Fibroblast growth factors (FGFs) comprise a family of at least eighteen structurally related proteins that are involved in a multitude of physiological and pathological cellular processes, including cell growth, differentiation, angiogenesis, wound healing and tumorgenesis. The biological activities of the FGFs are mediated by a family of type I transmembrane tyrosine kinases which undergo dimerization and autophosphorylation after ligand binding. Four distinct genes encoding closely related FGF receptors, FGF R1‑4, are known. All four genes for FGF Rs encode proteins with an N-terminal signal peptide, three immunoglobulin (Ig)-like domains, an acid-box region containing a run of acidic residues between the IgI and IgII domains, a transmembrane domain and the split tyrosine-kinase domain. Multiple forms of FGF R1‑3 are generated by alternative splicing of the mRNAs. A frequent splicing event involving FGF R1 and 2 results in receptors containing all three Ig domains, referred to as the alpha isoform, or only IgII and IgIII, referred to as the beta isoform. Only the alpha isoform has been identified for FGF R3 and FGF R4. Additional splicing events for FGF R1‑3, involving the C-terminal half of the IgIII domain encoded by two mutually exclusive alternative exons, generate FGF receptors with alternative IgIII domains (IIIb and IIIc). A IIIa isoform which is a secreted FGF binding protein containing only the N-terminal half of the IgIII domain plus some intron sequences has also been reported for FGF R1. Mutations in FGF R1‑3 have been found in patients with birth defects involving craniosynostosis. The complex patterns of expression of these receptors as well as the specificity of their interactions with the various FGF ligand family members are under investigation.

Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human
Applications
Validated:
Immunohistochemistry, Western Blot, Flow Cytometry, Immunocytochemistry, CyTOF-ready
Cited:
Immunohistochemistry
Label
Unconjugated
Antibody Source
Monoclonal Rat IgG2A Clone # 240929
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant human FGF R4
Leu22-Asp369 (predicted)
Accession # P22455
Leu22-Asp369 (predicted)
Accession # P22455
Specificity
Detects human FGF R4 in direct ELISAs and Western blots. In Western blots, less than 5% cross-reactivity with recombinant mouse FGF R4 and recombinant human FGF R5 is observed and no cross-reactivity with any isoform of rhFGF R1, rhFGF R2, or rhFGF R3 is observed.
Clonality
Monoclonal
Host
Rat
Isotype
IgG2A
Scientific Data Images for Human FGFR4 Antibody
Detection of Human FGF R4 by Western Blot.
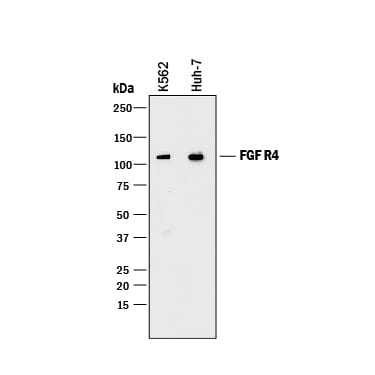
Western blot shows lysates of K562 human chronic myelogenous leukemia cell line and Huh-7 human hepatoma cell line. PVDF membrane was probed with 0.2 µg/mL of Rat Anti-Human FGF R4 Monoclonal Antibody (Catalog # MAB6852) followed by HRP-conjugated Anti-Rat IgG Secondary Antibody (HAF005). A specific band was detected for FGF R4 at approximately 110 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.FGF R4 in Human Pancreas.
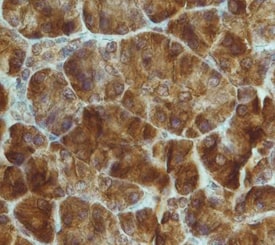
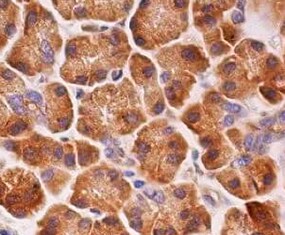
FGF R4 was detected in immersion fixed paraffin-embedded sections of human pancreas using Rat Anti-Human FGF R4 Monoclonal Antibody (Catalog # MAB6852) at 15 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Rat HRP-DAB Cell & Tissue Staining Kit (brown; CTS017) and counterstained with hematoxylin (blue). Specific staining was localized to cytoplasm. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.FGF R4 in MCF‑7 Human Cell Line.
