TREM-1 (Triggering Receptor Expressed on Myeloid cells) is a type I transmembrane protein having a single Ig-like domain. It associates with the adapter protein, DAP12, to deliver an activating signal. Several other TREM family members have been reported that are structurally similar but share less than 30% amino acid identity. TREM-1 is expressed on blood neutrophils and a subset of monocytes, and expression is up-regulated by bacterial LPS. Engagement of TREM-1 with a monoclonal antibody leads to expression of IL-8, MCP-1, and TNF-alpha suggesting that this receptor plays an important role in inflammatory responses. TREM-1 is expressed at high levels on neutrophils of patients with microbial sepsis and in mice with LPS-induced shock. Blockade of TREM-1 with a TREM-1/Fc fusion protein protected mice against LPS-induced shock.

Key Product Details
Validated by
Biological Validation
Species Reactivity
Validated:
Mouse
Cited:
Mouse
Applications
Validated:
Western Blot, Flow Cytometry, Immunocytochemistry, Agonist Activity, CyTOF-ready
Cited:
Immunohistochemistry, Neutralization, Flow Cytometry, Immunocytochemistry, ELISA Capture, In vivo assay, Functional Assay
Label
Unconjugated
Antibody Source
Monoclonal Rat IgG2A Clone # 174031
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant mouse TREM-1
Ala21-Ser202
Accession # Q9JKE2
Ala21-Ser202
Accession # Q9JKE2
