The heat shock protein-90 kDa (HSP90) is a composite name for a large group of genes whose molecular weights average 90 kDa. HSP90 functions primarily as a molecular chaperone, facilitating the folding of other cellular proteins, preventing protein aggregation, or targeting improperly folded proteins to specific degradative pathways. HSP90 is ubiquitously expressed, highly conserved, and accounts for 1‑2% of the total cellular protein. Recently introduced, standardized nomenclature has divided the 17 identified HSP90 genes into three related and one unrelated classes, HSP90aa, HSP90AB, HSP90BB, and TRAP, respectively. Six of these genes were functional while the remaining 11 are considered putative pseudogenes. Eukaryotic cells have two principal isoforms of HSP90. The antibody described here was raised against the inducible form, HSP90AA1, a 732 amino acid protein also known as HSP90-alpha, HSP90A, HSPC1, HSPCA, HSP89-alpha and LAP2. The constitutively expressed HSP90AB1-isoform 1 is a 724 amino acid protein that is also known as HSP90-beta, HSP90B, HSPCB, HSPC2, and HSP89-beta. HSP90AB1-1 and HSP90AA share 90% identity. In addition to its role as a molecular chaperone and stress response protein, HSP90 is a central component in a number of basic cellular processes including hormone signaling and cell cycle control.
Human/Mouse/Rat HSP90 Antibody
R&D Systems | Catalog # AF3775


Key Product Details
Species Reactivity
Human, Mouse, Rat
Applications
Western Blot, Simple Western
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant human HSP90 alpha
Met1-Asp732
Accession # P07900
Met1-Asp732
Accession # P07900
Specificity
Detects human, mouse, and rat HSP90 in Western blots. Detects recombinant human HSP90 alpha and HS90 beta in Western blots.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human/Mouse/Rat HSP90 Antibody
Detection of Human/Mouse/Rat HSP90 by Western Blot.
Western blot shows lysates of Jurkat human acute T cell leukemia cell line, A20 mouse B cell lymphoma cell line, and L6 rat myoblast cell line. PVDF membrane was probed with 0.5 µg/mL Human/Mouse/Rat HSP90 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3775) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF017). For additional reference, recombinant human HSP90a and HSP90b (5 ng/lane) were included. A specific band for HSP90 was detected at approximately 96 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 2.Detection of Human HSP90 by Simple WesternTM.
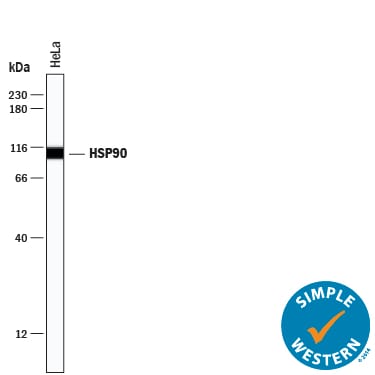
Simple Western lane view shows lysates of HeLa human cervical epithelial carcinoma cell line, loaded at 0.2 mg/mL. A specific band was detected for HSP90 at approximately 100 kDa (as indicated) using 5 µg/mL of Goat Anti-Human/Mouse/Rat HSP90 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3775) followed by 1:50 dilution of HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF019). This experiment was conducted under reducing conditions and using the 12-230 kDa separation system.Applications for Human/Mouse/Rat HSP90 Antibody
Application
Recommended Usage
Simple Western
5 µg/mL
Sample: HeLa human cervical epithelial carcinoma cell line
Sample: HeLa human cervical epithelial carcinoma cell line
Western Blot
0.5 µg/mL
Sample: Jurkat human acute T cell leukemia cell line, A20 mouse B cell lymphoma cell line, and L6 rat myoblast cell line
Sample: Jurkat human acute T cell leukemia cell line, A20 mouse B cell lymphoma cell line, and L6 rat myoblast cell line
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: HSP90
Long Name
Heat Shock Protein 90
Alternate Names
FLJ31884, Heat shock 86 kDa, heat shock 90kD protein 1, alpha, heat shock 90kD protein 1, alpha-like 4, heat shock 90kD protein, alpha-like 4, heat shock 90kDa protein 1, alpha, heat shock protein 90kDa alpha (cytosolic), class A member 1, heat shock protein HSP 90-alpha, Hsp89, Hsp90, HSP90AHSP86, HSP90N, HSPC1HSP 86, HSPCAHSP89A, HSPCAL1, HSPCAL4, HSPN, LAP2, Renal carcinoma antigen NY-REN-38
UniProt
Additional HSP90 Products
Product Documents for Human/Mouse/Rat HSP90 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human/Mouse/Rat HSP90 Antibody
For research use only
Related Research Areas
Citations for Human/Mouse/Rat HSP90 Antibody
Customer Reviews for Human/Mouse/Rat HSP90 Antibody
There are currently no reviews for this product. Be the first to review Human/Mouse/Rat HSP90 Antibody and earn rewards!
Have you used Human/Mouse/Rat HSP90 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways