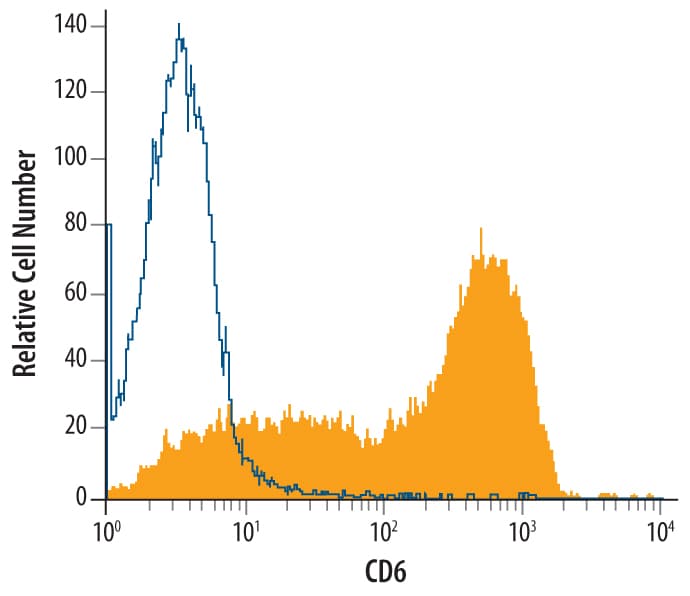
CD6 is a member of the group B scavenger receptor cysteine-rich (SRCR) superfamily. CD6 is a type I membrane glycoprotein and contains three extracellular SRCR domains. CD6 is expressed at low levels on immature thymocytes and at high levels on mature thymocytes. The majority of peripheral blood T cells, a subset of B cells, and a subset of neuronal cells express CD6. CD6 is a 668 amino acid protein with a 24 amino acid predicted signal sequence, a 374 amino acid extracellular domain, and a 244 amino acid transmembrane region. The 626 amino acid murine homolog has also been identified. The human and murine proteins share 70% amino acid identity over their full-lengths.
The role of CD6 has not been fully elucidated. However, it appears to play a role as both a co-stimulatory molecule in T cell activation and as an adhesion receptor. Studies demonstrating a mitogenic effect for T cells with some CD6 specific monoclonal antibodies, in conjunction with either accessory cells or PMA and anti-CD2 mAb, support the concept of CD6 as a co-stimulatory molecule. Anti-CD6 monoclonal antibody has been used as an immunosuppressive agent for patients undergoing kidney or bone marrow allograft rejection. It has also been used to remove CD6+ T cells from donor bone marrow prior to allogeneic bone marrow transplantation. Other studies have demonstrated an adhesive role for CD6, it has been demonstrated to bind the activated leukocyte cell adhesion molecule (ALCAM, CD166). CD6/ALCAM interactions have been postulated to play a role in thymocyte development. The presence of ALCAM on neuronal cells may provide a mechanism of interaction between CD6+ T cells and ALCAM+ neuronal cells. Phosphorylation of the CD6 molecule appears to play a role in CD6-mediated signal transduction. Serine and threonine residues become hyperphosphorylated and tyrosine residues become phosphorylated when T cells are activated with anti-CD6 mAb in conjunction with PMA, anti-CD2, or anti-CD3 mAb. The CD6 intracellular domain contains regions that can interact with SH2 or SH3 containing proteins. However, the signaling pathways have not been elucidated.