Specificity
Detects mouse TREM-1 in direct ELISAs and Western blots. In Western blots, no cross-reactivity with recombinant mouse TREM-2B, recombinant human (rh) TREM-1, or rhTREM-2 is observed.
Clonality
Monoclonal
Host
Rat
Isotype
IgG2A
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Mouse TREM‑1 Antibody
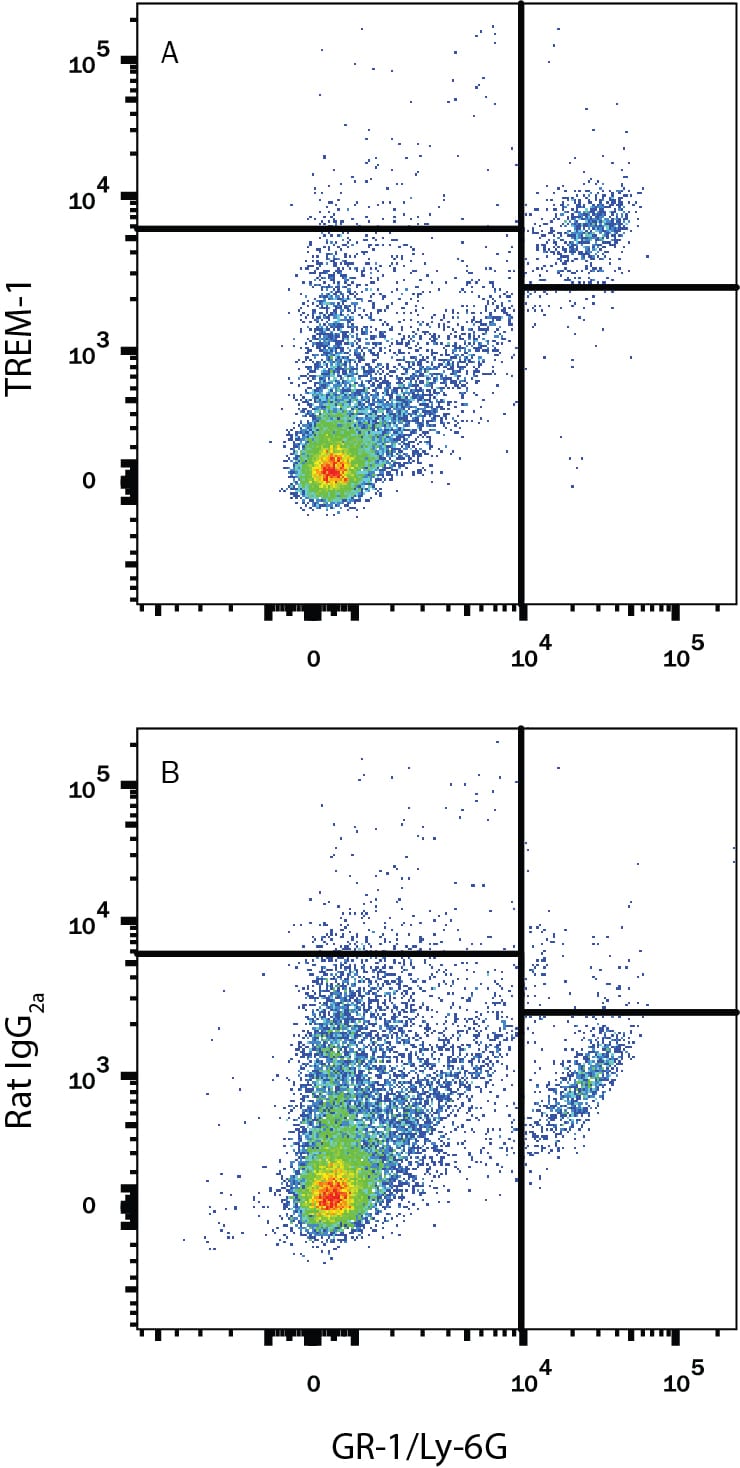
Detection of TREM‑1 in Mouse Splenocytes by Flow Cytometry.
Mouse splenocytes were stained with Rat Anti-Mouse Gr-1/Ly-6G Alexa Fluor® 405-conjugated Monoclonal Antibody (Catalog # FAB1037V) and either (A) Rat Anti-Mouse TREM-1 Monoclonal Antibody (Catalog # MAB1187) or (B) Rat IgG2AIsotype Control (Catalog # MAB006) followed by Phycoerythrin-conjugated Anti-Rat IgG Secondary Antibody (Catalog # F0105B).TREM‑1 in Mouse Splenocytes.
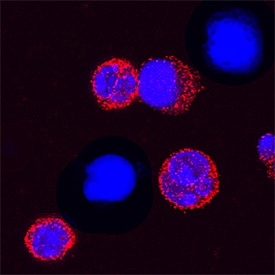
TREM-1 was detected in immersion fixed mouse splenocytes treated with LPS using Rat Anti-Mouse TREM-1 Monoclonal Antibody (Catalog # MAB1187) at 15 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Rat IgG Secondary Antibody (red; Catalog # NL013) and counterstained with DAPI (blue). Specific staining was localized to cytoplasm. View our protocol for Fluorescent ICC Staining of Non-adherent Cells.Detection of TREM-1 by Immunocytochemistry/ Immunofluorescence
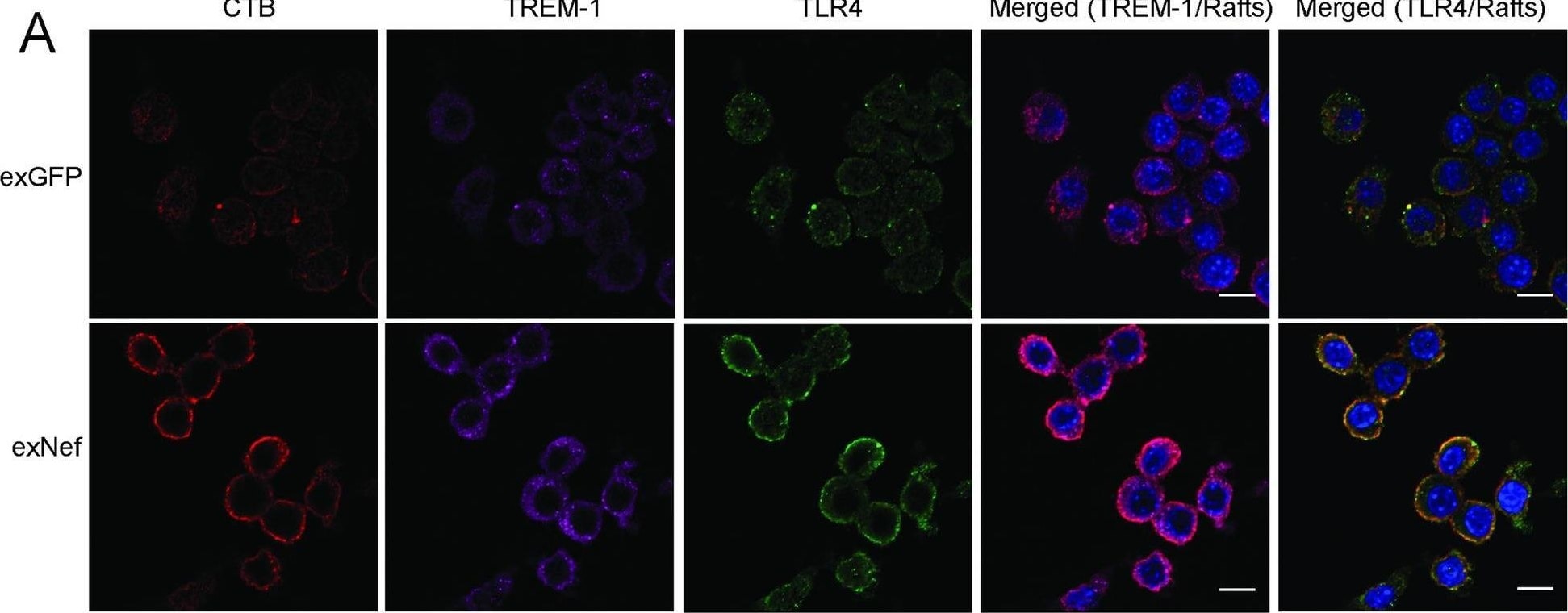
