Glut1 belongs to the facilitative glucose transport protein family that comprises 13 members. It is an integral membrane protein with 12 transmembrane domains and is expressed at variable levels in many tissues including brain endothelial cells, CD8+ T cells, and erythrocytes (1‑4). Glut1 is a major glucose transporter that mediates glucose transport across the mammalian blood‑brain barrier.
Human Glut1 Antibody
R&D Systems | Catalog # MAB1418

Key Product Details
Validated by
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Met1-Val492
Accession # AAA52571
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human Glut1 Antibody
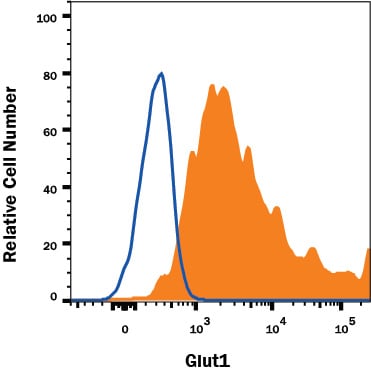
Detection of Glut1 in HepG2 Human Cell Line by Flow Cytometry.
HepG2 human hepatocellular carcinoma cell line was stained with Mouse Anti-Human Glut1 Monoclonal Antibody (Catalog # MAB1418, filled histogram) or isotype control antibody (Catalog # MAB0041, open histogram), followed by Allophycocyanin-conjugated Anti-Mouse IgG Secondary Antibody (Catalog # F0101B).Detection of Glut1 in Jurkat Human Cell Line by Flow Cytometry.
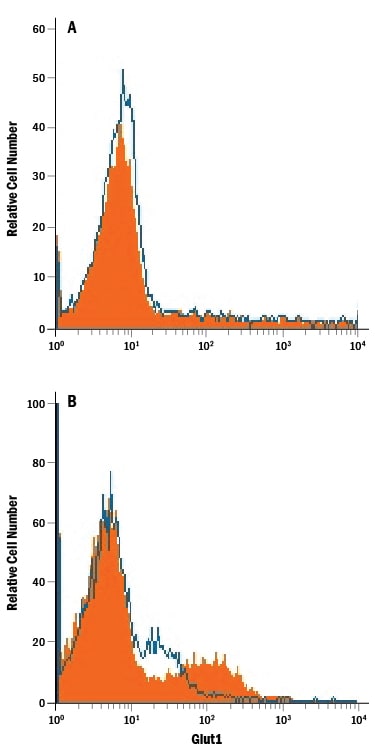
Jurkat human acute T cell leukemia cell line either (A) untreated or (B) cultured in nutrient-depleted media was stained with Mouse Anti-Human Glut1 Monoclonal Antibody (Catalog # MAB1418, filled histogram) or isotype control antibody (Catalog # MAB0041, open histogram), followed by Phycoerythrin-conjugated Anti-Mouse IgG Secondary Antibody (Catalog # F0102B).Glut1 in HepG2 Human Hepatocellular Carcinoma Cell Line.
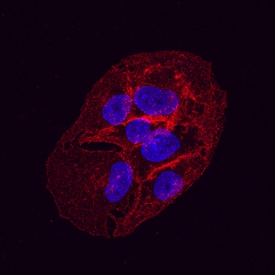
Glut1 was detected in immersion fixed HepG2 human hepatocellular carcinoma cell line using Mouse Anti-Human Glut1 Monoclonal Antibody (Catalog # MAB1418) at 10 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Mouse IgG Secondary Antibody (red; Catalog # NL007) and counterstained with DAPI(blue). Specific staining was localized to the plasma membrane. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Detection of Human Glut1 by Flow Cytometry