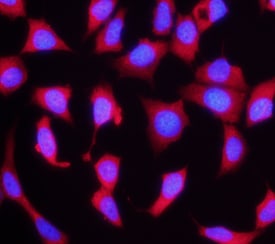
FGF R4 was detected in immersion fixed MCF-7 human breast cancer cell line using Rat Anti-Human FGF R4 Monoclonal Antibody (Catalog # MAB6852) at 10 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Rat IgG Secondary Antibody (red; NL013) and counterstained with DAPI (blue). Specific staining was localized to cytoplasm. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Detection of FGFR4 by Western Blot
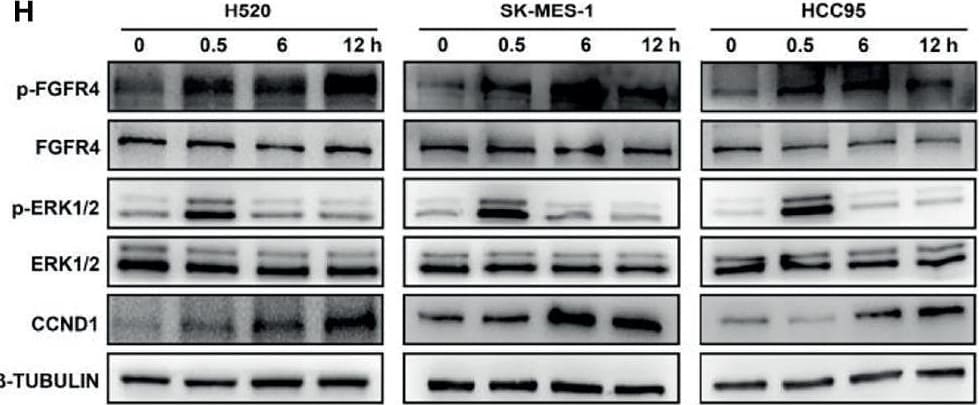
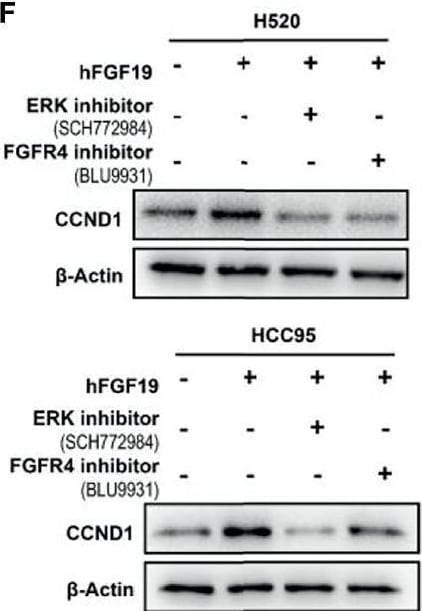
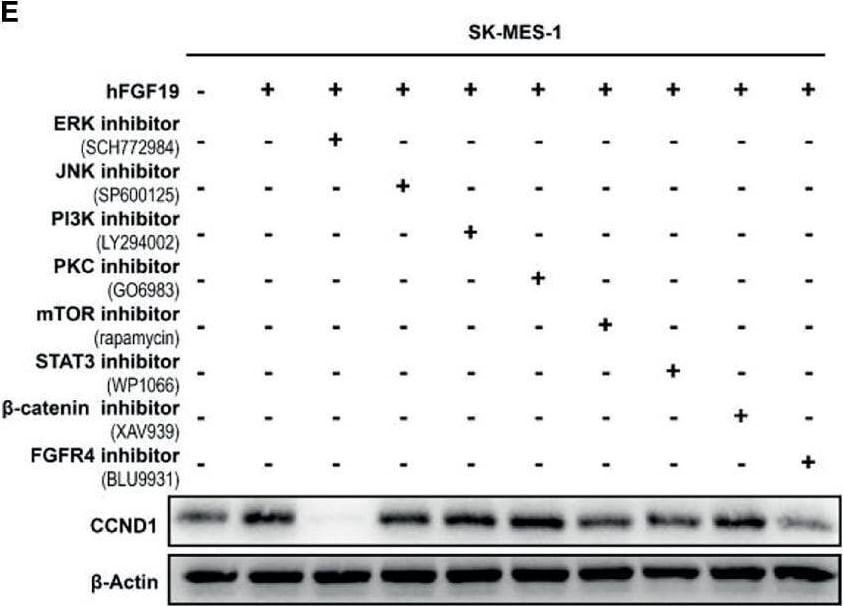
FGF19 enhances CCND1 expression by FGF19-FGFR4-ERK1/2 axis in LUSC cells. (A) Recombinant human FGF19 (rhFGF19) (25 ng/ml) promoted expression of CCND1 in SK-MES-1 and H520 cells (serum starved for 12 h before treatment) in a time-dependent manner. (B) CCND1 mRNA expression levels in LUSC cell lines after treatment with rhFGF2 for 12 h. (C) Expression of CCND1 in FGF19 overexpression LUSC cell line. (D) Expression of CCND1 in FGF19 knockdown LUSC cell line. (E) A panel of inhibitors against a number of signaling pathways was used to dissect the leading factors of regulated by FGF19. (F) Western blot and (G) qPCR analysis of CCND1 expression in H520 and HCC95 LUSC cells after treatment of FGF19, FGF19 and SCH772984, FGF19 & BLU9931, or DMSO as control. (H) Effects of the FGF19/FGFR4 pathway on protein levels of CCND1, p-FGFR4, and p-ERK1/2 by western blot analysis. H520, SK-MES-1 and HCC95 cells were treated with FGF19 (25 ng/ml, 0/0.5/6/12 h) to activate the FGF19/FGFR4 signaling pathway. Data were shown as mean ± SD bars and compared by unpaired t-test. **p <0.01; ****p <0.0001; ns, not significant. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35463335), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of FGFR4 in HepG2 cells by Flow Cytometry.
