IFITM3 (IFN-inducible tranemembrane protein 3; also IFN-inducible protein 1-8U and fragilis) is a 14.6 kDa (predicted) member of the CD225 family of proteins. It is expressed on primordial germ cells and select tumor types, and appears to regulate cell migration. Human IFITM3 is a 133 amino acid (aa), two-transmembrene protein that contains an N-terminal (aa 1-57) and a C-terminal (aa 108-128) extracellular domain. Over aa 3-57, human IFITM3 is 59% and 56% aa identical to bovine and mouse IFITM3, respectively. Over aa 3-57 human IFITM3 is 88% identical to human IFITM2.
Human IFITM2/IFITM3 Antibody
R&D Systems | Catalog # AF4834

Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human
Applications
Validated:
Western Blot
Cited:
Western Blot, Immunocytochemistry
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
E. coli-derived human IFITM3
His3-His57
Accession # Q01628
His3-His57
Accession # Q01628
Specificity
Detects human IFITM2 and human IFITM3 in direct ELISAs and Western blots. In direct ELISAs, less than 4% cross-reactivity with recombinant human IFITM1 is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human IFITM2/IFITM3 Antibody
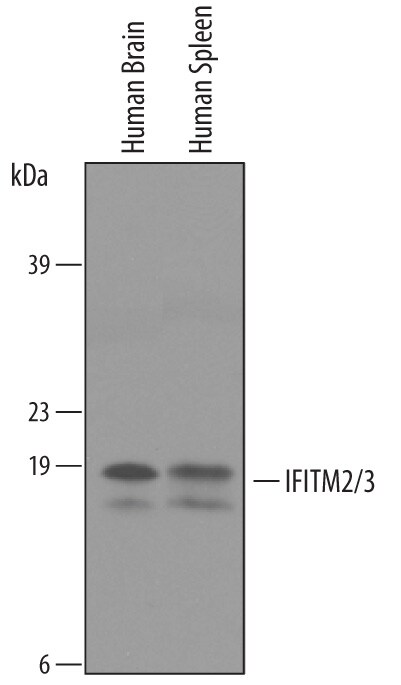
Detection of Human IFITM2/IFITM3/Fragilis by Western Blot.
Western blot shows lysates of human brain tissue and human spleen tissue. PVDF membrane was probed with 1 µg/mL of Goat Anti-Human IFITM2/3 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF4834) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF019). Specific bands were detected for IFITM2 and IFITM3 at approximately 15 and 17 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 8.Detection of IFITM2/IFITM3 by Western Blot
Native FL IFITM3 and delta 21 IFITM3 restrict IAVs.(A) A549 cells transduced with the vector alone or with tet-inducible native FL IFITM3 or delta 21 IFITM3 were treated with the indicated concentrations of doxycycline or interferon-beta. Two days later, cells were infected by MLV-GFP pseudotyped with the indicated viral entry glycoproteins. Infected cells were harvested, fixed with formaldehyde, and analyzed by flow cytometry 48 hours after infection. The relative infectivity was determined as the percentage of GFP positive cells normalized to that of vector-transduced A549 cells. (B) The same aliquots of cells used in (A) were incubated with infectious influenza A/PR/8/34 (H1N1) virus at an M.O.I. of 1 or with influenza A/England/42/72 (H3N2) virus at an M.O.I of 0.5. Infected cells were labeled with anti-H1 or anti-H3 antibodies and with an PE-conjugated secondary antibody. Cells were analyzed by flow cytometry 16 hours after infection and the relative infectivity was determined as the percentage of PE positive cells normalized to that of vector-transduced A549 cells. (C) Experiments were similar to that in (B) except that cells were incubated with infectious influenza A/Virginia/ATCC3/2009 (H1N1) virus at an M.O.I. of 1. (D) The same aliquots of cells used in (A) or (B) were analyzed by western blotting. The expression of IFITM3 isoforms was measured using the indicated antibodies. The results are the averages of three independent infection replicates. The error bars indicate standard deviations. Each panel represents at least two sets of experiments with similar results. * indicates statistical significance (p<0.03 by Student's t test) as compared with vector-transduced cells. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/25314048), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of IFITM2/IFITM3 by Western Blot
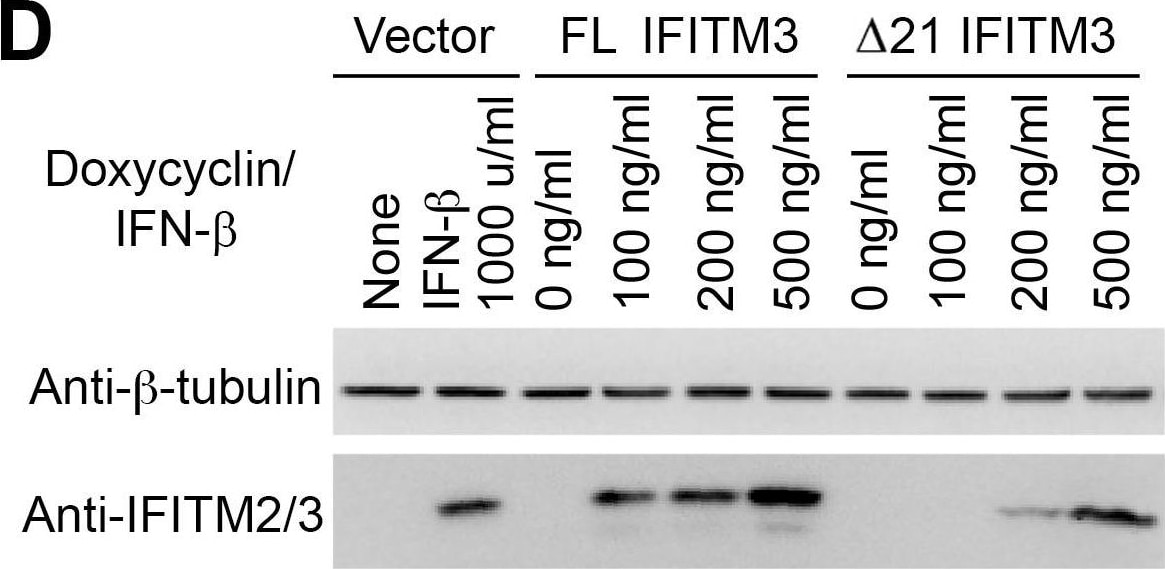
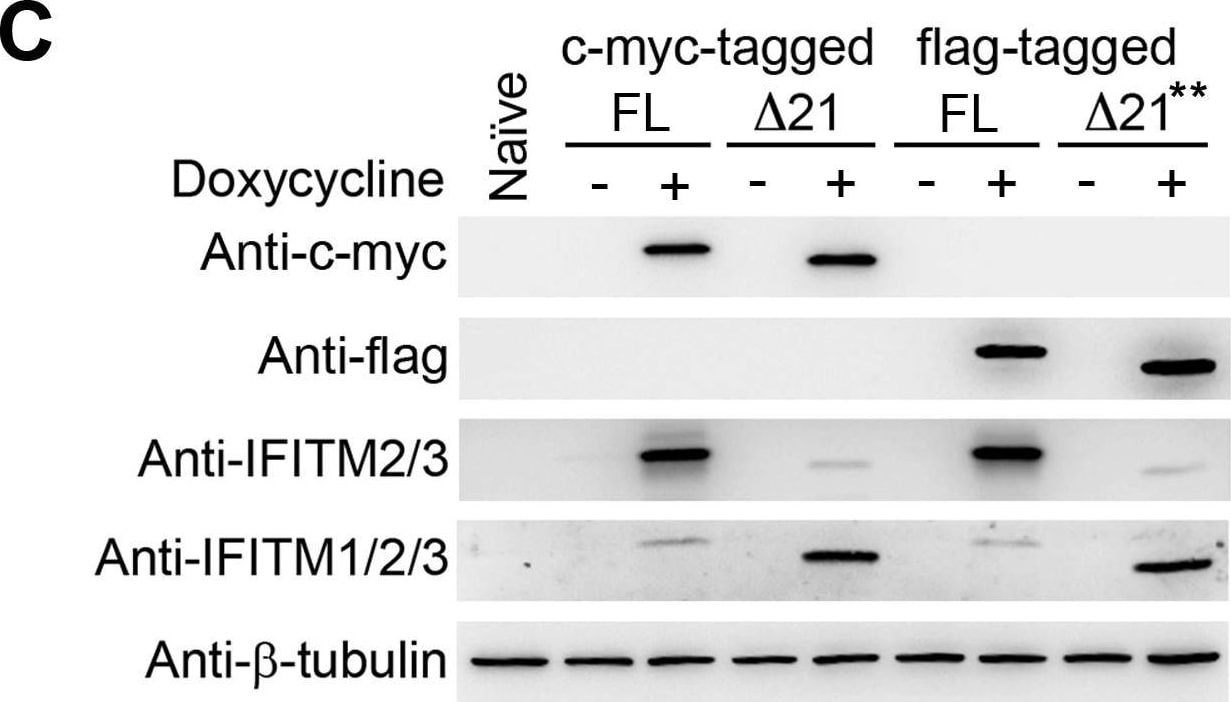
Epitope-tagged FL IFITM3 and delta 21 IFITM3 restrict IAVs.Naïve A549 cells and A549 cells transduced with tet-inducible c-myc-tagged (A), flag-tagged (B) FL IFITM3 or delta 21 IFITM3 were treated with or without 1000 ng/ml doxycycline (** 600 ng/ml doxycycline). Two days later, cells were infected with MLV-GFP pseudotyped with the indicated viral entry glycoproteins. Infected cells were harvested, fixed with formaldehyde, and analyzed by flow cytometry 48 hours after infection. The relative infectivity was determined as the percentage of GFP positive cells normalized to that of naïve A549 cells. (C) The same aliquots of cells used in (A) or (B) were analyzed by western blotting. The expression of IFITM3 isoforms was measured using the indicated antibodies. The expression of beta –tubulin was used as our loading controls. The results are the averages of three independent infection replicates. The error bars indicate standard deviations. Each panel represents at least two sets of experiments with similar results. * indicates statistical significance (p<0.03 by Student's t test) as compared with naïve cells. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/25314048), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of IFITM2/IFITM3 by Western Blot
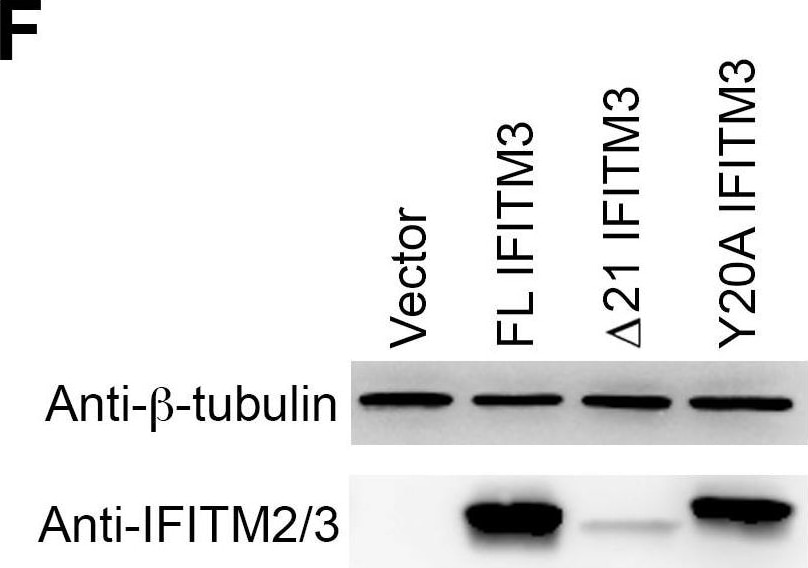
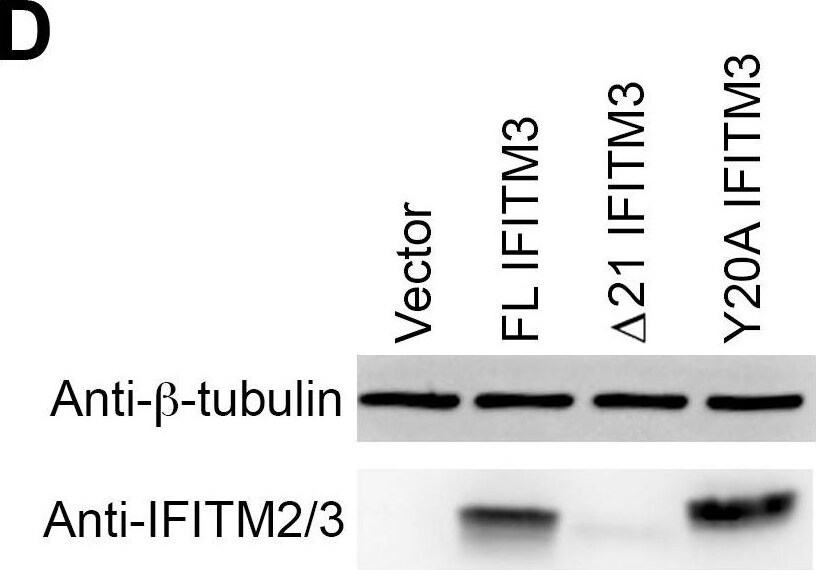
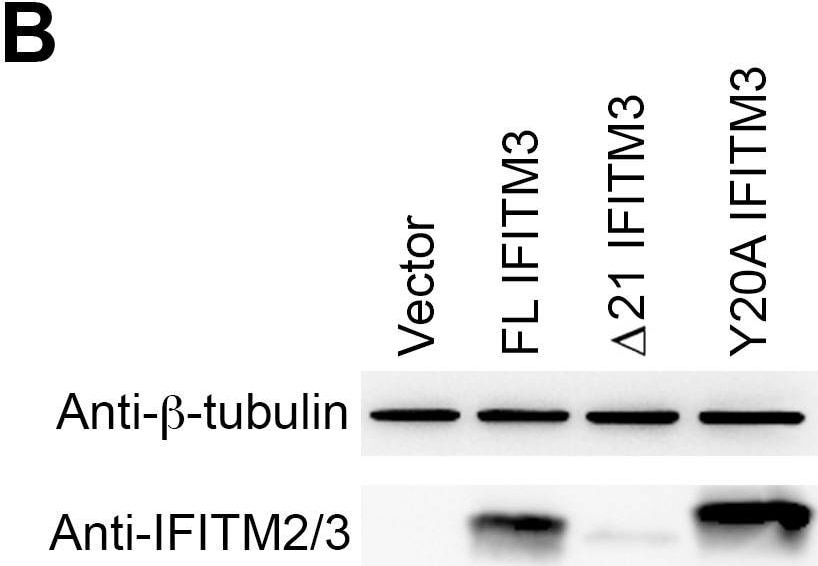
Y20A IFITM3 restricts IAV entry.A549 (A), 293T (C), or Vero E6 (E) cells transduced with vector alone or to express FL IFITM3, delta 21 IFITM3, or Y20A IFITM3 were incubated with MLV-GFP pseudotyped with the indicated viral entry glycoproteins. Infected cells were harvested, fixed with formaldehyde, and analyzed by flow cytometry 48 hours after infection. The relative infectivity was determined as the percentage of GFP positive cells normalized to that of vector-transduced A549 cells. Expression of the indicated IFITM3 variants in A549 (B), 293T (D), or Vero E6 (F) cells were detected by western blotting using the indicated antibodies. The results are the averages of three independent infection replicates. The error bars indicate standard deviations. Each panel represents at least two sets of experiments with similar results. * indicates statistical significance (p<0.03 by Student's t test) as compared with vector-transduced cells. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/25314048), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of IFITM2/IFITM3 by Western Blot
Y20A IFITM3 restricts IAV entry.A549 (A), 293T (C), or Vero E6 (E) cells transduced with vector alone or to express FL IFITM3, delta 21 IFITM3, or Y20A IFITM3 were incubated with MLV-GFP pseudotyped with the indicated viral entry glycoproteins. Infected cells were harvested, fixed with formaldehyde, and analyzed by flow cytometry 48 hours after infection. The relative infectivity was determined as the percentage of GFP positive cells normalized to that of vector-transduced A549 cells. Expression of the indicated IFITM3 variants in A549 (B), 293T (D), or Vero E6 (F) cells were detected by western blotting using the indicated antibodies. The results are the averages of three independent infection replicates. The error bars indicate standard deviations. Each panel represents at least two sets of experiments with similar results. * indicates statistical significance (p<0.03 by Student's t test) as compared with vector-transduced cells. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/25314048), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of IFITM2/IFITM3 by Western Blot
Y20A IFITM3 restricts IAV entry.A549 (A), 293T (C), or Vero E6 (E) cells transduced with vector alone or to express FL IFITM3, delta 21 IFITM3, or Y20A IFITM3 were incubated with MLV-GFP pseudotyped with the indicated viral entry glycoproteins. Infected cells were harvested, fixed with formaldehyde, and analyzed by flow cytometry 48 hours after infection. The relative infectivity was determined as the percentage of GFP positive cells normalized to that of vector-transduced A549 cells. Expression of the indicated IFITM3 variants in A549 (B), 293T (D), or Vero E6 (F) cells were detected by western blotting using the indicated antibodies. The results are the averages of three independent infection replicates. The error bars indicate standard deviations. Each panel represents at least two sets of experiments with similar results. * indicates statistical significance (p<0.03 by Student's t test) as compared with vector-transduced cells. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/25314048), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human IFITM2/IFITM3 Antibody
Application
Recommended Usage
Western Blot
1 µg/mL
Sample: Human brain tissue and human spleen tissue
Sample: Human brain tissue and human spleen tissue
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: IFITM2/IFITM3
Alternate Names
1-8D;1-8U;DSPA2b;DSPA2c;IFITM2;IFITM3;IP15
UniProt
Additional IFITM2/IFITM3 Products
Product Documents for Human IFITM2/IFITM3 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human IFITM2/IFITM3 Antibody
For research use only
Related Research Areas
Citations for Human IFITM2/IFITM3 Antibody
Customer Reviews for Human IFITM2/IFITM3 Antibody
There are currently no reviews for this product. Be the first to review Human IFITM2/IFITM3 Antibody and earn rewards!
Have you used Human IFITM2/IFITM3 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...