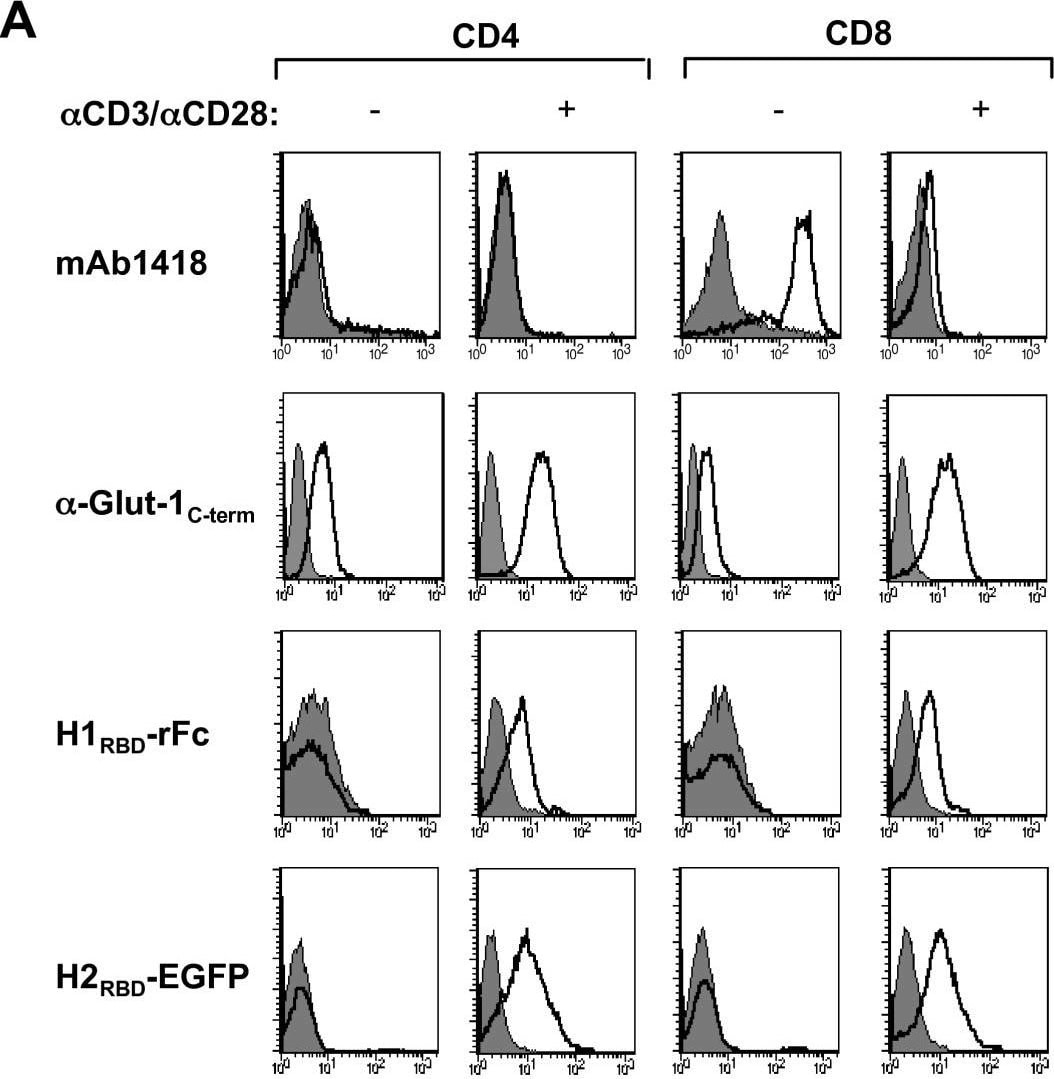
TCR stimulation results in Glut-1 expression and concomitant glucose uptake in CD4 and CD8 lymphocytes: Induction of H1RBD and H2RBD binding. CD4+ and CD8+ T lymphocytes were isolated by negative selection and stimulated via the TCR using alpha CD3/ alpha CD28 mAbs. (A) Non-activated and TCR-activated T cells were used for binding assays with mAb1418 followed by incubation with a FITC-conjugated alpha mouse IgG. Intracellular Glut-1 levels were monitored in permeabilized cells using the C-term Glut-1 polyclonal antibody followed by incubation with a FITC-conjugated sheep alpha rabbit IgG antibody. Filled histograms depict binding in the presence of the secondary FITC-conjugated antibody alone. Expression of the HTLV-1 Env receptor was detected by a 30 min incubation of the non-activated and TCR-activated cells with rabbit rFc-tagged H1RBD fusion protein at 37°C and binding was revealed by a 20 min incubation at 4°C with a FITC-conjugated sheep alpha rabbit IgG antibody. Binding to the H2RBD domain fused directly to EGFP (H2RBD-EGFP) was detected following a 30 min incubation at 37°C. (B) Glucose uptake was assayed by incubating non-activated and TCR-activated CD4 and CD8 T cells (1 × 106) with 2-deoxy-D [1-3H]glucose (2 μCi) for 45 min at 37°C. Uptake for each cell population is expressed as mean counts per minute (CPM) for triplicate samples, error bars indicate SD. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/17504522), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Glut1 by Flow Cytometry
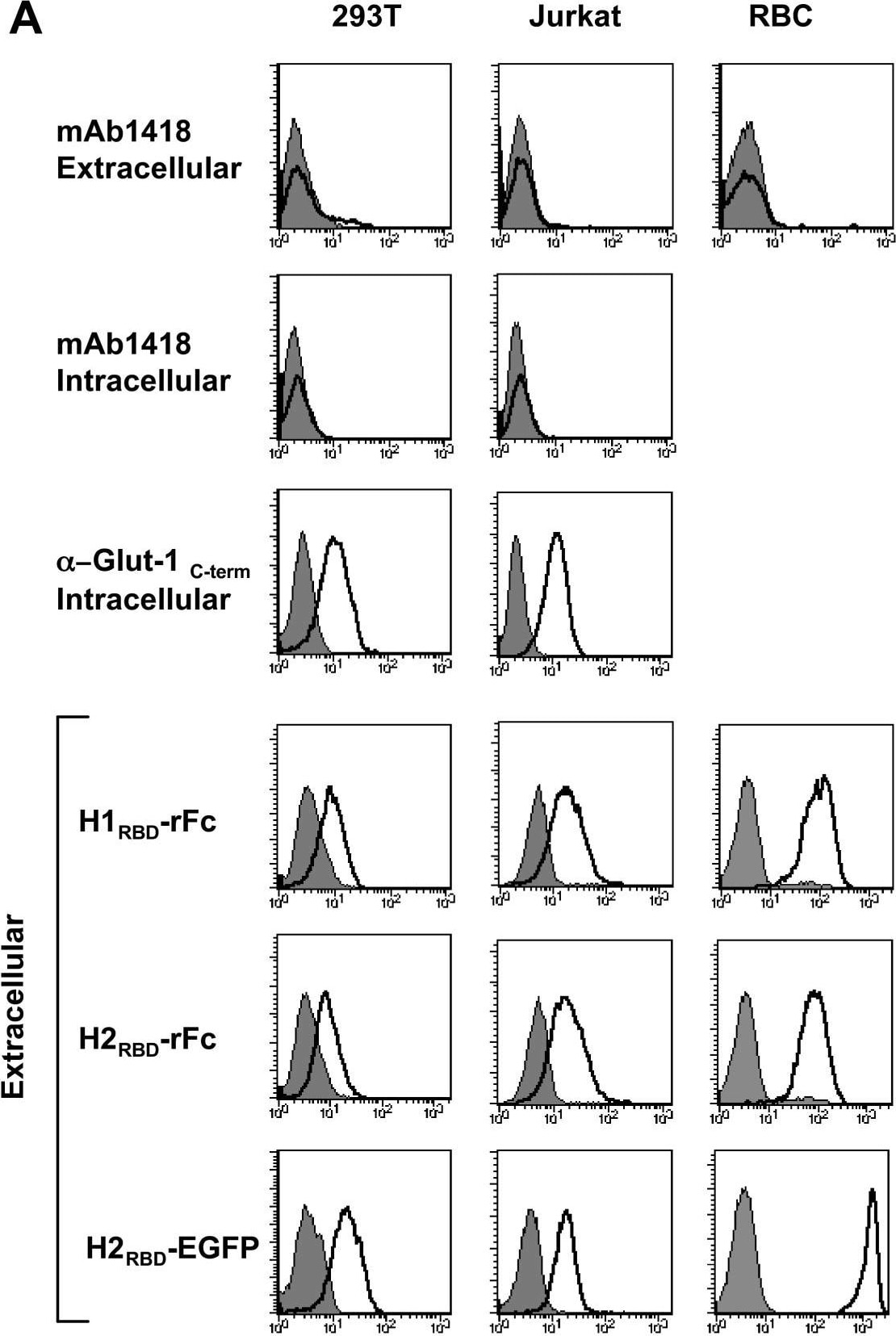
Endogenous Glut-1 expression in diverse cell types is not reflected by mAb1418 reactivity but correlates with binding of the HTLV-1 and HTLV-2 Env RBDs. (A) 293T, Jurkat and primary human erythrocytes were stained with mAb1418 and control binding with the secondary FITC-conjugated antibody is shown in all histograms (filled). Intracellular Glut-1 levels in permeabilized 293T and Jurkat cells were monitored with mAb1418 as well as the C-term polyclonal Glut-1 antibody. Expression of the HTLV-1 and HTLV-2 receptor was monitored by incubation of cells for 30 min at 37°C with the rFc-tagged H1RBD and H2RBD fusion proteins as well as the H2RBD-EGFP fusion protein. Binding is shown in solid line histograms whereas control immunofluorescence is shown in filled histograms. (B) Total Glut-1 protein levels in cell extracts from 293T, Jurkat and human erythrocytes were monitored by immunoblotting with an anti-C-term Glut-1 antibody. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/17504522), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Glut1 by Flow Cytometry
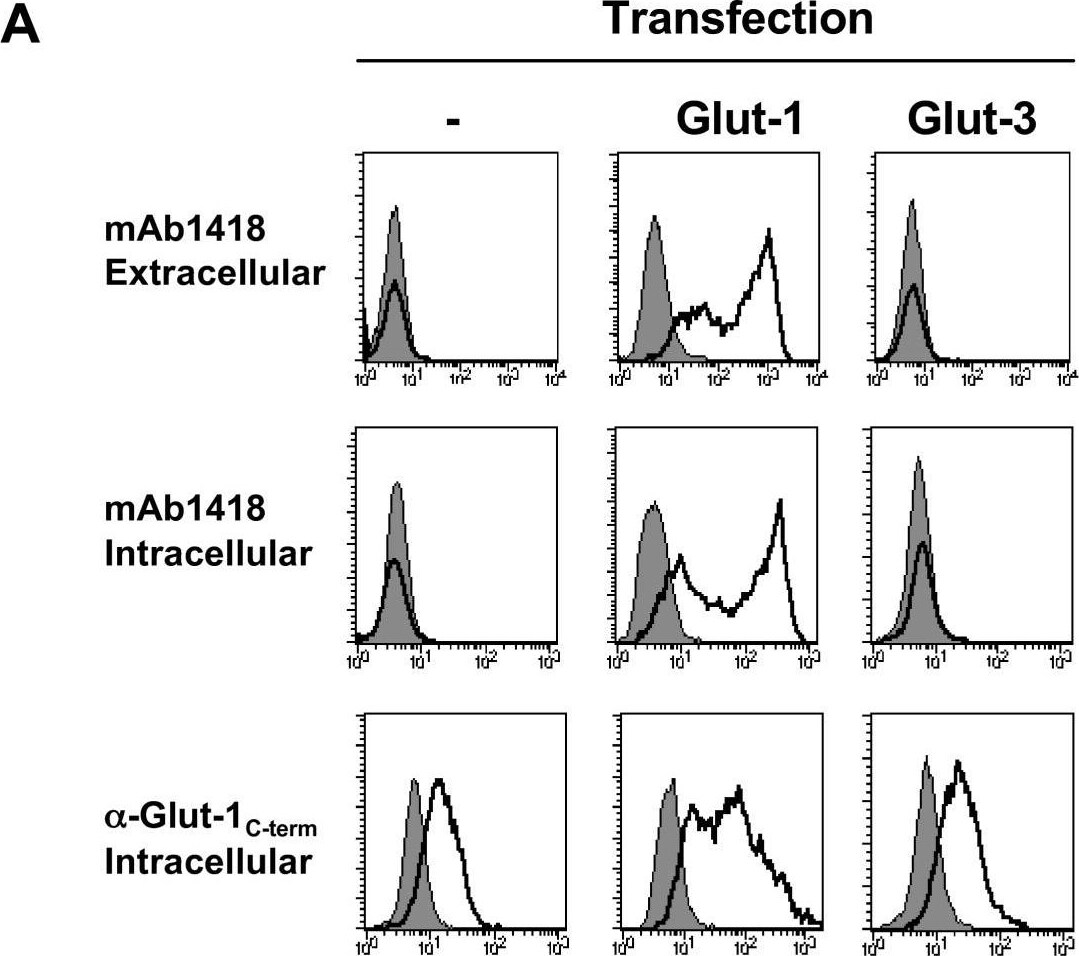
Antibody and HRBD binding following transfection of the Glut-1 and Glut-3 glucose transporters. (A) 293T cells were transfected with Glut-1 or Glut-3 expression vectors and assayed for binding to the mAb1418 and C-term anti-Glut-1 polyclonal Ab. The former stainings were performed on whole cells as well as permeabilized cells to determine cell surface and total binding, respectively. All stainings using the anti-Glut-1 pAb were performed on permeabilized cells as the recognized epitope is intracellular. Staining was performed at 4°C. Specific binding and background fluorescence due to the secondary conjugated Ab are indicated in solid line and filled histograms, respectively. (B) Control and transfected 293T cells were incubated with rFc-tagged H1RBD and H2RBD fusion proteins for 30 min at 37°C followed by incubation with a FITC-conjugated alpha rabbit-Fc antibody at 4°C. Direct binding to H2RBD was demonstrated by incubation of cells with an EGFP-tagged envelope (H2RBD-EGFP). Binding is shown in solid line histograms whereas control immunofluorescence is shown in filled histograms. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/17504522), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Glut1 by Flow Cytometry