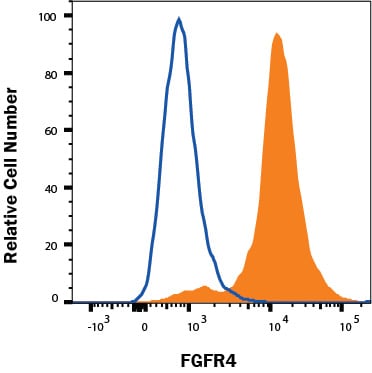
HepG2 cells were stained with Rat Anti-Human FGFR4 Monoclonal Antibody (Catalog # MAB6852, filled histogram) or isotype control antibody (Catalog # MAB006, open histogram), followed by Phycoerythrin-conjugated Anti-Rat IgG Secondary Antibody (Catalog # F0105B). View our protocol for Staining Membrane-associated Proteins.Detection of FGFR4 by Western Blot
FGF19 enhances CCND1 expression by FGF19-FGFR4-ERK1/2 axis in LUSC cells. (A) Recombinant human FGF19 (rhFGF19) (25 ng/ml) promoted expression of CCND1 in SK-MES-1 and H520 cells (serum starved for 12 h before treatment) in a time-dependent manner. (B) CCND1 mRNA expression levels in LUSC cell lines after treatment with rhFGF2 for 12 h. (C) Expression of CCND1 in FGF19 overexpression LUSC cell line. (D) Expression of CCND1 in FGF19 knockdown LUSC cell line. (E) A panel of inhibitors against a number of signaling pathways was used to dissect the leading factors of regulated by FGF19. (F) Western blot and (G) qPCR analysis of CCND1 expression in H520 and HCC95 LUSC cells after treatment of FGF19, FGF19 and SCH772984, FGF19 & BLU9931, or DMSO as control. (H) Effects of the FGF19/FGFR4 pathway on protein levels of CCND1, p-FGFR4, and p-ERK1/2 by western blot analysis. H520, SK-MES-1 and HCC95 cells were treated with FGF19 (25 ng/ml, 0/0.5/6/12 h) to activate the FGF19/FGFR4 signaling pathway. Data were shown as mean ± SD bars and compared by unpaired t-test. **p <0.01; ****p <0.0001; ns, not significant. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35463335), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of FGFR4 by Western Blot
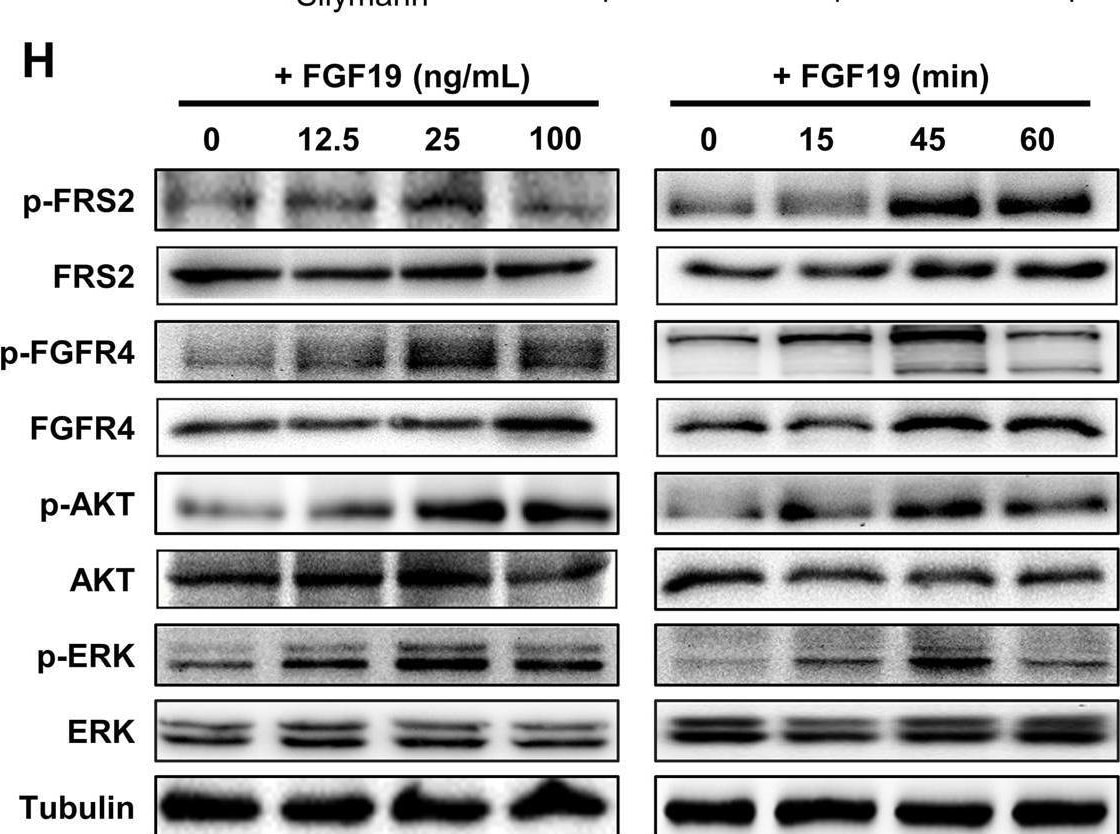
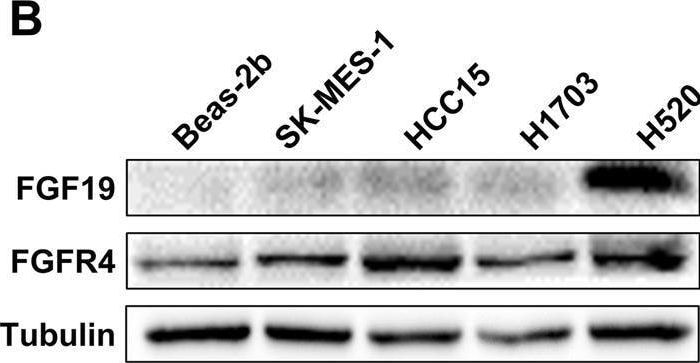
CDCA and ER stress induce FGF19 upregulation and hFGF19 activates ERK/AKT signaling to promote LSQ cell proliferation. A FGF19 (a) and FGFR4 (b) mRNA expression levels in LSQ cell lines and Beas-2b. b Protein expression levels of FGF19 and FGFR4 in LSQ cell lines and bronchial epithelial cell line Beas-2b. c The effects of chenodeoxycholic acid (CDCA) treatment on FGF19 protein levels in Beas-2b cells in a dose-dependent (ranging from 0 to 100 µM, upper panel) and time-dependent (within 48 h, lower panel) manner. d The effect of chenodeoxycholic acid treatment (50 µM) on the apoptosis of Beas-2b cells with quantifications of the results on the right panel. e mRNA expression of FGF19 (left panel) and GRP78 (right panel) of LSQ cell lines H520 and SK-MES-1 treated with thapsigargin (TG) or tunicamycin (TM) for 24 h. f FGF19 protein expression of H520 and SK-MES-1 