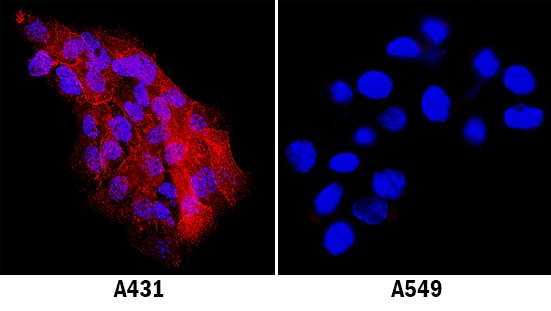
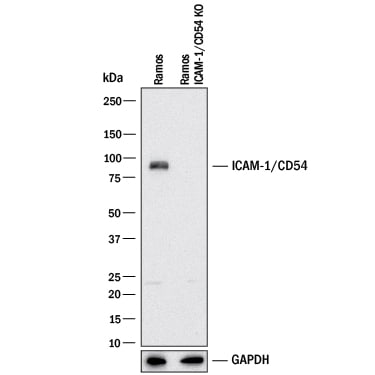
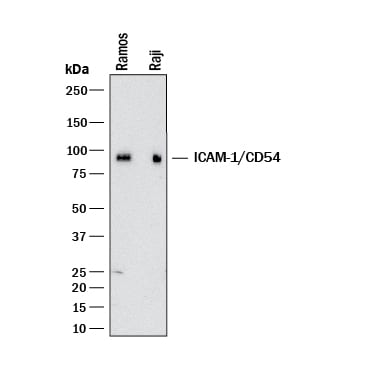
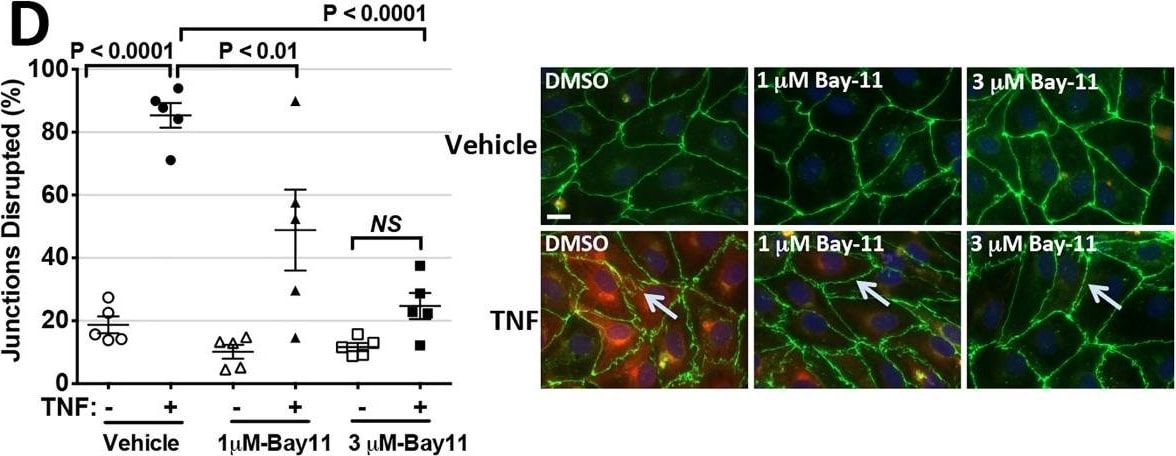
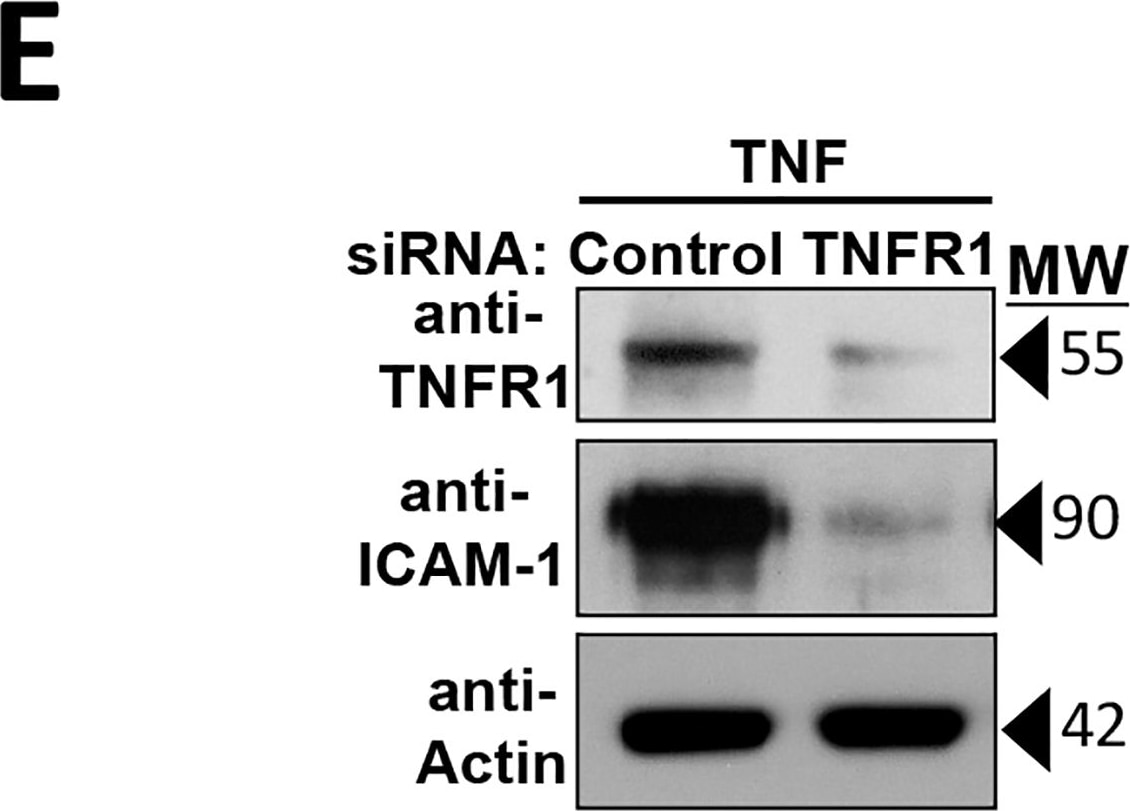
