Mouse Progranulin, also known as Proepithelin, PC Cell-derived Growth Factor and Acrogranin, is a secreted protein of 572 amino acid residues. It contains seven and one half granulin repeats which are characterized by 6 disulfide bridges. Progranulin can be converted in vivo to granulins by proteolysis. Progranulin is a mitogen that stimulates proliferation and survival of various epithelial, mesenchymal and tumor cells. Several granulins have also been shown to be biologically active and may play a role in inflammation and wound healing. Mouse Progranulin shares 86% and 75% amino acid sequence homology with rat and human Progranulin, respectively.
Mouse Progranulin/PGRN Antibody
R&D Systems | Catalog # AF2557

Key Product Details
Validated by
Biological Validation
Species Reactivity
Validated:
Mouse
Cited:
Human, Mouse, Rat, Transgenic Mouse
Applications
Validated:
Immunohistochemistry, Western Blot, Immunocytochemistry
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Immunohistochemistry-Frozen, Western Blot, Immunofluorescence, Immunocytochemistry, Immunoprecipitation, ELISA Capture, ELISA Development (Detection), In vivo assay
Label
Unconjugated
Antibody Source
Polyclonal Sheep IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant mouse Progranulin
Thr18-Leu589
Accession # P28798
Thr18-Leu589
Accession # P28798
Specificity
Detects mouse Progranulin in direct ELISAs and Western blots. In Western blots, approximately 15% cross-reactivity with recombinant human Progranulin is observed.
Clonality
Polyclonal
Host
Sheep
Isotype
IgG
Scientific Data Images for Mouse Progranulin/PGRN Antibody
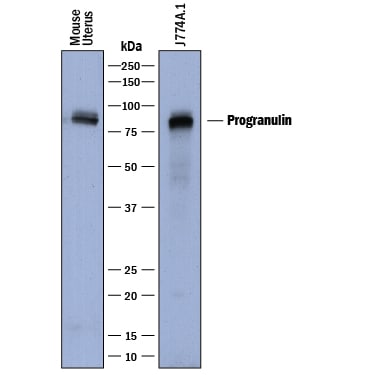
Detection of Mouse Progranulin/PGRN by Western Blot.
Western blot shows lysates of mouse uterus tissue and J774A.1 mouse reticulum cell sarcoma macrophage cell line. PVDF membrane was probed with 0.5 µg/mL of Sheep Anti-Mouse Progranulin/PGRN Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2557) followed by HRP-conjugated Anti-Sheep IgG Secondary Antibody (Catalog # HAF016). A specific band was detected for Progranulin/PGRN at approximately 85-90 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Progranulin/PGRN in Mouse Splencytes.
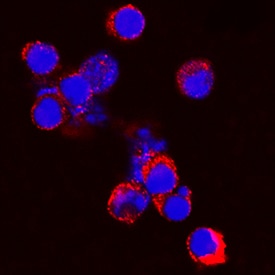
Progranulin/PGRN was detected in immersion fixed mouse splenocytes using Sheep Anti-Mouse Progranulin/PGRN Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2557) at 5 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Sheep IgG Secondary Antibody (red; Catalog # NL010) and counterstained with DAPI (blue). Specific staining was localized to cytoplasm. View our protocol for Fluorescent ICC Staining of Non-adherent Cells.Progranulin in Mouse Kidney.
Progranulin was detected in immersion fixed frozen sections of mouse kidney using Sheep Anti-Mouse Progranulin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2557) at 15 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Sheep HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS019) and counterstained with hematoxylin (blue). View our protocol for Chromogenic IHC Staining of Frozen Tissue Sections.Detection of Mouse Progranulin/PGRN by Immunocytochemistry/Immunofluorescence
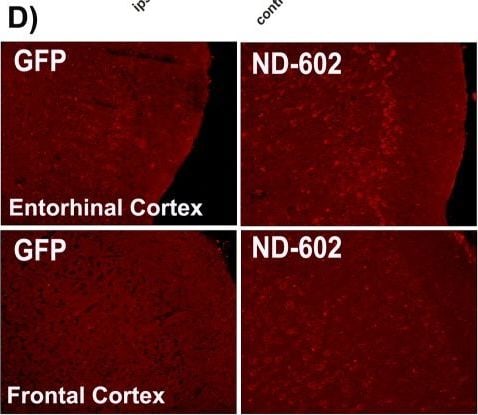
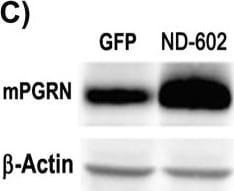
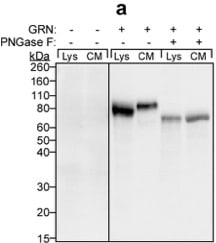
PGRN immunolabeling following ND-602.(A) Representative photomicrographs depicting PGRN immunolabeling in the dentate gyrus of the Tg2576 mouse brain following unilateral intracerebral administration of either LV-GFP, or LV-PGRN (ND-602). (B) The hippocampal density of PGRN immunolabeling was significantly elevated following ND-602 administration in both the ipsilateral and contralateral hemispheres. (C) PGRN protein expression (progranulin/ beta -actin gray values) in the hippocampus was detected by western blot assay. (D) Representative fluorescent photomicrographs depicting PGRN immunolabeling in the entorhinal cortex and frontal cortex following unilateral intracerebral administration of either LV-GFP, or LV-PGRN (ND-602). (E) The density of PGRN immunolabeling was significantly elevated following ND-602 administration in both hemispheres. By contrast, elevations in the (F) frontal cortex did not reach statistical significance. (G) Representative fluorescent photomicrograph depicting GFP (green) and NeuN (red) immunolabeling throughout the ipsilateral hippocampus following lentiviral delivery. The image is a tiled composite of multiple images obtained at 20X magnification. A closer view, (H) shows viral transduction of both NeuN-positive and–negative cells. Each bar represents the mean (± S.E.M.) (n = 8–10) optical density measured across 4 coronal sections. ** sig. diff. from GFP-treated controls, p < 0.001; * p < 0.05. ++ sig. diff. from contralateral hemisphere, p < 0.001. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/28837568), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Progranulin/PGRN by Immunocytochemistry/Immunofluorescence
