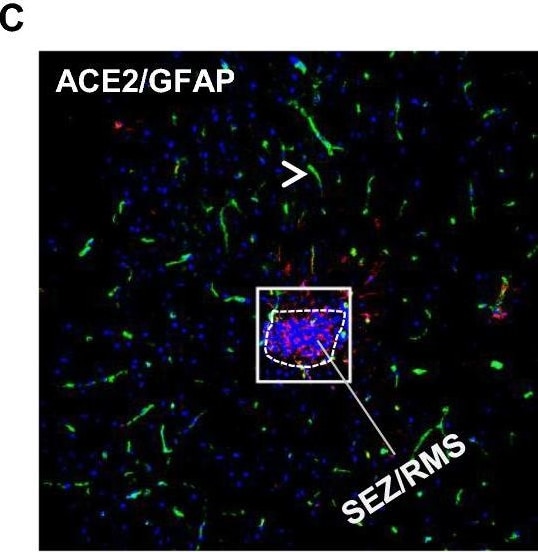
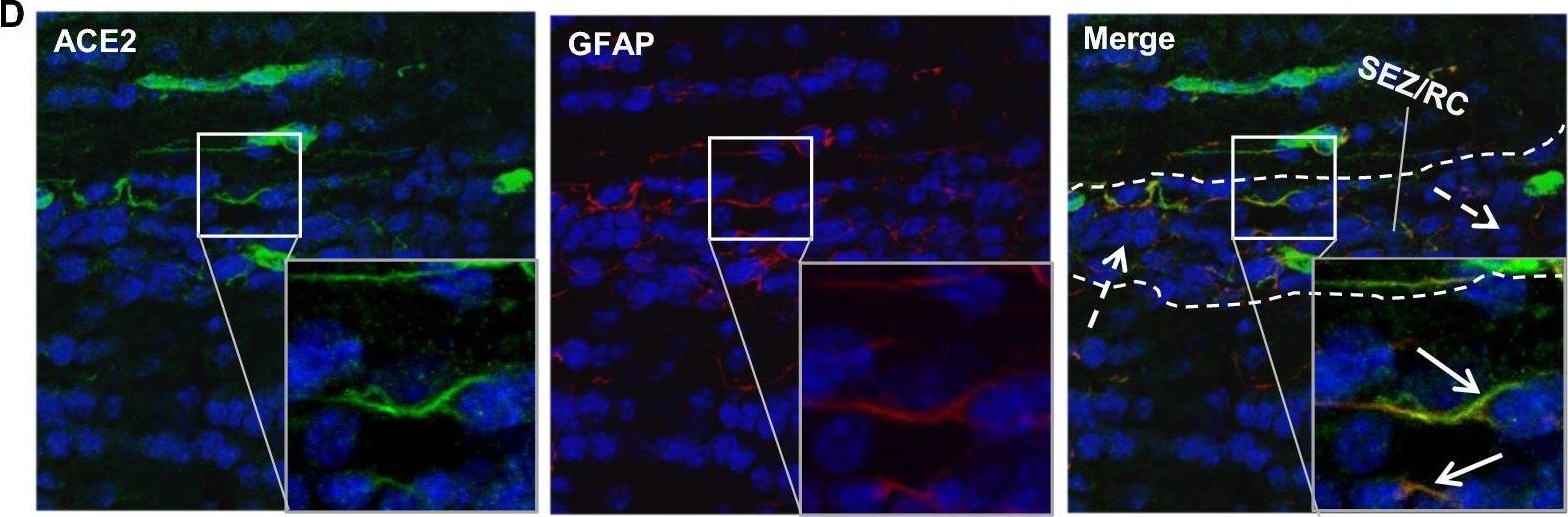
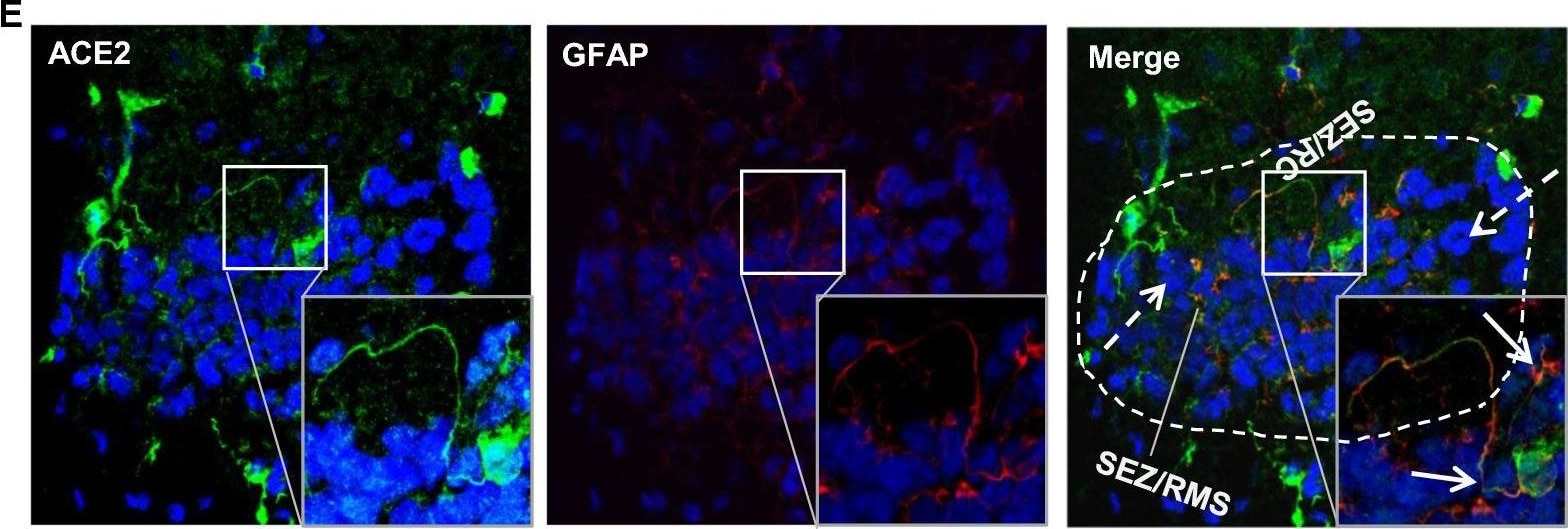
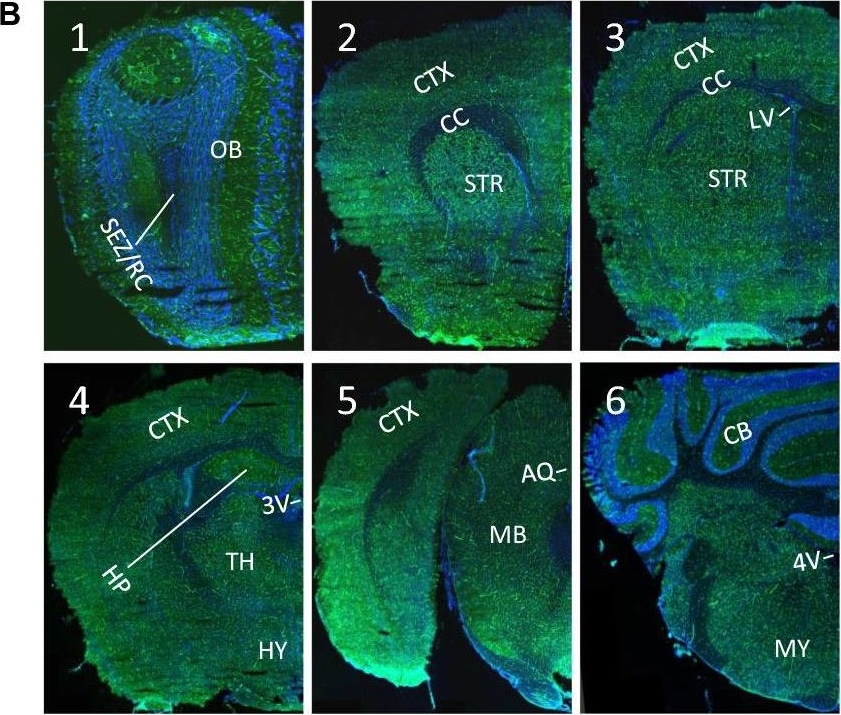
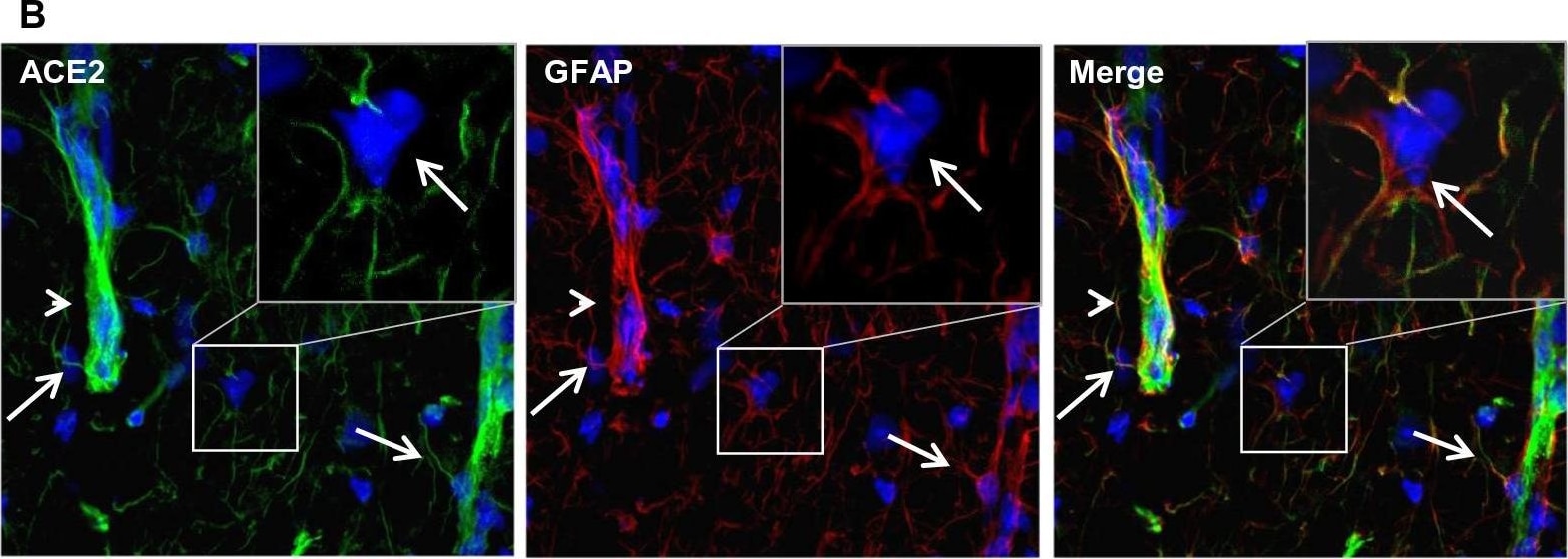
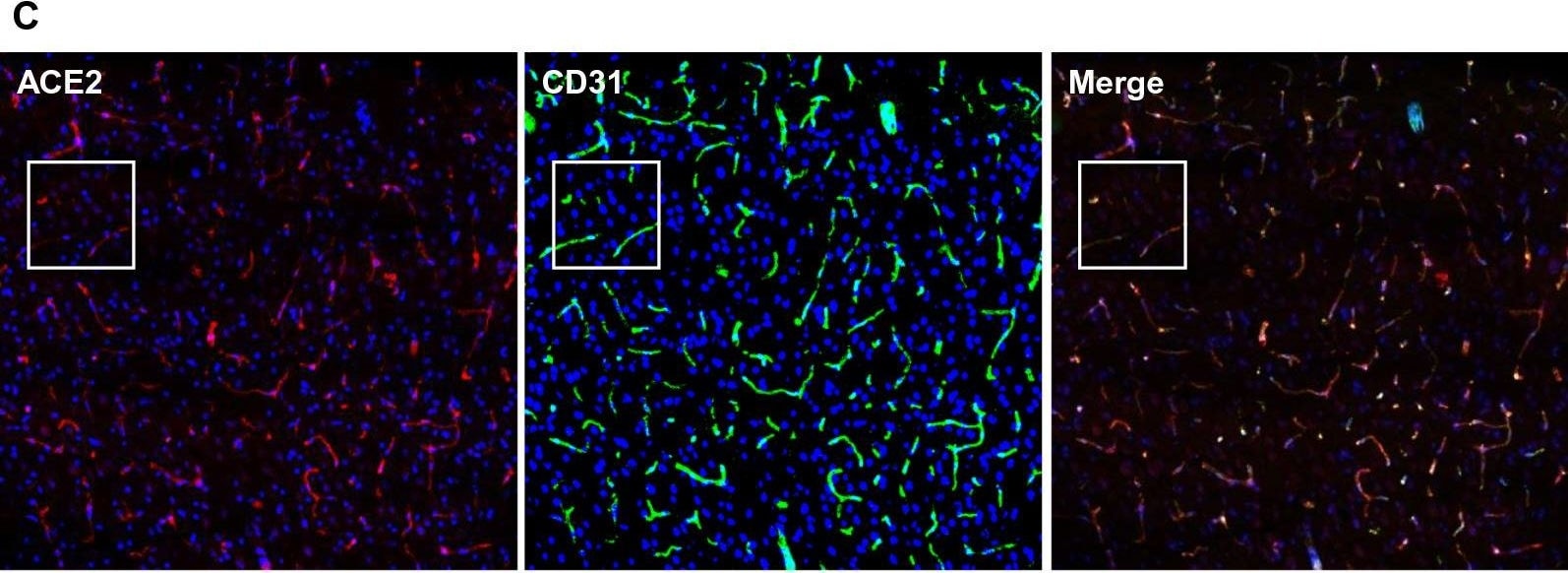
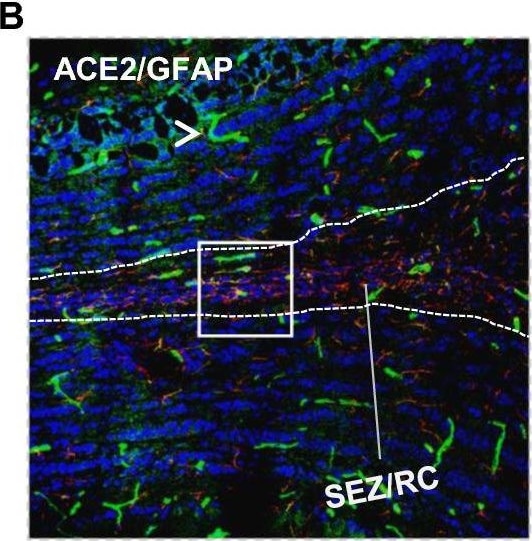
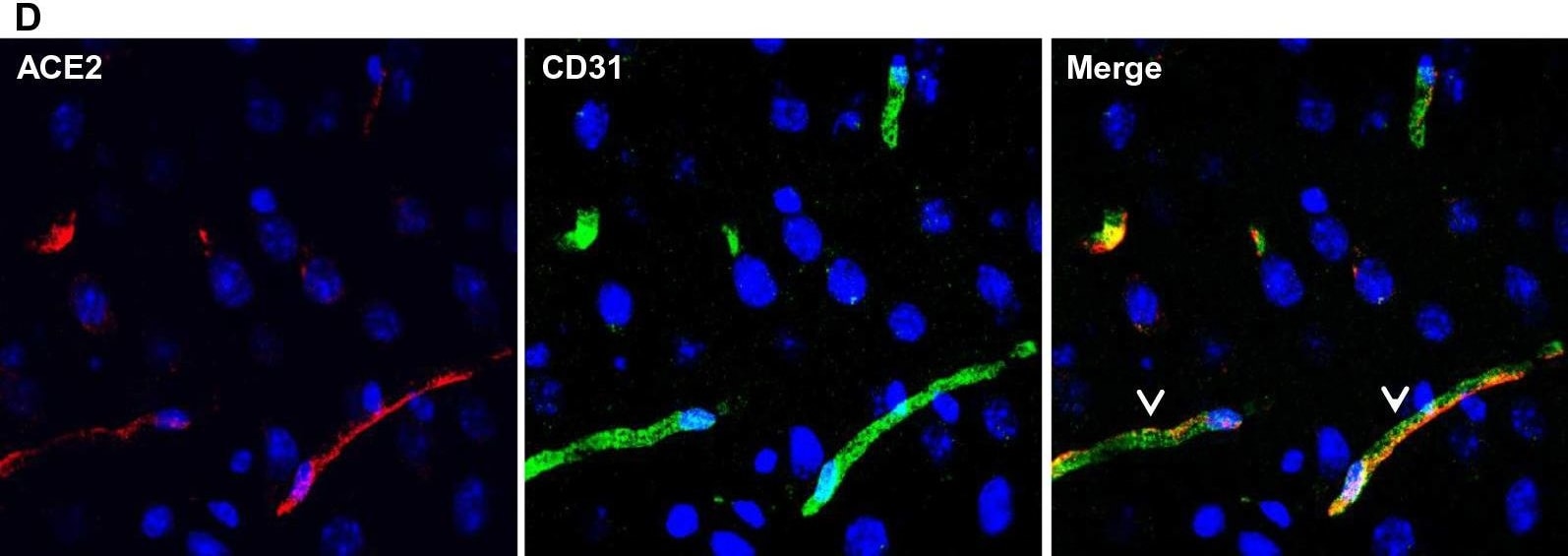
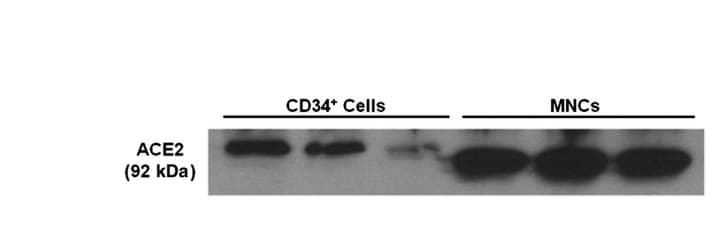
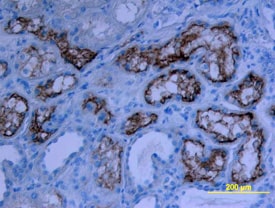
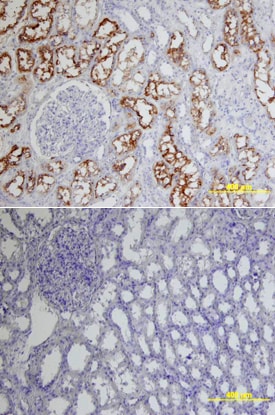
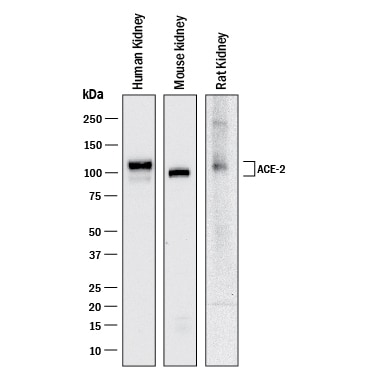
ACE-2, also called ACEH (ACE homolog), is an integral membrane protein and a zinc metalloprotease of the ACE family that also includes somatic and germinal ACE (1). Human ACE-2 has about 40% amino acid identity to the N- and C-terminal domains of human somatic ACE. The predicted human ACE-2 protein sequence consists of 805 amino acids, including a N-terminal signal peptide, a single catalytic domain, a C-terminal membrane anchor, and a short cytoplasmic tail. ACE-2 cleaves angiotensins I and II as a carboxypeptidase. ACE-2 mRNA is found at high levels in testis, kidney, and heart and at moderate levels in colon, small intestine, and ovary. Classical ACE inhibitors such as captopril and lisinopril do not inhibit ACE-2 activity. Novel peptide inhibitors of ACE-2 do not inhibit ACE activity (2). Genetic data from Drosophila, mice and rats show that ACE-2 is an essential regulator of heart function in vivo (3).
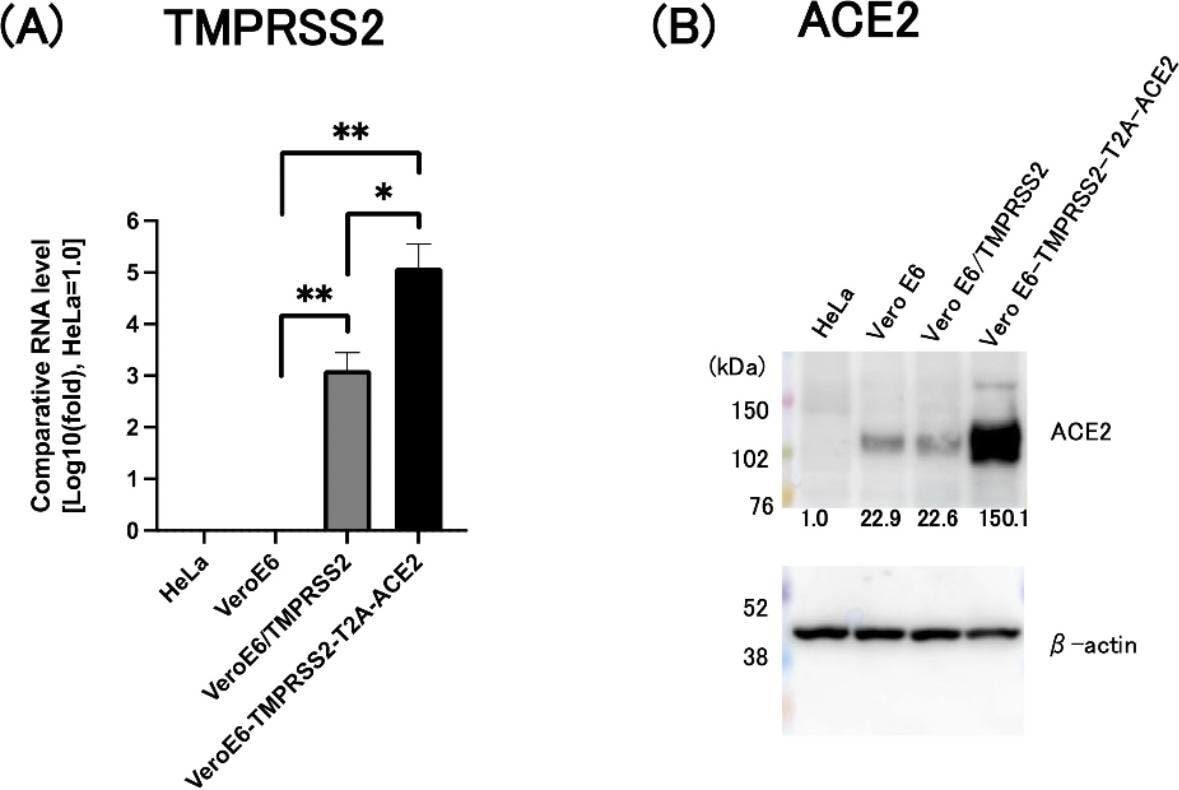
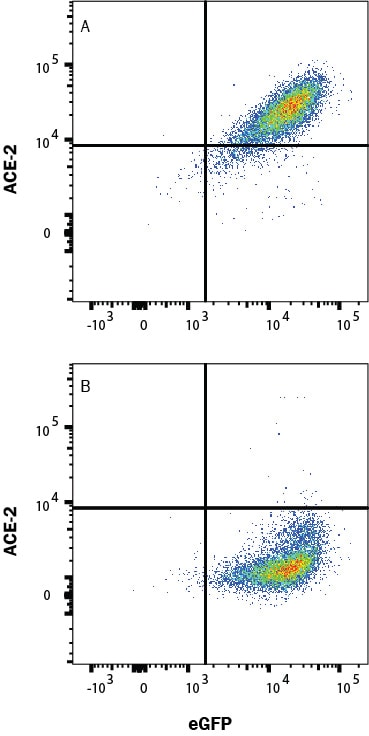
ACE2 has been shown to be a functional receptor of the human coronaviruses SARS-CoV and SARS-CoV-2 (4, 5). This Human anti-ACE2 antibody (catalog # AF933) was used to block the variant SARS-CoV-2 and ACE2 interaction to elucidate