400 kDa complex that interacts with the cytoskeleton. The human DAG-1 preprocursor is a type I transmembrane protein 895 amino acids (aa) in length. It contains a 27 aa signal sequence plus an 868 aa proform that undergoes autocatalysis to generate a 626 aa alpha -chain (aa 28-653), and a 242 aa beta -chain. Mature DAG-1 is a heterodimer composed of noncovalently linked alpha ‑ and beta ‑chains. The alpha -chain possesses one potential Ig-like domain (aa 64-162), a mucin-like region (aa 316-485), and a peptidase S72 domain (aa 500-733). It is O‑glycosylated and runs from 100-160 kDa in SDS-PAGE. The beta -chain is N-glycosylated and runs at 42-44 kDa in SDS-Page. It possesses a short 95 aa extracellular region (aa 654-749) plus a 120 aa cytoplasmic domain (aa 776-895). Membrane cleavage of the beta -chain causes dissociation of the heterodimer and generates a 30 kDa truncated form. Over aa 28-749, human DAG-1 shares 93% aa identity with mouse DAG-1.
Human Dystroglycan Antibody
R&D Systems | Catalog # AF6868

Key Product Details
Validated by
Knockout/Knockdown, Biological Validation
Species Reactivity
Validated:
Human
Cited:
Human, Mouse, Canine, Rabbit, Transgenic Mouse
Applications
Validated:
Knockout Validated, Immunohistochemistry, Western Blot
Cited:
Immunohistochemistry, Western Blot, Flow Cytometry
Label
Unconjugated
Antibody Source
Polyclonal Sheep IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant human Dystroglycan
Gln28-Val749
Accession # Q14118
Gln28-Val749
Accession # Q14118
Specificity
Detects human Dystroglycan in direct ELISAs and Western blots.
Clonality
Polyclonal
Host
Sheep
Isotype
IgG
Scientific Data Images for Human Dystroglycan Antibody
Detection of Human Dystroglycan by Western Blot.
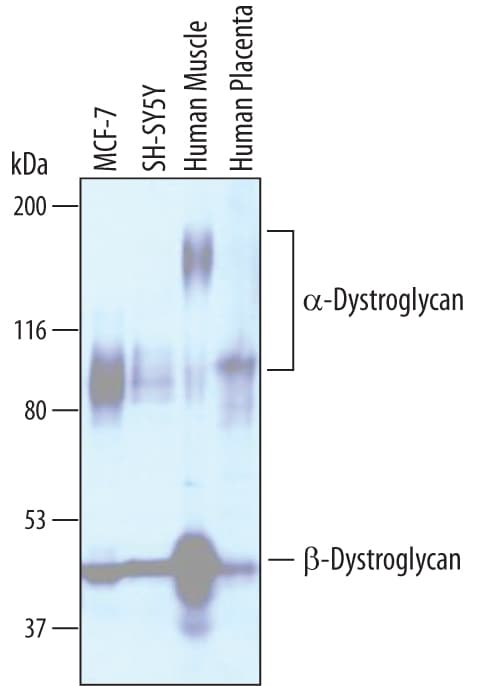
Western blot shows lysates of MCF-7 human breast cancer cell line, SH-SY5Y human neuroblastoma cell line, human muscle tissue, and human placenta tissue. PVDF membrane was probed with 1 µg/mL of Sheep Anti-Human Dystroglycan Antigen Affinity-purified Polyclonal Antibody (Catalog # AF6868) followed by HRP-conjugated Anti-Sheep IgG Secondary Antibody (Catalog # HAF016). Specific bands were detected for a-Dystroglycan at approximately 100-160 kDa (as indicated) and beta -Dystroglycan at approximately 42-44 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Dystroglycan in Human Skeletal Muscle.
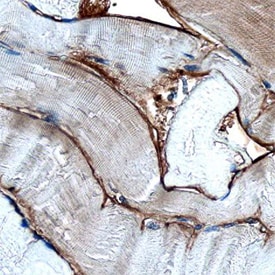
Dystroglycan was detected in immersion fixed paraffin-embedded sections of human skeletal muscle using Sheep Anti-Human Dystroglycan Antigen Affinity-purified Polyclonal Antibody (Catalog # AF6868) at 3 µg/mL overnight at 4 °C. Before incubation with the primary antibody, tissue was subjected to heat-induced epitope retrieval using Antigen Retrieval Reagent-Basic (Catalog # CTS013). Tissue was stained using the Anti-Sheep HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS019) and counterstained with hematoxylin (blue). Specific staining was localized to basement membrane. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Western Blot Shows Human Dystroglycan Specificity by Using Knockout Cell Line.