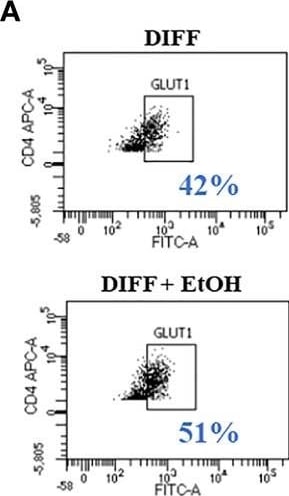
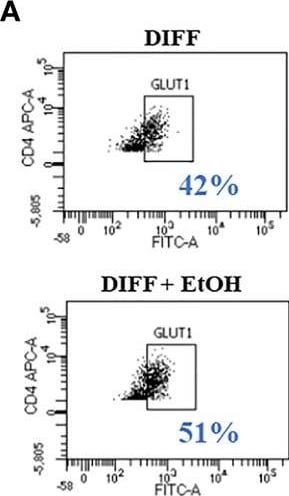
Effect of ethanol on CD4+ T cell glucose metabolism. (A) Representative FACS plots of GLUT1 expression in DIFF and DIFF + EtOH CD4+ T cells. (B) Ethanol increased CD4+ T cell GLUT1 expression. (C) Representative FACS histograms of 2-NBDG uptake in DIFF and DIFF + EtOH CD4+ T cells. (D) Ethanol increased glucose uptake within differentiated CD4+ T cells. (E) All experimental groups were analyzed using the Glycolytic Stress test. Ethanol increased glycolysis (F) and glycolytic capacity (G) within differentiated groups. MFI = mean fluorescent intensity, 2-NBDG = 2-Deoxy-2-[(7-nitro-2,1,3-benzoxadiazol-4-yl)amino]-D-glucose, ECAR = extracellular acidification rate, Naïve = undifferentiated and no ethanol treatment, EtOH = undifferentiated and ethanol-treated, DIFF = differentiated and no ethanol treatment, and DIFF + EtOH = differentiated and ethanol-treated. Data represents average values using CD4+ T cells from 6 donors expressed as mean ± SEM. Significant differences (p<0.05) were determined by repeated measures 2-way ANOVA. *p ≤ 0.05. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35634279), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Glut1 by Flow Cytometry
Effect of ethanol on CD4+ T cell glucose metabolism. (A) Representative FACS plots of GLUT1 expression in DIFF and DIFF + EtOH CD4+ T cells. (B) Ethanol increased CD4+ T cell GLUT1 expression. (C) Representative FACS histograms of 2-NBDG uptake in DIFF and DIFF + EtOH CD4+ T cells. (D) Ethanol increased glucose uptake within differentiated CD4+ T cells. (E) All experimental groups were