viral transmission and potential therapeutic strategies. (5)


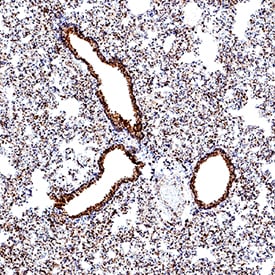
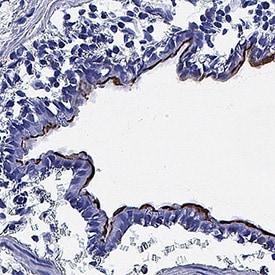
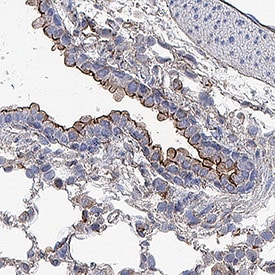
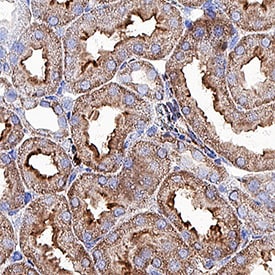
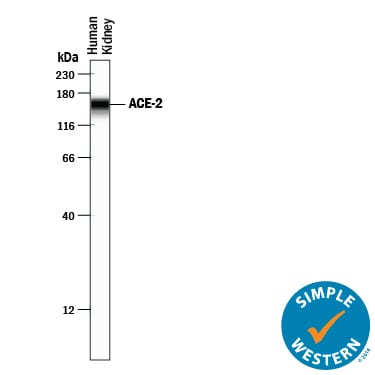
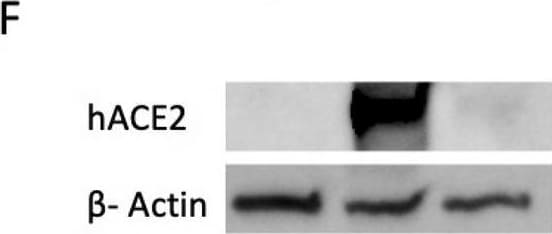

