F-Spondin (“Floor plate” and “thrombospondin” homology; also Spondin-1 and VSGP) is a member of the F-Spondin family of proteins that collectively belong to a subgroup of TSR (thrombospondin) type I class molecules (1). Class I molecules are either membrane-bound or ECM-associated. F-Spondin is a 110 kDa, secreted, heparin-binding extracellular matrix glycoprotein first characterized in rat for its high expression in embryonic floor plate (2‑4). Human F-Spondin is synthesized as an 807 amino acid (aa) precursor that contains a 28 aa signal sequence and a 779 aa mature region (3, 4). The mature region includes an N-terminal reelin-like domain (aa 1‑200), a centrally placed F-Spondin (FS) type segment (aa 201‑440), and six C-terminal class 2 thrombospondin type I repeats (1, 3, 5). Class 1 and 2 repeats differ in the placement of their cysteine residues. The fifth and sixth TSP repeats (aa 668‑806) apparently bind ECM, while TSP repeats 1‑4 (aa 442‑666), plus the spondin segment, are suggested to mediate either repulsive activity (on motor neurons), or outgrowth promoting activity (on sensory neurons) (1, 6). At least two isoforms of F-Spondin are known. Both are proteolytically-generated, one by plasmin, another by an unidentified protease. Plasmin cleaves the C-terminus at two points, generating a soluble, 95 kDa, 656 aa F‑Spondin that contains all but TSP repeats #5 and 6 (7). The unidentified protease appears to cleave F-Spondin between the FS segment and the first TSP repeat, generating 60 kDa and 50 kDa fragments, respectively (6). F-Spondin shows highly unusual glycosylation, exhibiting both C-mannosylation (mannose bound to Trp) and O-fucosylation (fucose bound to Ser/Thr) (4). The significance of these glycosidic modifications is unknown. Mature human F-Spondin is 98%, 97%, 98%, and 97% aa identical to mature canine, rat, bovine and mouse F-Spondin, respectively. Mammalian cells known to express F‑Spondin include floor plate epithelium, ventral motor neurons, Schwann cells, fibroblasts, hippocampal pyramidal cells, endothelial cells, vascular smooth muscle cells and some tumor cells (6, 8, 9).
Human F‑Spondin/SPON1 Antibody
R&D Systems | Catalog # AF3135

Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human
Applications
Validated:
Western Blot, Neutralization, Simple Western
Cited:
Western Blot, Neutralization
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant human F‑Spondin/SPON1
Phe29-Cys807
Accession # Q9HCB6
Phe29-Cys807
Accession # Q9HCB6
Specificity
Detects human F‑Spondin/SPON1 in direct ELISAs and Western blots.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human F‑Spondin/SPON1 Antibody
Detection of Human F‑Spondin/SPON1 by Western Blot.
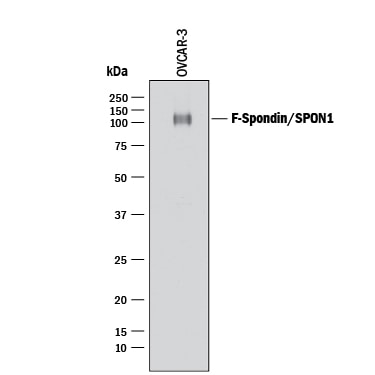
Western blot shows lysates of OVCAR-3 human ovarian carcinoma cell line. PVDF membrane was probed with 1 µg/mL of Goat Anti-Human F-Spondin/SPON1 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3135) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (HAF017). A specific band was detected for F-Spondin/ SPON1 at approximately 110 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Detection of Human F‑Spondin/SPON1 by Simple WesternTM.
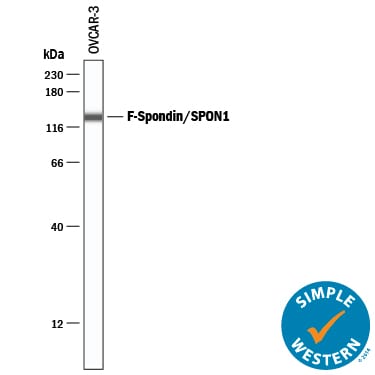
Simple Western lane view shows lysates of OVCAR-3 human ovarian carcinoma cell line, loaded at 0.2 mg/mL. A specific band was detected for F-Spondin/SPON1 at approximately 134 kDa (as indicated) using 10 µg/mL of Goat Anti-Human F-Spondin/SPON1 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3135) followed by 1:50 dilution of HRP-conjugated Anti-Goat IgG Secondary Antibody (HAF109). This experiment was conducted under reducing conditions and using the 12-230 kDa separation system.Applications for Human F‑Spondin/SPON1 Antibody
Application
Recommended Usage
Simple Western
10 µg/mL
Sample: OVCAR‑3 human ovarian carcinoma cell line
Sample: OVCAR‑3 human ovarian carcinoma cell line
Western Blot
1 µg/mL
Sample: OVCAR‑3 human ovarian carcinoma cell line
Sample: OVCAR‑3 human ovarian carcinoma cell line
Neutralization
Measured by its ability to neutralize the enhancement of neurite outgrowth of dorsal root ganglion neurons from E13 chick embryos induced by F‑Spondin. 3 µg/mL is sufficient to block neurite outgrowth induced by 90 ng/3 μL drop/well of F-Spondin immobilized on nitrocellulose coated plate.
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: F-Spondin/SPON1
References
- Feinstein, Y. and A. Klar (2004) Int. J. Biochem. Cell Biol. 36:975.
- Klar, A. et al. (1992) Cell 69:95.
- Miyamoto, K. et al. (2001) Arch. Biochem. Biophys. 390:93.
- Gonzalez de Peredo, A. et al. (2002) Mol. Cell. Proteomics 1:11.
- Adams, J.C. and R.P. Tucker (2000) Dev. Dyn. 218:280.
- Burstyn-Cohen, T. et al. (1998) J. Neurosci. 18:8875.
- Tzarfaty-Majar, V. et al. (2001) J. Biol. Chem. 276:28233.
- Feinstein, Y. et al. (1999) Development 126:3637.
- Pyle-Chenault, R.A. et al. (2005) Tumor Biol. 26:245.
- Mattes, J. and P.S. Foster (2003) Curr. Drug Targets Inflamm. Allergy 2:169.
Alternate Names
FSpondin, SPON1, VSGP
Gene Symbol
SPON1
UniProt
Additional F-Spondin/SPON1 Products
Product Documents for Human F‑Spondin/SPON1 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human F‑Spondin/SPON1 Antibody
For research use only
Related Research Areas
Citations for Human F‑Spondin/SPON1 Antibody
Customer Reviews for Human F‑Spondin/SPON1 Antibody
There are currently no reviews for this product. Be the first to review Human F‑Spondin/SPON1 Antibody and earn rewards!
Have you used Human F‑Spondin/SPON1 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...