ExNef potentiate inflammatory signalling cascade via re-localization of TLR4 & TREM-1 to lipid rafts.A–The effect of exNef (0.4 ng/ml, 48h) on abundance of lipid rafts & re-localization of TLR4 & TREM-1 to the plasma membrane in RAW264.7 macrophages. Left column, CTB staining; second column, anti-TREM-1 staining; third column anti-TLR4 staining; fourth column–merge TREM-1/rafts, right column–merge TLR4/rafts. Scale bars 10 μm. Image collected & cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/31344124), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of TREM-1 by Immunocytochemistry/ Immunofluorescence
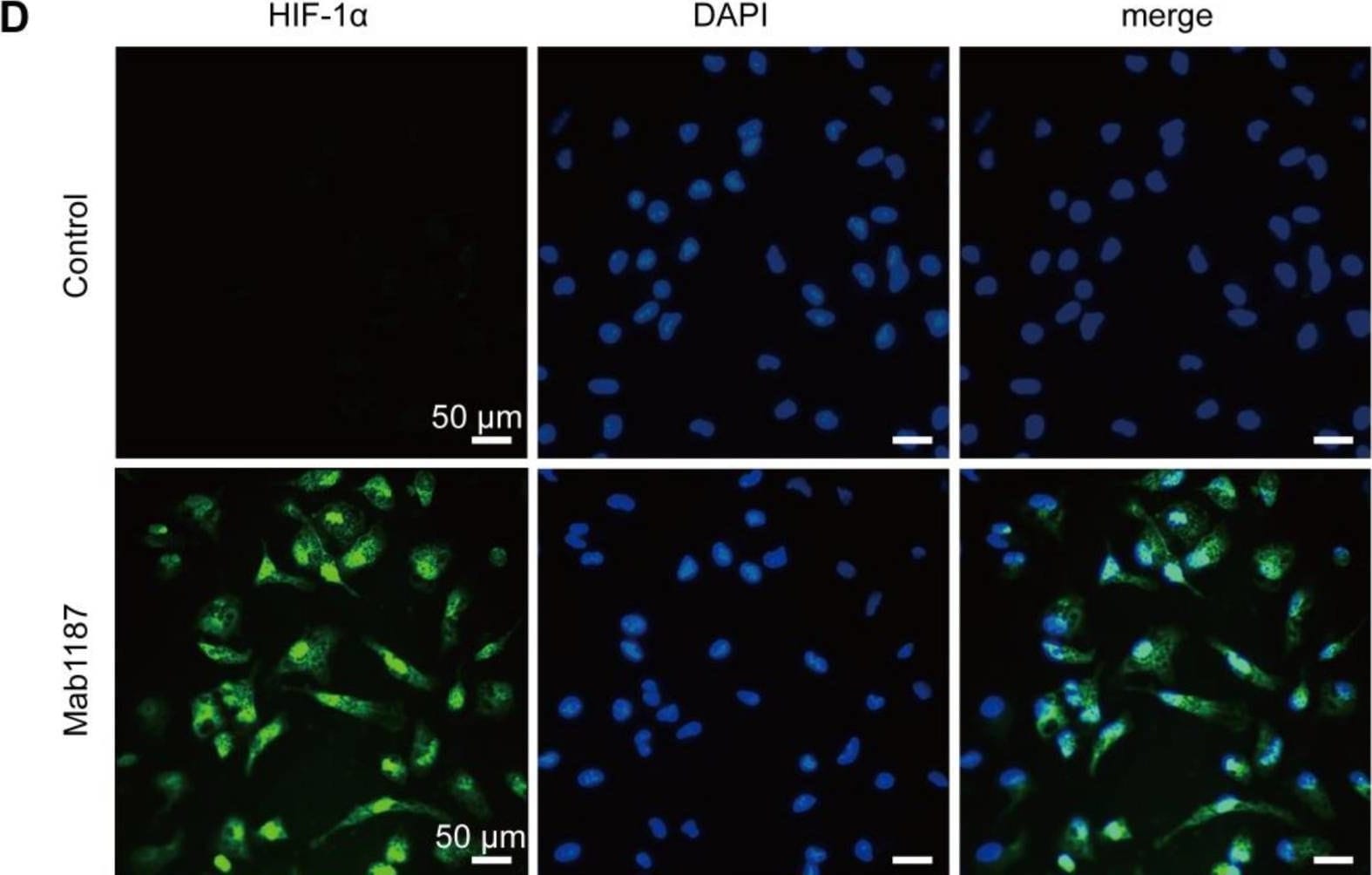
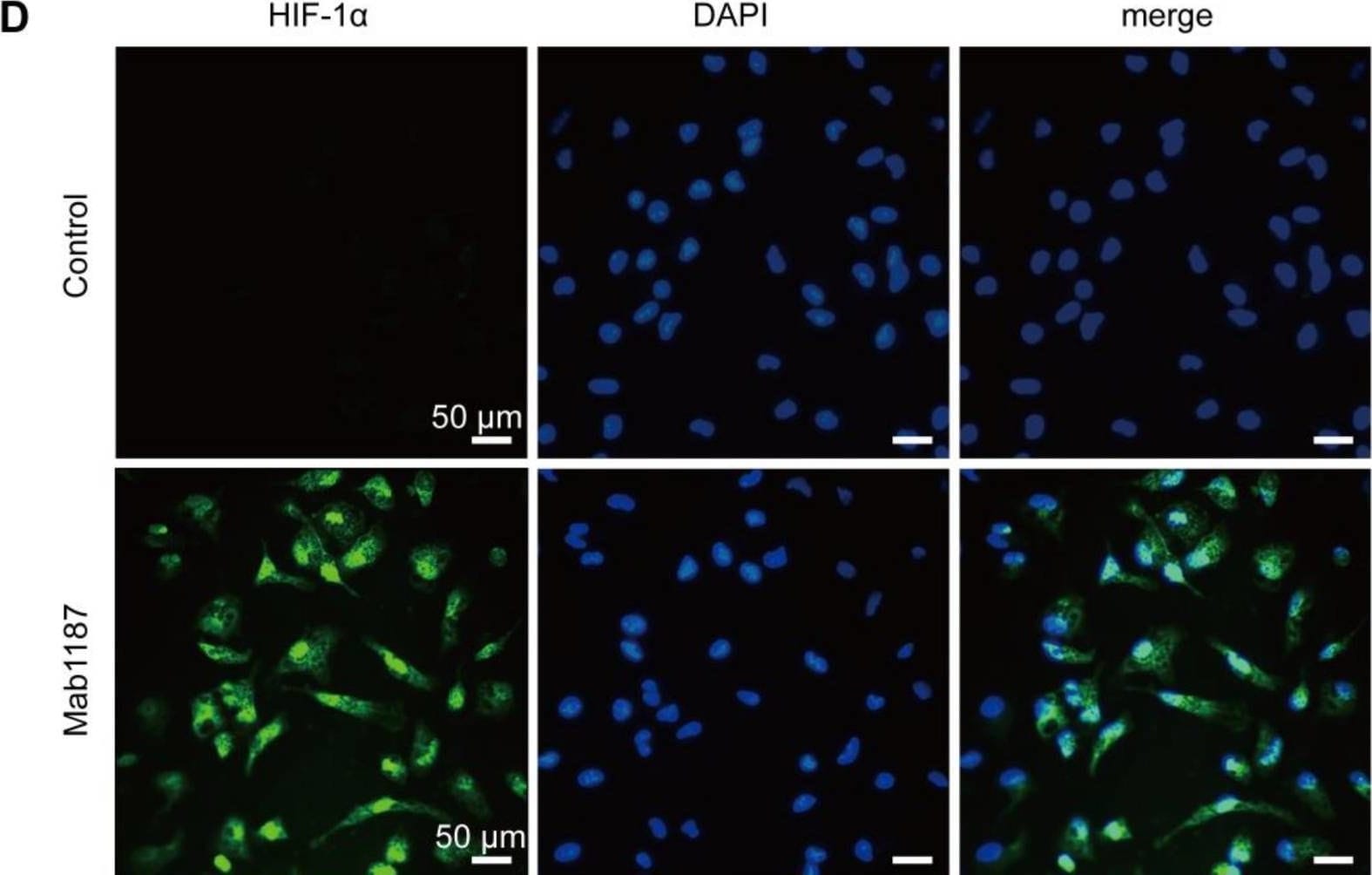
TREM-1 activation induced HIF-1 alpha accumulation and translocation to the nucleus in macrophages. Macrophages were incubated with anti-TREM-1 mAb (10 μg/mL) in a normoxia condition. (A) Six hours later, Hif‐1 alpha gene levels were measured using qPCR, n=4-6. (B-C) Twenty-four hours later, the protein of HIF‐1 alpha was performed by western blot with GAPDH as a loading control, n=3. (D) Macrophages were cultured on anti-TREM-1 for 24 h and then subjected to immunofluorescence examination to analyze the HIF-1 alpha accumulation and translocation to the nucleus (scale bar, 50 μm). (E) Average fluorescent intensity was calculated by HIF-1 alpha + fluorescence intensity (IntDen)/area of the region (Area) using ImageJ, n=6. (F-G) 1×106 cells/well were premixed with PBS control or PX-478 (25 μM) for 30 min, then plated into 12-well plates with agonist anti-TREM-1 mAb (10 μg/mL). HK2 protein levels were measured after an additional incubation for 24 h, n=3. n represents experiments performed on different macrophages from separate mice. Data are expressed as the mean ± SD. Student's t-test (two-tailed, unpaired) was used to compare Mab1187 and Control in (A-E): * P < 0.05, ** P < 0.01, and *** P < 0.001. One-way ANOVA adjusted by Tukey's multiple comparison test was used in G: *** P < 0.001. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36594089), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of TREM-1 by Immunocytochemistry/ Immunofluorescence
TREM-1 activation induced HIF-1 alpha accumulation and translocation to the nucleus in macrophages. Macrophages were incubated with anti-TREM-1 mAb (10 μg/mL) in a normoxia condition. (A) Six hours later, Hif‐1 alpha gene levels were measured using qPCR, n=4-6. (B-C) Twenty-four hours later, the protein of HIF‐1 alpha was performed by western blot with GAPDH as a loading control, n=3. (D) Macrophages were cultured on anti-TREM-1 for 24 h and then subjected to immunofluorescence examination to analyze the HIF-1 alpha accumulation and translocation to the nucleus (scale bar, 50 μm). (E) Average fluorescent