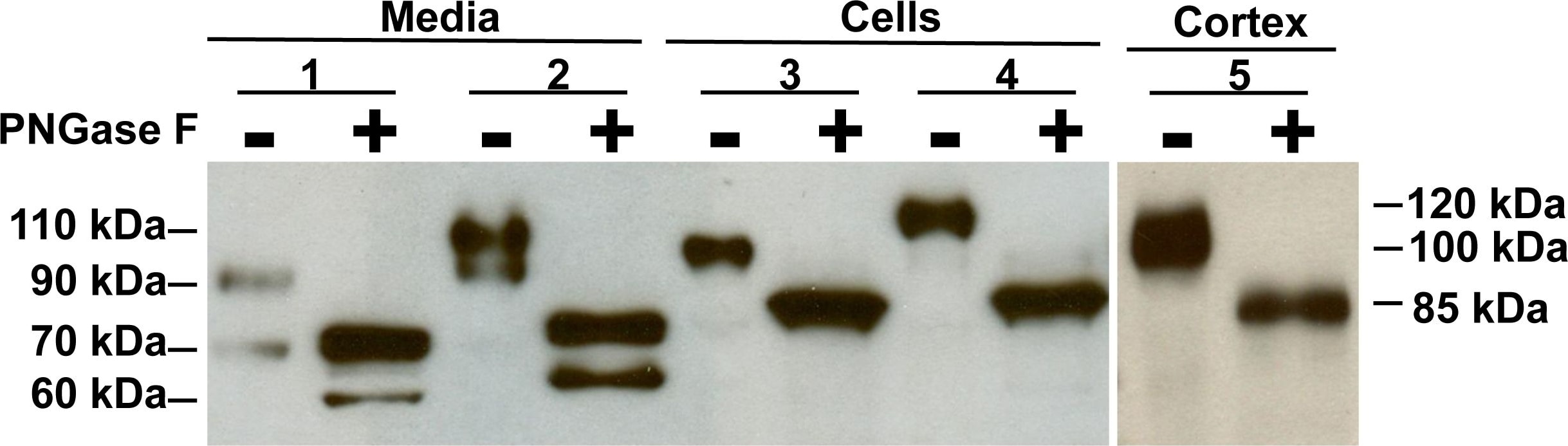
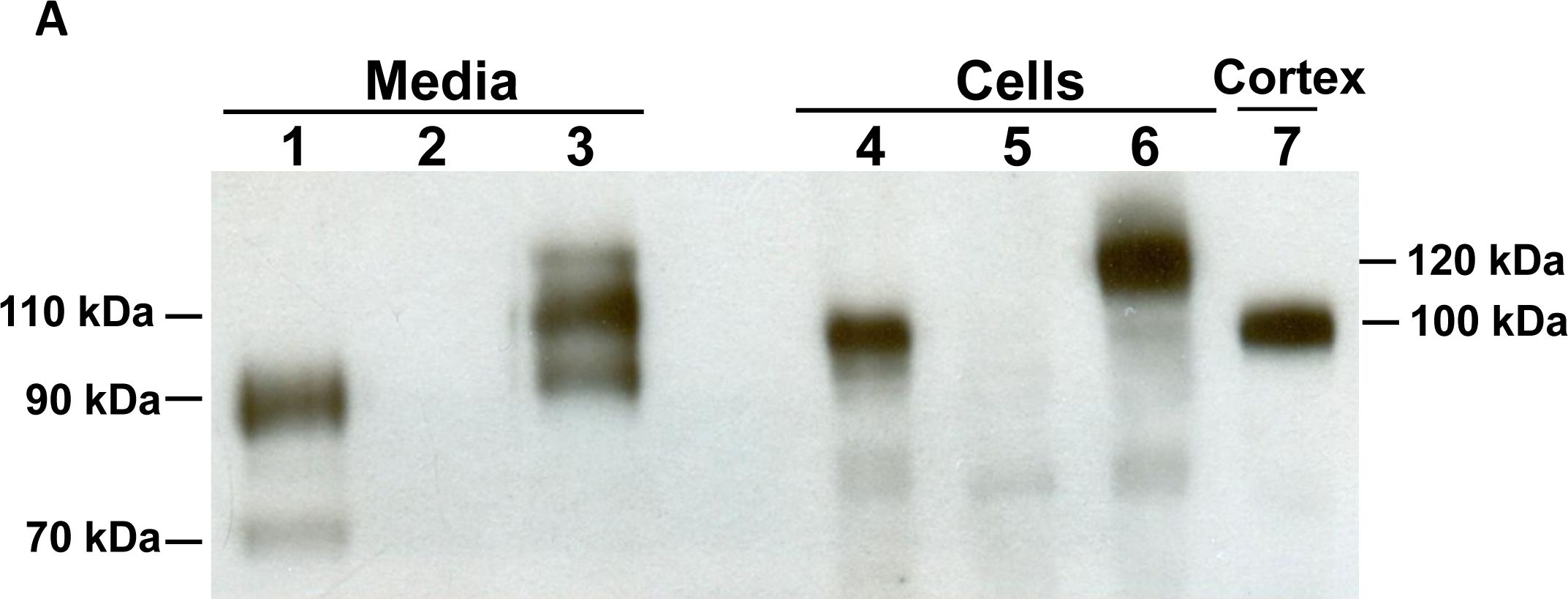
![Simple Western: ACE-2 Antibody [Unconjugated] [AF933] - Human/Mouse/Rat/Hamster ACE‑2 Antibody](https://resources.rndsystems.com/images/products/af933_human-ace-2-ectodomain-affinity-purified-polyclonal-ab-30820241044556.jpg)