treated with TG or TM for 24 h, in the presence or absence of ER stress suppressor silymarin. g Cell proliferation of SK-MES-1 cells treated with conditioned medium from H520 cells overexpressing FGF19 was determined by CCK-8 assays. h The effects of hFGF19 on the activation of ERK/AKT signaling in SK-MES-1 cells in a dose-dependent (left panel) or time-dependent (right panel) manner. i Enrichment plots of mTOR expression signatures according to FGF19 expression levels in an LSQ cohort. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32111983), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of FGFR4 by Western Blot
FGF19 enhances CCND1 expression by FGF19-FGFR4-ERK1/2 axis in LUSC cells. (A) Recombinant human FGF19 (rhFGF19) (25 ng/ml) promoted expression of CCND1 in SK-MES-1 and H520 cells (serum starved for 12 h before treatment) in a time-dependent manner. (B) CCND1 mRNA expression levels in LUSC cell lines after treatment with rhFGF2 for 12 h. (C) Expression of CCND1 in FGF19 overexpression LUSC cell line. (D) Expression of CCND1 in FGF19 knockdown LUSC cell line. (E) A panel of inhibitors against a number of signaling pathways was used to dissect the leading factors of regulated by FGF19. (F) Western blot and (G) qPCR analysis of CCND1 expression in H520 and HCC95 LUSC cells after treatment of FGF19, FGF19 and SCH772984, FGF19 & BLU9931, or DMSO as control. (H) Effects of the FGF19/FGFR4 pathway on protein levels of CCND1, p-FGFR4, and p-ERK1/2 by western blot analysis. H520, SK-MES-1 and HCC95 cells were treated with FGF19 (25 ng/ml, 0/0.5/6/12 h) to activate the FGF19/FGFR4 signaling pathway. Data were shown as mean ± SD bars and compared by unpaired t-test. **p <0.01; ****p <0.0001; ns, not significant. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35463335), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of FGFR4 by Western Blot
CDCA and ER stress induce FGF19 upregulation and hFGF19 activates ERK/AKT signaling to promote LSQ cell proliferation. A FGF19 (a) and FGFR4 (b) mRNA expression levels in LSQ cell lines and Beas-2b. b Protein expression levels of FGF19 and FGFR4 in LSQ cell lines and bronchial epithelial cell line Beas-2b. c The effects of chenodeoxycholic acid (CDCA) treatment on FGF19 protein levels in Beas-2b cells in a dose-dependent (ranging from 0 to 100 µM, upper panel) and time-dependent (within 48 h, lower panel) manner. d The effect of chenodeoxycholic acid treatment (50 µM) on the apoptosis of Beas-2b cells with quantifications of the results on the right panel. e mRNA expression of FGF19 (left panel) and GRP78 (right