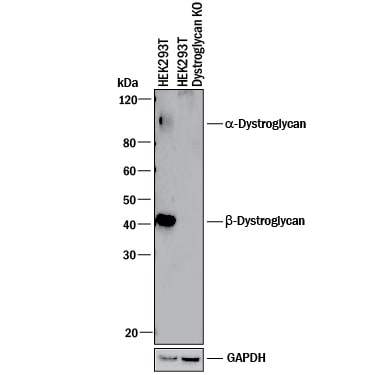
Western blot shows lysates of HEK293T human embryonic kidney parental cell line and Dystroglycan knockout HEK293T cell line (KO). PVDF membrane was probed with 1 µg/mL of Sheep Anti-Human Dystroglycan Antigen Affinity-purified Polyclonal Antibody (Catalog # AF6868) followed by HRP-conjugated Anti-Sheep IgG Secondary Antibody (Catalog # HAF016). Specific bands were detected for a--Dystroglycan at approximately 110 kDa -and beta -Dystroglycan at approximately 42 kDa (as indicated) in the parental HEK293T cell line, but is not detectable in knockout HEK293T cell line. GAPDH (Catalog # AF5718) is shown as a loading control. This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Detection of Human Dystroglycan by Western Blot
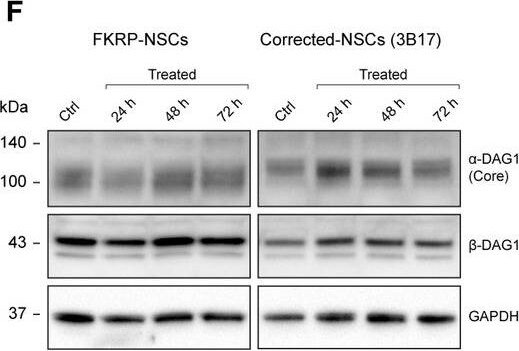
Hit compound 4BPPNit validation in the FKRP dystroglycanopathy human iPSC modelsFKRP‐NSCs treated with 4BPPNit showed increased IIH6 reactivity, compared with untreated control cells. Note that the augmented IIH6 reactivity by 4BPPNit is much weaker than that in CRISPR‐corrected‐NSCs. Under the same exposure time, the signal intensity of IIH6 reactivity is saturated in corrected‐NSCs.Representative immunoblots with the IIH6 antibody using FKRP‐ and corrected‐NSCs treated with 4BPPNit for 24, 48, and 72 h, and DMSO only controls.Quantification of alpha ‐dystroglycan glycosylation (IIH6), compared with DMSO only controls.Representative immunoblots of laminin‐binding analysis using FKRP‐ and corrected‐NSCs treated with 4BPPNit and DMSO only controls.Quantification of laminin‐binding activities, compared with DMSO only controls.Representative immunoblots with the AF6868 antibody detecting both alpha ‐dystroglycan core protein and beta ‐dystroglycan in FKRP‐ and corrected‐NSCs treated with 4BPPNit and DMSO only controls.Quantification of alpha ‐dystroglycan core protein and beta ‐dystroglycan expression, compared with DMSO only controls.Data information: Intensities of glycosylation, laminin‐binding activity, or protein expression are normalized to GAPDH protein expression. Note that the left and right panels of the immunoblots in (B) and (D) are not equivalent exposure time. See also Appendix Fig S5. Values indicate mean ± s.d. (n = 3 or 4 biological replicates; one‐way ANOVA; NS, not significant; *P < 0.05; **P < 0.01). Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31566294), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human Dystroglycan Antibody
Application
Recommended Usage
Immunohistochemistry
5-15 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human skeletal muscle
Sample: Immersion fixed paraffin-embedded sections of human skeletal muscle
Knockout Validated
Dystroglycan
is specifically detected in HEK293T human embryonic kidney parental cell line but is not detectable in
Dystroglycan knockout HEK293T cell line.
Western Blot
1 µg/mL
Sample: MCF‑7 human breast cancer cell line, SH‑SY5Y human neuroblastoma cell line, human muscle tissue, and human placenta tissue
Sample: MCF‑7 human breast cancer cell line, SH‑SY5Y human neuroblastoma cell line, human muscle tissue, and human placenta tissue
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Sterile PBS to a final concentration of 0.2 mg/mL. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Dystroglycan
Long Name
Dystrophin-associated Glycoprotein 1
Alternate Names
AGRNR, Dag-1, DAG1
Gene Symbol
DAG1
UniProt
Additional Dystroglycan Products
Product Documents for Human Dystroglycan Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Dystroglycan Antibody
For research use only
Related Research Areas
Citations for Human Dystroglycan Antibody
Customer Reviews for Human Dystroglycan Antibody
There are currently no reviews for this product. Be the first to review Human Dystroglycan Antibody and earn rewards!
Have you used Human Dystroglycan Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways
 Blood-Brain Barrier Pathway: Anatomy
Blood-Brain Barrier Pathway: Anatomy