PGRN immunolabeling following ND-602.(A) Representative photomicrographs depicting PGRN immunolabeling in the dentate gyrus of the Tg2576 mouse brain following unilateral intracerebral administration of either LV-GFP, or LV-PGRN (ND-602). (B) The hippocampal density of PGRN immunolabeling was significantly elevated following ND-602 administration in both the ipsilateral and contralateral hemispheres. (C) PGRN protein expression (progranulin/ beta -actin gray values) in the hippocampus was detected by western blot assay. (D) Representative fluorescent photomicrographs depicting PGRN immunolabeling in the entorhinal cortex and frontal cortex following unilateral intracerebral administration of either LV-GFP, or LV-PGRN (ND-602). (E) The density of PGRN immunolabeling was significantly elevated following ND-602 administration in both hemispheres. By contrast, elevations in the (F) frontal cortex did not reach statistical significance. (G) Representative fluorescent photomicrograph depicting GFP (green) and NeuN (red) immunolabeling throughout the ipsilateral hippocampus following lentiviral delivery. The image is a tiled composite of multiple images obtained at 20X magnification. A closer view, (H) shows viral transduction of both NeuN-positive and–negative cells. Each bar represents the mean (± S.E.M.) (n = 8–10) optical density measured across 4 coronal sections. ** sig. diff. from GFP-treated controls, p < 0.001; * p < 0.05. ++ sig. diff. from contralateral hemisphere, p < 0.001. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/28837568), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Progranulin/PGRN by Western Blot
PGRN immunolabeling following ND-602.(A) Representative photomicrographs depicting PGRN immunolabeling in the dentate gyrus of the Tg2576 mouse brain following unilateral intracerebral administration of either LV-GFP, or LV-PGRN (ND-602). (B) The hippocampal density of PGRN immunolabeling was significantly elevated following ND-602 administration in both the ipsilateral and contralateral hemispheres. (C) PGRN protein expression (progranulin/ beta -actin gray values) in the hippocampus was detected by western blot assay. (D) Representative fluorescent photomicrographs depicting PGRN immunolabeling in the entorhinal cortex and frontal cortex following unilateral intracerebral administration of either LV-GFP, or LV-PGRN (ND-602). (E) The density of PGRN immunolabeling was significantly elevated following ND-602 administration in both hemispheres. By contrast, elevations in the (F) frontal cortex did not reach statistical significance. (G) Representative fluorescent photomicrograph depicting GFP (green) and NeuN (red) immunolabeling throughout the ipsilateral hippocampus following lentiviral delivery. The image is a tiled composite of multiple images obtained at 20X magnification. A closer view, (H) shows viral transduction of both NeuN-positive and–negative cells. Each bar represents the mean (± S.E.M.) (n = 8–10) optical density measured across 4 coronal sections. ** sig. diff. from GFP-treated controls, p < 0.001; * p < 0.05. ++ sig. diff. from contralateral hemisphere, p < 0.001. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/28837568), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Mouse Progranulin/PGRN Antibody by Western Blot
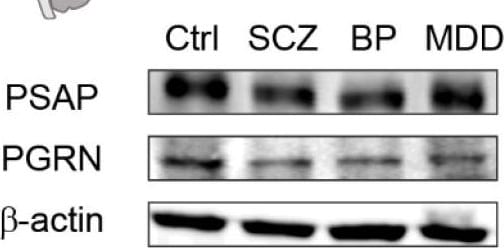
PSAP and PGRN levels are decreased in cingulate cortex tissue of schizophrenia patients. (A) Representative immunoblots of PSAP, PGRN, and beta -actin in cingulate cortex tissue from subjects of healthy control (Ctrl), schizophrenia (SCZ), bipolar disorder (BP), and major depressive disorder (MDD). Upper panel, illustration of cingulate cortex in human brain. (B,C) Bar graphs presenting quantification of the ratio of protein levels of PSAP/ beta -actin (B) and PGRN/ beta -actin (C). (D,E) Scatter plots showing the Pearson’s correlation analysis of PSAP and lifetime quantity of fluphenazine or equivalent (FLU) in schizophrenia (D) and bipolar disorder group (E), respectively. Each dot represents one patient of SCZ (blue) or BP (magenta). (F,G) Scatter plots showing the Pearson’s correlation analysis of PGRN and FLU in schizophrenia (F) and bipolar disorder group (G), respectively. One-way ANOVA with Sidak’s post hoc test. N = 11–12 samples in each group. Data are presented as mean ± S.E.M. * p ˂ 0.05 compared to Ctrl. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/36233357), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Mouse Progranulin/PGRN Antibody by Immunohistochemistry
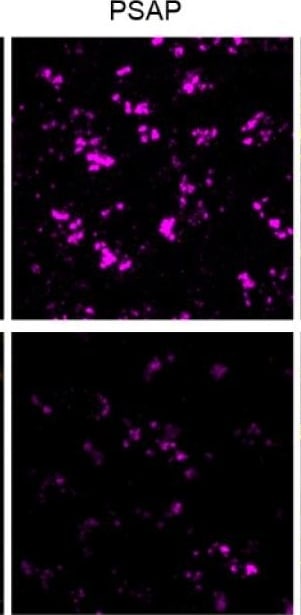
Conditional deletion of neuronal PSAP in the cingulate cortex causes downregulation of neuronal PGRN. (A) Representative fluorescent in situ hybridization images of PSAP mRNA in the cingulate cortex of mice injected with AAV-GFP and AAV-GFP-Cre. Upper row, schematic depiction of stereotaxic injection sites in one hemisphere (the same in the other hemisphere). Scale bar, 50 μm. (B) Representative immunofluorescent staining images of GFP (cyan), Cre (orange), PSAP (magenta), and PGRN (yellow) in the cingulate cortex of mice injected with AAV-GFP and AAV-GFP-Cre, respectively. Scale bar, 20 μm. (C,D) Bar graphs showing quantification of mean fluorescence intensity (MFI) of PSAP (C) and PGRN (D) staining in GFP- or Cre-positive cells. A.U, arbitrary unit. Student’s t-test, N = 34 cells in each group. Data are presented as mean ± S.E.M. **** p ˂ 0.0001. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/36233357), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Progranulin/PGRN by Immunocytochemistry/ Immunofluorescence
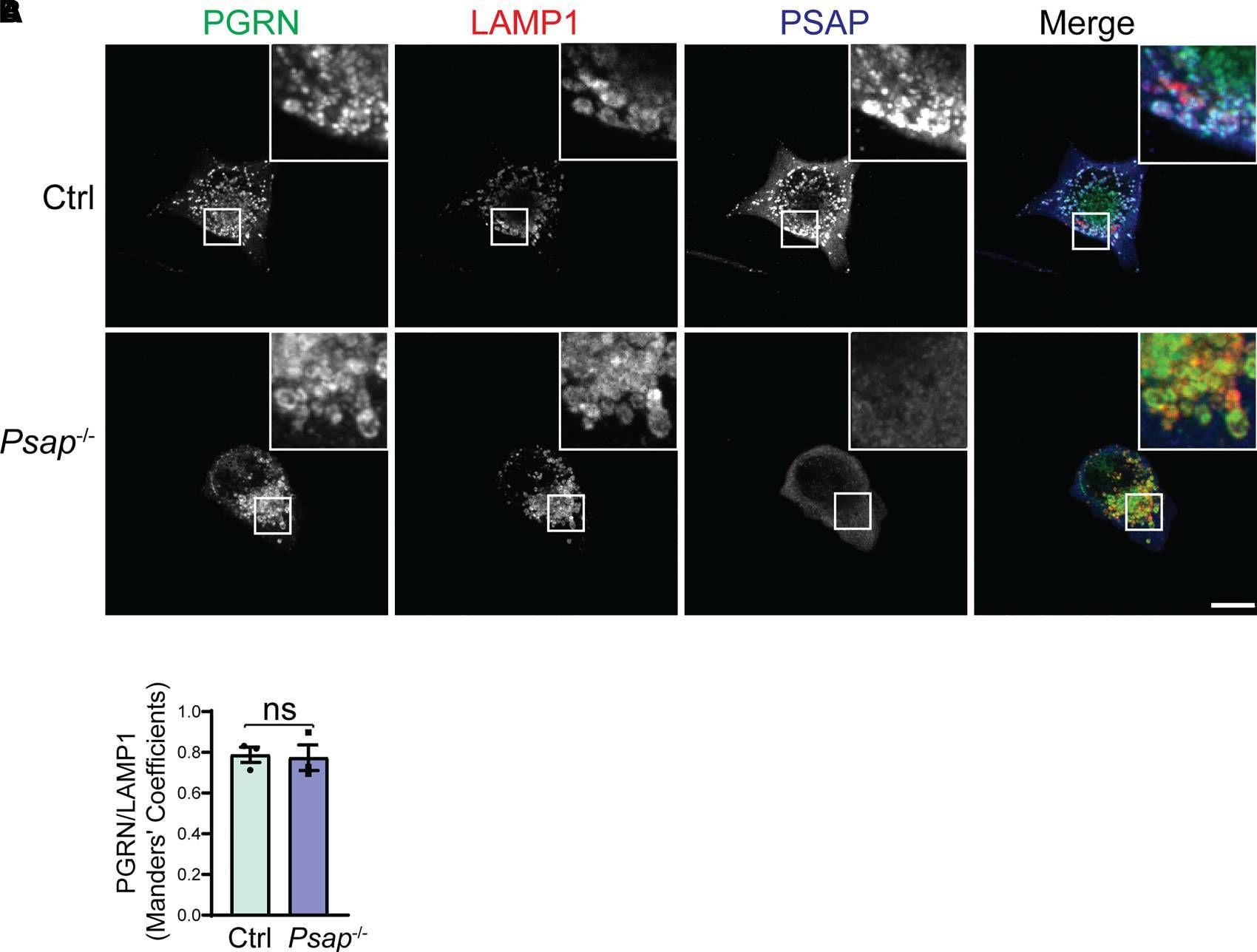
PSAP ablation does not affect PGRN lysosomal trafficking in BV2 cells. (A) Representative confocal images of BV2 cells stained with sheep anti-PGRN, rat anti-LAMP1 and rabbit anti-PSAP antibodies. Scale bar, 10 µm. (B) Quantification of PGRN signals inside LAMP1-positive vesicles in A. Mean ± SEM; n = 3, Student’s t-test, ns, not significant. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35169707), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Progranulin/PGRN by Western Blot
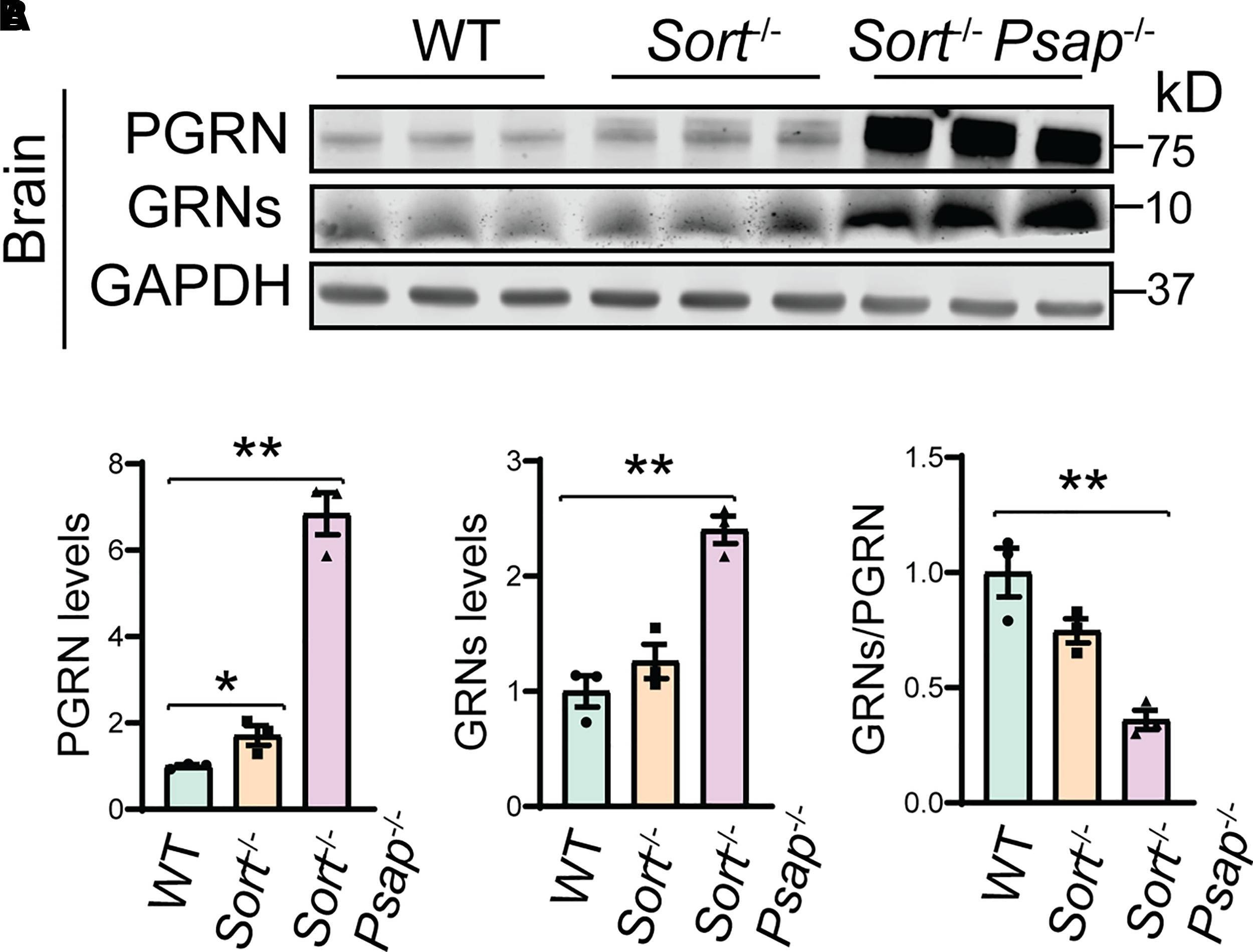
PGRN processing in Sortilin- and PSAP-deficient mice. (A,B) Western blot analysis of PGRN and granulin peptides in the cortical lysates from P21 WT, Sort−/− and Sort−/− Psap−/− mice. The levels of PGRN and granulin peptides (GRNs) were quantified and normalized to GAPDH. Lysates from three mice were analysed for each genotype (n = 3), one-way ANOVA, *P < 0.05; **P < 0.01. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35169707), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Mouse Progranulin/PGRN Antibody
Application
Recommended Usage
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed mouse splenocytes
Sample: Immersion fixed mouse splenocytes
Immunohistochemistry
5-15 µg/mL
Sample: Perfusion fixed frozen sections of mouse brain, kidney, liver, and thymus
Sample: Perfusion fixed frozen sections of mouse brain, kidney, liver, and thymus
Western Blot
0.5 µg/mL
Sample: Mouse uterus tissue and J774A.1 mouse reticulum cell sarcoma macrophage cell line
Sample: Mouse uterus tissue and J774A.1 mouse reticulum cell sarcoma macrophage cell line
Reviewed Applications
Read 8 reviews rated 4.9 using AF2557 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Progranulin/PGRN
Alternate Names
Acrogranin, GEP, GP88, GRN, PCDGF, PEPI, PGRN, Proepithelin
Gene Symbol
GRN
UniProt
Additional Progranulin/PGRN Products
Product Documents for Mouse Progranulin/PGRN Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse Progranulin/PGRN Antibody
For research use only
Related Research Areas
Citations for Mouse Progranulin/PGRN Antibody
Customer Reviews for Mouse Progranulin/PGRN Antibody (8)
4.9 out of 5
8 Customer Ratings
Have you used Mouse Progranulin/PGRN Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
5 of
8 reviews
Showing All
Filter By:
-
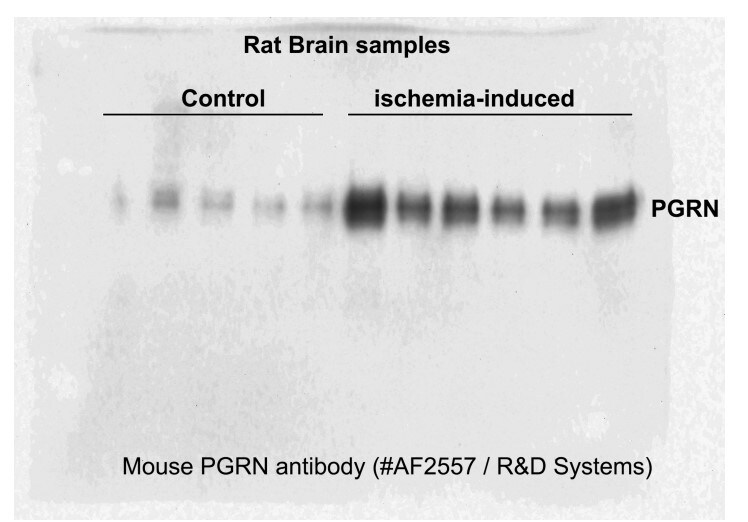
Application: Western BlotSample Tested: Adult brainSpecies: RatVerified Customer | Posted 10/14/2023We had good results using this antibody for adult rat brain samples. We were very pleased with the bands we got. We used 5microliters of primary antibody and 10ml of milk during the primary antibody incubation.
-
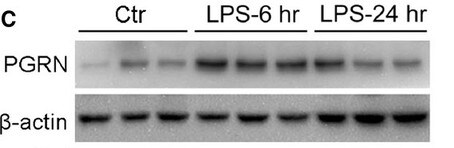
Application: Western BlotSample Tested: Spinal cordSpecies: MouseVerified Customer | Posted 07/21/2021
-
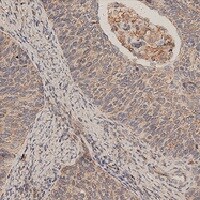
Application: ImmunohistochemistrySample Tested: Cancer TissueSpecies: MouseVerified Customer | Posted 07/09/2021
-
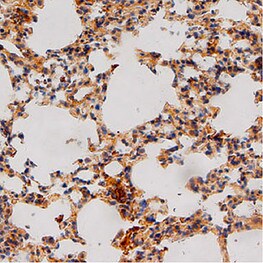
Application: ImmunohistochemistrySample Tested: Lung tissueSpecies: MouseVerified Customer | Posted 04/30/2021
-
Application: Western BlotSample Tested: Adult lungSpecies: MouseVerified Customer | Posted 04/29/2021
-
Application: Western BlotSample Tested: Embryonic stem cellsSpecies: MouseVerified Customer | Posted 07/21/2018
-
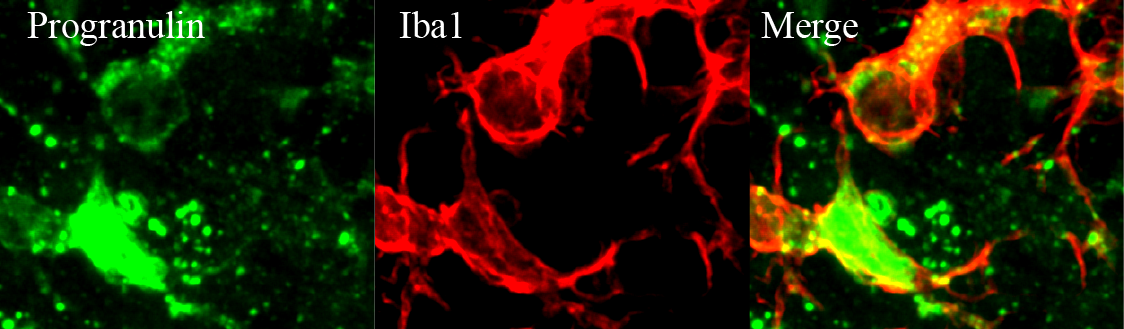
Application: Immunohistochemistry with free floating sectionSample Tested: Mouse brainSpecies: MouseVerified Customer | Posted 03/19/2018Green is Progranulin and red is Iba1 staining.antigen retrieval for 15 mins, 1:250-1:500 dilution
-
Application: Western BlotSample Tested: HEK293 cell lysates transfected with mPGRN cDNASpecies: MouseVerified Customer | Posted 06/12/2016Image is from: Naphade et al., Acta Neuropathologica, 2010, 119(1):123-33
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...