panel) of LSQ cell lines H520 and SK-MES-1 treated with thapsigargin (TG) or tunicamycin (TM) for 24 h. f FGF19 protein expression of H520 and SK-MES-1 treated with TG or TM for 24 h, in the presence or absence of ER stress suppressor silymarin. g Cell proliferation of SK-MES-1 cells treated with conditioned medium from H520 cells overexpressing FGF19 was determined by CCK-8 assays. h The effects of hFGF19 on the activation of ERK/AKT signaling in SK-MES-1 cells in a dose-dependent (left panel) or time-dependent (right panel) manner. i Enrichment plots of mTOR expression signatures according to FGF19 expression levels in an LSQ cohort. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32111983), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human FGFR4 Antibody
Application
Recommended Usage
CyTOF-ready
Ready to be labeled using established conjugation methods. No BSA or other carrier proteins that could interfere with conjugation.
Flow Cytometry
0.25 µg/106 cells
Sample: HepG2 cells
Sample: HepG2 cells
Immunocytochemistry
8-25 µg/mL
Sample: Immersion fixed BG01V human embryonic stem cells and MCF‑7 human breast cancer cell line
Sample: Immersion fixed BG01V human embryonic stem cells and MCF‑7 human breast cancer cell line
Immunohistochemistry
8-25 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human pancreas
Sample: Immersion fixed paraffin-embedded sections of human pancreas
Western Blot
0.2 µg/mL
Sample: K562 human chronic myelogenous leukemia cell line and Huh‑7 human hepatoma cell line
Sample: K562 human chronic myelogenous leukemia cell line and Huh‑7 human hepatoma cell line
Reviewed Applications
Read 1 review rated 5 using MAB6852 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Protein A or G purified from hybridoma culture supernatant
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: FGFR4
References
- Galzie, Z. et al. (1997) Biochem. Cell Biol. 75:669.
- Burke, D. et al. (1998) Trends Biochem. Sci. 23:59.
Long Name
Fibroblast Growth Factor Receptor 4
Alternate Names
CD334, FGF R4
Gene Symbol
FGFR4
UniProt
Additional FGFR4 Products
Product Documents for Human FGFR4 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human FGFR4 Antibody
For research use only
Related Research Areas
Citations for Human FGFR4 Antibody
Customer Reviews for Human FGFR4 Antibody (1)
5 out of 5
1 Customer Rating
Have you used Human FGFR4 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
Application: ImmunohistochemistrySample Tested: Pancreas tissueSpecies: HumanVerified Customer | Posted 07/21/2022
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars