IGFBP-rp1, also known as Mac25/Angiomodulin (AGM), tumor-derived adhesion factor (TAF) and prostacyclin-stimulating factor (PSF), is a secreted protein that contains three protein domain modules. Human IGFBP-rp1 cDNA encodes 282 amino acid (aa) residue precursor protein with a putative 26 aa signal peptide. Mature IGFBP-rp1 is a glycosylated protein with an N-terminal IGFBP domain, followed by a Kazal-type serine proteinase inhibitor domain and a C-terminal
immunoglobulin-like C2-type domain. The similarity of IGFBP-rp1 with the IGFBPs is confined to the N-terminal IGFBP domain, which contains all 12 of the conserved cysteine residues found in IGFBP-1 through 5. Human and mouse IGFBP-rp1 are highly homologous. Discounting a segment of 43 aa near the N-terminus that is missing in the mouse homologue, human and mouse IGFBP-rp1 share 94% aa sequence identity. IGFBP-rp1 is expressed in many normal tissues and in cancer cells. It is abundantly expressed in high endothelial venules (HEVs) of blood vessels in the secondary lymphoid tissues. The expression of IGFBP-rp1 is upregulated in senescing epithelial cells and by retinoic acid. IGFBP-rp1 binds IGF and insulin with very low affinity and has been shown to enhance the mitogenic actions of IGF and insulin. IGFBP-rp1 also has IGF/insulin-independent activities. It interacts with heparan sulfate proteoglycans, type IV collagen, and specific chemokines.
IGFBP‑rp1 supports weak cell adhesion, promotes cell spreading on type IV collagen, and stimulates the production of the potent vasodilator PGI2. It modulates tumor cell growth and has also been implicated in angiogenesis. IGFBP-rp1 is proteolytically cleaved between lysine 97 and alanine 98. Cleaved IGFBP-rp1 has enhanced cell attachment activity but can no longer bind IGF/insulin (1-3).
Human IGFBP‑rp1/IGFBP‑7 Antibody
R&D Systems | Catalog # MAB13341
Recombinant Monoclonal Antibody.

Key Product Details
Species Reactivity
Human
Applications
Immunohistochemistry
Label
Unconjugated
Antibody Source
Recombinant Monoclonal Goat IgG Clone # 40012B
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant human IGFBP-rp1
Arg98-Arg277
Accession # AAA16187
Arg98-Arg277
Accession # AAA16187
Specificity
Detects human IGFBP-rp1 in direct ELISAs.
Clonality
Monoclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human IGFBP‑rp1/IGFBP‑7 Antibody
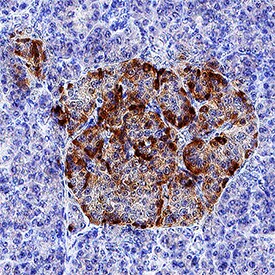
IGFBP‑rp1/IGFBP‑7 in Human Pancreas.
IGFBP-rp1/IGFBP-7 was detected in immersion fixed paraffin-embedded sections of human pancreas using Goat Anti-Human IGFBP-rp1/IGFBP-7 Monoclonal Antibody (Catalog # MAB13341) at 1.7 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS008) and counterstained with hematoxylin (blue). Specific staining was localized to islet cell cytoplasm. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Applications for Human IGFBP‑rp1/IGFBP‑7 Antibody
Application
Recommended Usage
Immunohistochemistry
1-25 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human pancreas
Sample: Immersion fixed paraffin-embedded sections of human pancreas
Formulation, Preparation, and Storage
Purification
Protein A or G purified from cell culture supernatant
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized
from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: IGFBP-rp1/IGFBP-7
References
- Hwa, V. et al. (1999) Endocrinology Rev. 20:761.
- Nagakubo, D. et al. (2003) J. Immunol. 171:553.
- Ahmed, S. et al. (2003) Biochem. Biophys. Res. Commun. 310:612.
Long Name
Insulin-like Growth Factor Binding Protein Related Protein 1
Alternate Names
IGFBP-7, IGFBP7, IGFBPrp1, Mac25, PSF
Gene Symbol
IGFBP7
UniProt
Additional IGFBP-rp1/IGFBP-7 Products
Product Documents for Human IGFBP‑rp1/IGFBP‑7 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human IGFBP‑rp1/IGFBP‑7 Antibody
For research use only
Related Research Areas
Customer Reviews for Human IGFBP‑rp1/IGFBP‑7 Antibody
There are currently no reviews for this product. Be the first to review Human IGFBP‑rp1/IGFBP‑7 Antibody and earn rewards!
Have you used Human IGFBP‑rp1/IGFBP‑7 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
