Semaphorin 3C (Sema 3C; previously semaE) is one of six Class 3 secreted semaphorins which share 40-50% amino acid (aa) identity. Class 3 semaphorins are potent chemorepellents that function in axon and/or vascular guidance during development, and may be upregulated in tumor progression (1, 2). The 751 amino acid (aa) mouse Sema3C is highly modular. It contains a 20 aa signal sequence, an ~500 aa N-terminal Sema domain that forms a beta -propeller structure similar to that found in integrin molecules, a cysteine knot, a furin-type cleavage site, an Ig-like domain, and a C-terminal basic domain (1-3). Covalent dimerization plus cleavage at the C-terminus are required for activity of class 3 semaphorins (4). Mouse Sema 3C shares at least 95% aa identity with human, rat, cow and dog Sema 3C, and 89% and 75% aa sequence identity with chick and zebrafish Sema 3C, respectively. Type 3 semaphorins transduce signals through transmembrane plexins, either directly or by binding associated neuropilin receptors (1, 2). Sema 3C signaling is transduced by Plexin-D1 indirectly via neuropilin-1 or neuropilin-2 receptors (5). Sema 3C is expressed in all somitic motor neurons, in lung buds and in cardiac neural crest cells during development (1, 5-8). Sema 3C activates integrins in certain cells so, in addition to its repulsive activities, it sometimes acts as a chemoattractant (6, 9). In the developing nervous system, this chemoattraction appears to complement Sema 3A repulsion in adjacent cell layers (1, 6, 7). Sema 3C also provides an attractive force opposing Sema 6A and Sema 6B to guide migration of neural crest endothelial cells to the cardiac outflow tract (10). Consequently, defects in aortic arch formation occur when Sema 3C or Plexin-D1 genes or Sema 3C-neuropilin interactions are disrupted (5, 11, 12).
Mouse Semaphorin 3C Antibody
R&D Systems | Catalog # AF1728

Key Product Details
Species Reactivity
Validated:
Mouse
Cited:
Mouse
Applications
Validated:
Immunohistochemistry, Western Blot, Immunocytochemistry
Cited:
Immunohistochemistry, Western Blot
Label
Unconjugated
Antibody Source
Polyclonal Sheep IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant mouse Semaphorin 3C
Gln24-Ser751 (Arg48Ala, Arg52Ala)
Accession # Q62181
Gln24-Ser751 (Arg48Ala, Arg52Ala)
Accession # Q62181
Specificity
Detects mouse Semaphorin 3C in direct ELISAs and Western blots. In direct ELISAs, less than 5% cross-reactivity with recombinant mouse (rm) Semaphorin 3A, rmSemaphorin 3B, rmSemaphorin 3E, and rmSemaphorin 3F is observed.
Clonality
Polyclonal
Host
Sheep
Isotype
IgG
Scientific Data Images for Mouse Semaphorin 3C Antibody
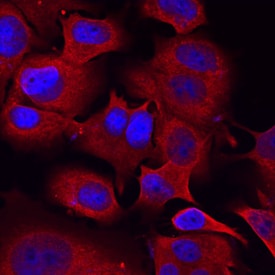
Semaphorin 3C in MCF‑7 Human Cell Line.
Semaphorin 3C was detected in immersion fixed MCF-7 human breast cancer cell line using Sheep Anti-Mouse Semaphorin 3C Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1728) at 10 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Sheep IgG Secondary Antibody (red; Catalog # NL010) and counterstained with DAPI (blue). Specific staining was localized to cytoplasm. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Semaphorin 3C in Mouse Embryo.
Semaphorin 3C was detected in immersion fixed frozen sections of mouse embryo (15 d.p.c.) using Sheep Anti-Mouse Semaphorin 3C Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1728) at 1.7 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Sheep HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS019) and counterstained with hematoxylin (blue). Specific labeling was localized to the plasma membrane of mesenchymal cells. View our protocol for Chromogenic IHC Staining of Frozen Tissue Sections.Applications for Mouse Semaphorin 3C Antibody
Application
Recommended Usage
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed MCF‑7 human breast cancer cell line
Sample: Immersion fixed MCF‑7 human breast cancer cell line
Immunohistochemistry
5-15 µg/mL
Sample: Immersion fixed frozen sections of mouse embryo (E13.5-15.5)
Sample: Immersion fixed frozen sections of mouse embryo (E13.5-15.5)
Western Blot
0.1 µg/mL
Sample:
Sample:
Recombinant Mouse Semaphorin 3C Fc Chimera, Truncated
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Semaphorin 3C
References
- Hinck, L. (2004) Dev. Cell 7:783.
- Neufeld, G. et al. (2005) Front. Biosci. 10:751.
- Gherardi, E. et al. (2004) Curr. Opin. Struct. Biol. 14:669.
- Adams, R.H. et al. (1997) EMBO J. 16:6077.
- Gitler, A.D. et al. (2004) Dev. Cell 7:107.
- Bagnard, D. et al. (1998) Development 125:5043.
- Cohen, S. et al. (2005) Eur. J. Neurosci. 21:1767.
- Puschel, A. W. et al. (1995) Neuron 14:941.
- Herman, J.G. and G.G. Meadows (2007) Int. J. Oncol. 30:1231.
- Toyofuku, T. et al. (2008) Dev. Biol. 321:251.
- Feiner, L. et al. (2001) Development 128:3061.
- Gu, C. et al. (2003) Dev. Cell 5:45.
Alternate Names
SEMA3C, SEMAE, Semaphorin E, SEME
Gene Symbol
SEMA3C
UniProt
Additional Semaphorin 3C Products
Product Documents for Mouse Semaphorin 3C Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse Semaphorin 3C Antibody
For research use only
Related Research Areas
Citations for Mouse Semaphorin 3C Antibody
Customer Reviews for Mouse Semaphorin 3C Antibody
There are currently no reviews for this product. Be the first to review Mouse Semaphorin 3C Antibody and earn rewards!
Have you used Mouse Semaphorin 3C Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...

