Klotho beta, a divergent structural member of the glycosidase I superfamily, is expressed primarily in the liver and pancreas, with lower expression in adipose tissue (1, 2). Like Klotho, Klotho beta facilitates binding between FGF19 subfamily members and their receptors via formation of a ternary complex (3). The Klotho beta mediated interaction of human FGF19 (mouse FGF15) with FGF Receptor 4 in the liver negatively regulates bile acid synthesis by controlling the secretion of two key bile acid synthase genes, cholesterol 7-alpha hydroxylase (Cyp7a1) and sterol 12-alpha hydroxylase (Cyp8b1) (2-5). Klotho beta is also a cofactor for the interaction of FGF21 with FGF Receptor 1c in adipocytes, which allows FGF21 to stimulate GLUT1 expression, upregulating adipocyte insulin-dependent glucose uptake (2-4, 6). The 1043 amino acid (aa) type I transmembrane protein is composed of a 51 aa signal sequence, a 943 aa extracellular domain (ECD) containing two glycosidase-like regions, a 21 aa transmembrane domain, and 28 aa intracellular tail. Since Klotho-related proteins lack critical active site Glu residues present in beta -glycosidases, it was initially unclear whether they were functional enzymes (1, 7). However, glucuronidase activity has since been demonstrated for Klotho, indicating that physiologically relevant enzymatic activity for Klotho beta is also possible (8). The extracellular domain shares 79%, 87%, 87% and 67% identity with mouse, equine, canine and rat Klotho beta, respectively. The low identity with rat reflects aa discordance within rodent ECD.
Human Klotho beta Antibody
R&D Systems | Catalog # MAB58891

Key Product Details
Species Reactivity
Applications
Label
Antibody Source
Product Specifications
Immunogen
Phe53-Leu997
Accession # Q86Z14
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human Klotho beta Antibody
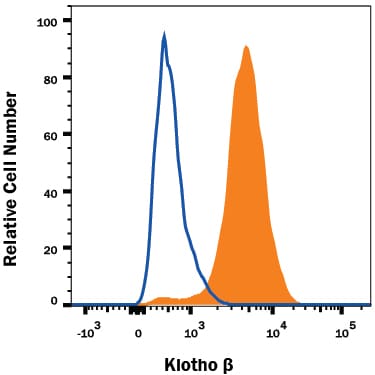
Detection of Klotho beta in HepG2 Human Cell Line by Flow Cytometry.
HepG2 human hepatocellular carcinoma cell line was stained with Rabbit Anti-Human Klotho beta Monoclonal Antibody (Catalog # MAB58891, filled histogram) or isotype control antibody (Catalog # AB-105-C, open histogram), followed by Allophycocyanin-conjugated Anti-Rabbit IgG Secondary Antibody (Catalog # F0111). View our protocol for Staining Membrane-associated Proteins.Klotho beta in HepG2 Human Cell Line.
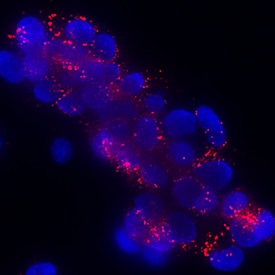
Klotho beta was detected in immersion fixed HepG2 human hepatocellular carcinoma cell line using Rabbit Anti-Human Klotho beta Monoclonal Antibody (Catalog # MAB58891) at 3 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Rabbit IgG Secondary Antibody (red; Catalog # NL004) and counterstained with DAPI (blue). Specific staining was localized to cytoplasm. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Applications for Human Klotho beta Antibody
Flow Cytometry
Sample: HepG2 human hepatocellular carcinoma cell line
Immunocytochemistry
Sample: Immersion fixed HepG2 human hepatocellular carcinoma cell line
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS For liquid material, refer to CoA for concentration.
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Klotho beta
References
- Mian, I. S. (1998) Blood Cells Mol. Dis. 24:83.
- Kurosu, H. and M. Kuro-o (2009) Mol. Cell. Endocrinol. 299:72.
- Ito, S. et al. (2005) J. Clin. Invest. 115:2202.
- Kurosu, H. et al. (2007) J. Biol. Chem. 282:26687.
- Lin, B. C. et al. (2007) J. Biol. Chem. 282:27277.
- Ogawa, Y. et al. (2007) Proc. Natl. Acad. Sci USA 104:7432.
- Chang, Q. et al. (2005) Science 310:490.
- Goetz, R. et al. (2007) Mol. Cell. Biol. 27:3417.
Alternate Names
Gene Symbol
UniProt
Additional Klotho beta Products
Product Documents for Human Klotho beta Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Klotho beta Antibody
For research use only
Related Research Areas
Citations for Human Klotho beta Antibody
Customer Reviews for Human Klotho beta Antibody
There are currently no reviews for this product. Be the first to review Human Klotho beta Antibody and earn rewards!
Have you used Human Klotho beta Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- View all Protocols, Troubleshooting, Illustrated assays and Webinars