analyzed using the Glycolytic Stress test. Ethanol increased glycolysis (F) and glycolytic capacity (G) within differentiated groups. MFI = mean fluorescent intensity, 2-NBDG = 2-Deoxy-2-[(7-nitro-2,1,3-benzoxadiazol-4-yl)amino]-D-glucose, ECAR = extracellular acidification rate, Naïve = undifferentiated and no ethanol treatment, EtOH = undifferentiated and ethanol-treated, DIFF = differentiated and no ethanol treatment, and DIFF + EtOH = differentiated and ethanol-treated. Data represents average values using CD4+ T cells from 6 donors expressed as mean ± SEM. Significant differences (p<0.05) were determined by repeated measures 2-way ANOVA. *p ≤ 0.05. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35634279), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human Glut1 Antibody
CyTOF-ready
Flow Cytometry
Sample: HepG2 human hepatocellular carcinoma cell line (untreated) and Jurkat human acute T cell leukemia cell line cultured in nutrient-depleted media
Immunocytochemistry
Sample: Immersion fixed HepG2 human hepatocellular carcinoma cell line
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Glut1
References
- Mueckler, M. et al. 1994, Eur. J. Biochem. 219:713.
- Meuckler, M. et al. 1985, Science 229:941.
- Jones, K.S. et al. 2006, J. Virol. 8291.
- Takenouchi, N. et al. 2007, J. Virol. 1506.
- Kinet, S. et al. 2007, Retrovirology 4:31.
Long Name
Alternate Names
Gene Symbol
UniProt
Additional Glut1 Products
Product Documents for Human Glut1 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Glut1 Antibody
For research use only
Related Research Areas
Citations for Human Glut1 Antibody
Customer Reviews for Human Glut1 Antibody
There are currently no reviews for this product. Be the first to review Human Glut1 Antibody and earn rewards!
Have you used Human Glut1 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- View all Protocols, Troubleshooting, Illustrated assays and Webinars