intensity was calculated by HIF-1 alpha + fluorescence intensity (IntDen)/area of the region (Area) using ImageJ, n=6. (F-G) 1×106 cells/well were premixed with PBS control or PX-478 (25 μM) for 30 min, then plated into 12-well plates with agonist anti-TREM-1 mAb (10 μg/mL). HK2 protein levels were measured after an additional incubation for 24 h, n=3. n represents experiments performed on different macrophages from separate mice. Data are expressed as the mean ± SD. Student's t-test (two-tailed, unpaired) was used to compare Mab1187 and Control in (A-E): * P < 0.05, ** P < 0.01, and *** P < 0.001. One-way ANOVA adjusted by Tukey's multiple comparison test was used in G: *** P < 0.001. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36594089), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Mouse TREM‑1 Antibody
Application
Recommended Usage
Agonist Activity
Measured by its ability to stimulate TNF-alpha secretion by P388D1 mouse lymphoma cells (ATCC # TIB-63). Bouchon, A. et al. (2001) Nature, 410:1103 and Bouchon, A. et al. (2000) J. Immunology, 164:4991.
The ED50 for this effect is typically 0.4-3 μg/mL.
The ED50 for this effect is typically 0.4-3 μg/mL.
CyTOF-ready
Ready to be labeled using established conjugation methods. No BSA or other carrier proteins that could interfere with conjugation.
Flow Cytometry
0.25 µg/106 cells
Sample: Mouse splenocytes
Sample: Mouse splenocytes
Immunocytochemistry
8-25 µg/mL
Sample: Immersion fixed mouse splenocytes treated with LPS
Sample: Immersion fixed mouse splenocytes treated with LPS
Western Blot
1 µg/mL
Sample: Recombinant Mouse TREM‑1 Fc Chimera (Catalog # 1187-TR)
Sample: Recombinant Mouse TREM‑1 Fc Chimera (Catalog # 1187-TR)
Reviewed Applications
Read 1 review rated 4 using MAB1187 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Protein A or G purified from hybridoma culture supernatant
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: TREM-1
References
- Bouchon, A. (2000) J. Immunol. 164:4991.
- Bouchon, A. (2001) Nature 410:1103.
- Nathan, C. and A. Ding (2001) Nature Med. 7:530.
Long Name
Triggering Receptor Expressed on Myeloid Cells 1
Alternate Names
CD354, TREM1
Gene Symbol
TREM1
UniProt
Additional TREM-1 Products
Product Documents for Mouse TREM‑1 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse TREM‑1 Antibody
For research use only
Related Research Areas
Citations for Mouse TREM‑1 Antibody
Customer Reviews for Mouse TREM‑1 Antibody (1)
4 out of 5
1 Customer Rating
Have you used Mouse TREM‑1 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
Application: Flow CytometrySample Tested: Spleen tissueSpecies: MouseVerified Customer | Posted 08/28/2022Works well with flow!
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...