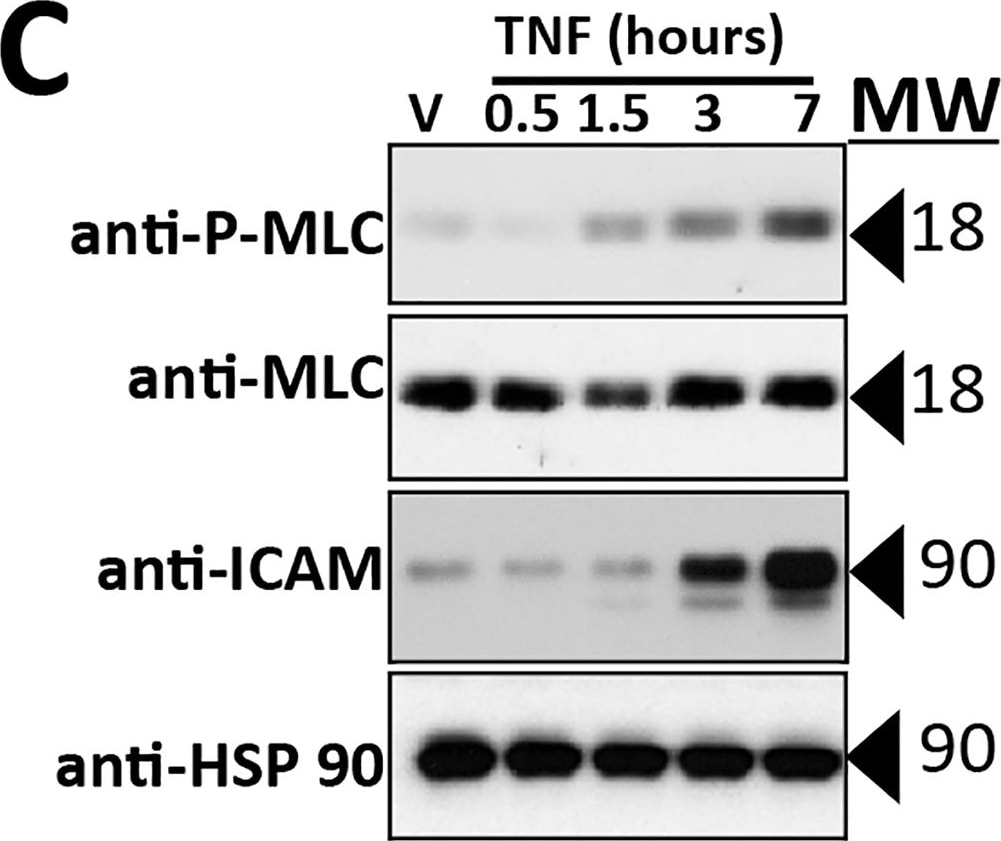
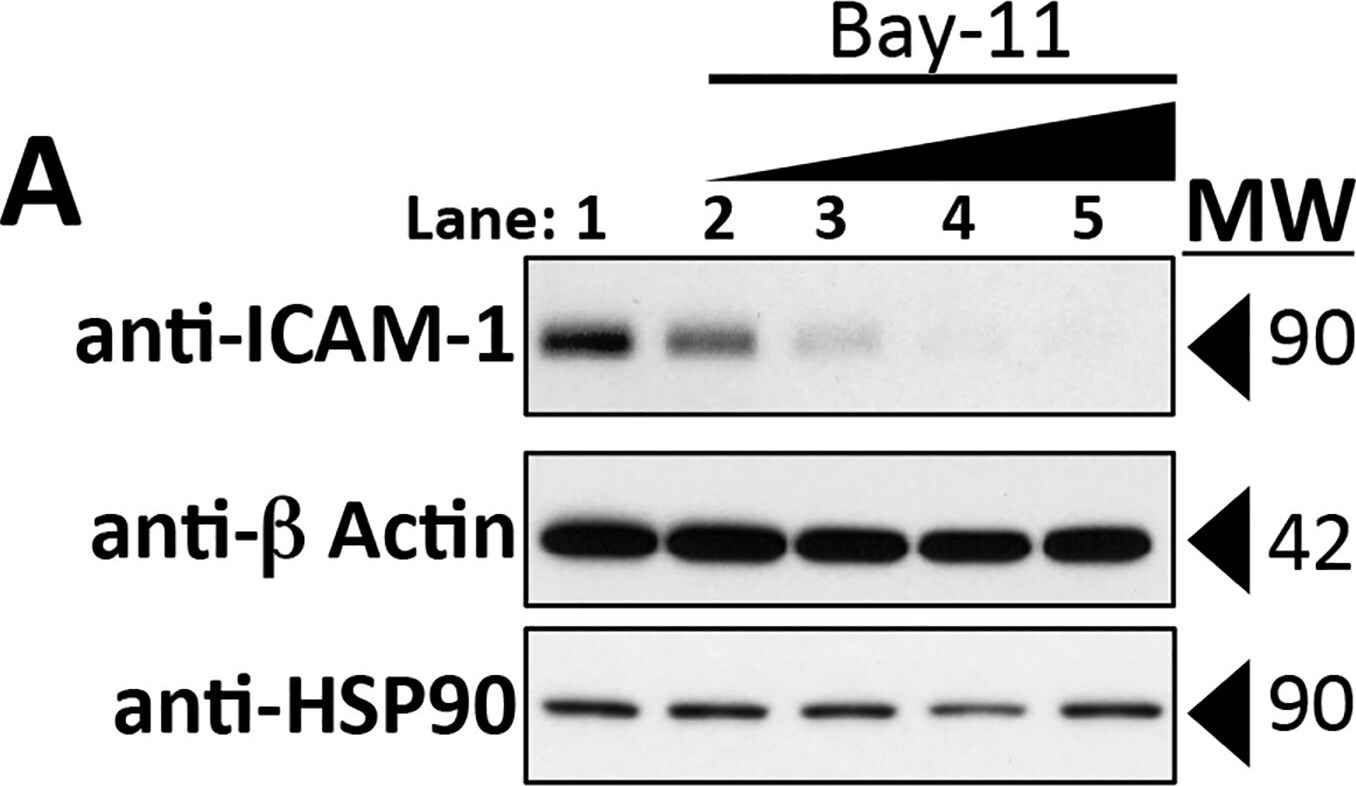
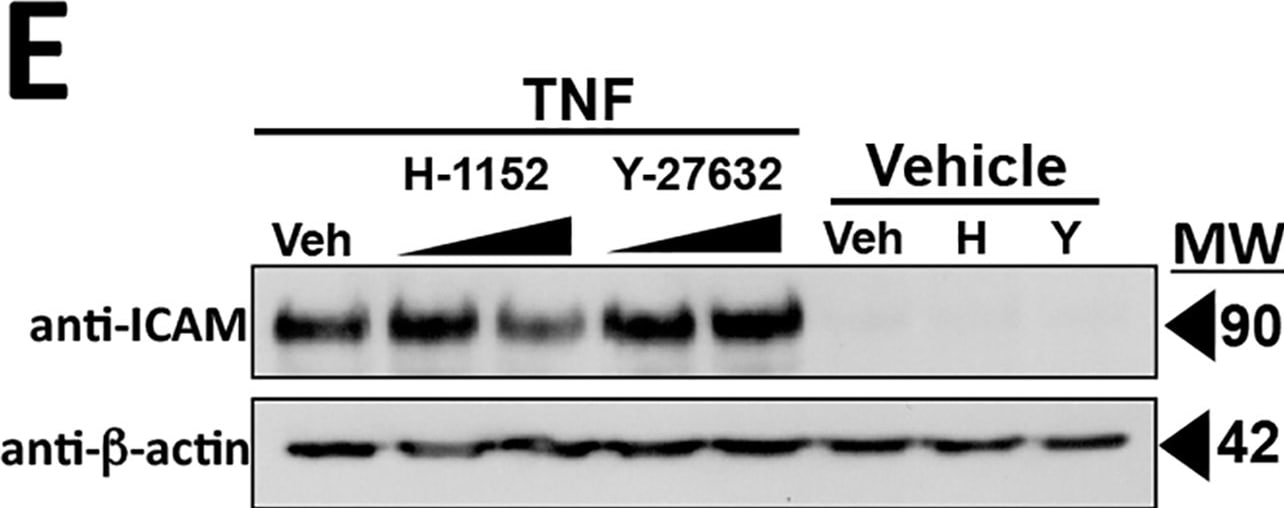
Intercellular Adhesion Molecule-1 (ICAM-1, CD54) binds the leukocyte integrins LFA-1 and Mac-1. ICAM-1 expression is weak on leukocytes, epithelial and resting endothelial cells, as well as some other cell types, but expression can be stimulated by IFN-gamma, TNF-alpha, IL-1 beta and LPS.
Soluble ICAM-1 is found in a biologically active form in serum, probably as a result of proteolytic cleavage from the cell surface, and is elevated in patients with various inflammatory syndromes such as septic shock, LAD, cancer and transplantation.