Interleukin 16, also named lymphocyte chemoattractant factor (LCF), was originally identified as a CD8+ T-cell-derived chemoattractant for CD4+ cells. The biologically active form of IL-16 was originally proposed to be a homotetramer of 14 kDa chains containing 130 amino acid residue subunits. The complete pro-IL-16 cDNA was subsequently cloned and shown to encode a 631 amino acid residue hydrophilic protein that lacked a signal peptide. The original 130 amino acid residue polypeptide is now believed to have been derived from the C terminus of the precursor. IL-16 precursor protein has been detected in the lysates of various cells including mitogen stimulated PBMCs. The biologically active and secreted natural IL-16 is assumed to be a proteolytic cleavage product of pro-IL-16 generated by proteases present in or on activated CD8+ cells. A likely cleavage site was proposed to be at aspartate residue 510. This would yield a 121 amino acid residue protein, smaller than the 130 aa residue protein first described. The expression of IL-16 precursor mRNA has been detected in various tissues including spleen, thymus, lymph nodes, peripheral leukocytes, bone marrow and cerebellum. The gene for IL-16 precursor has been localized to chromosome 15. The biological activities ascribed to IL-16 are reported to be dependent on the cell surface expression of CD4, suggesting that IL-16 is a CD4 ligand. Besides its chemotactic properties, IL-16 has also been shown to suppress HIV-1 replication in vitro. Recombinant E. coli-derived IL-16 produced at R&D Systems is present mostly as a monomer, exhibits chemotactic activity for lymphocytes at high concentrations, lacks chemotactic activites for monocytes, and binds the extracellular domain of CD4 with low affinity.


Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human
Applications
Validated:
Western Blot, ELISA Capture (Matched Antibody Pair), Simple Western
Cited:
Western Blot, Luminex Development
Label
Unconjugated
Antibody Source
Monoclonal Mouse IgG1 Clone # 70719
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant human IL-16 isoform 1
Met1203-Ser1332
Accession # Q14005
Met1203-Ser1332
Accession # Q14005
Specificity
Detects human IL-16 in direct ELISAs and Western blots.
Clonality
Monoclonal
Host
Mouse
Isotype
IgG1
Scientific Data Images for Human IL‑16 Antibody
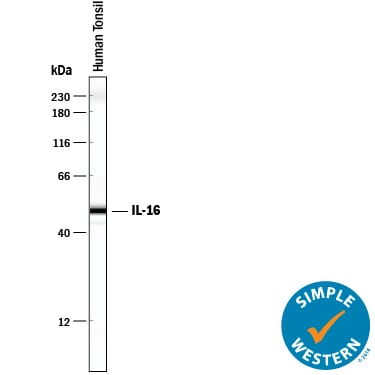
Detection of Human IL‑16 by Western Blot.
Western blot shows lysate of human tonsil tissue. PVDF membrane was probed with 2 µg/mL of Mouse Anti-Human IL-16 Monoclonal Antibody (Catalog # MAB316) followed by HRP-conjugated Anti-Mouse IgG Secondary Antibody (Catalog # HAF018). Specific bands were detected for IL-16 at approximately 45-55 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Detection of Human IL‑16 by Simple WesternTM.
Simple Western lane view shows lysates of human tonsil tissue, loaded at 0.2 mg/mL. A specific band was detected for IL‑16 at approximately 50 kDa (as indicated) using 20 µg/mL of Mouse Anti-Human IL‑16 Monoclonal Antibody (Catalog # MAB316). This experiment was conducted under reducing conditions and using the 12-230 kDa separation system.Applications for Human IL‑16 Antibody
Application
Recommended Usage
Simple Western
20 µg/mL
Sample: Human tonsil tissue
Sample: Human tonsil tissue
Western Blot
2 µg/mL
Sample: Human tonsil tissue
Sample: Human tonsil tissue
Human IL-16 Sandwich Immunoassay
Please Note: Optimal dilutions of this antibody should be experimentally determined.
Reviewed Applications
Read 2 reviews rated 4.5 using MAB316 in the following applications:
Formulation, Preparation, and Storage
Purification
Protein A or G purified from ascites
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: IL-16
References
- Cruikshank, W.W. et al. (1994) Proc. Natl. Acad. Sci. USA 91:5109.
- Baier, M. et al. (1997) Proc. Natl. Acad. Sci. USA 94:5273.
- Zhou, A. et al. (1997) Nature Medicine 3:659.
- Bazan, J.F. and T.J. Schall (1996) Nature 381:29.
Long Name
Interleukin 16
Alternate Names
IL16, LCF
Gene Symbol
IL16
UniProt
Additional IL-16 Products
Product Documents for Human IL‑16 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human IL‑16 Antibody
For research use only
Related Research Areas
Citations for Human IL‑16 Antibody
Customer Reviews for Human IL‑16 Antibody (2)
4.5 out of 5
2 Customer Ratings
Have you used Human IL‑16 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
2 of
2 reviews
Showing All
Filter By:
-
Application: ELISASample Tested: Serum and PlasmaSpecies: HumanVerified Customer | Posted 08/20/2021Works as the capture in an ELISA.
-
Application: ELISASample Tested: Serum and PlasmaSpecies: HumanVerified Customer | Posted 11/16/2017A sandwich ELISA was made with MAB316 as the capture and biotinylated AF-316 as the detection using 316-IL as the immunoassay calibrator.
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...