Nectins are a small family of Ca++-independent immunoglobulin (Ig)-like Cell Adhesion Molecules (CAMs) that organize intercellular junctions (1). The Nectin family has at least four members (Nectin-1-4), all of which show alternate splicing (except for Nectin-4), a transmembrane (TM) region (except for Nectin-1 gamma ), and three extracellular Ig-domains. Nectins are highly homologous to the human receptor for poliovirus, and as such have been alternately named poliovirus receptor-related proteins. They do not, however, appear to bind poliovirus (1). Nectin-2 is a 60 or 65 kDa type I TM glycoprotein that is found on a variety of cell types (2, 3). It has two splice forms (4, 5). Nectin-2 delta is a 65 kDa long form and is synthesized as a 538 amino acid precursor. It contains a 31 amino acid (aa) signal sequence, a 329 aa extracellular region, a 21 aa TM segment, and a 157 aa cytoplasmic domain. The extracellular region contains one N-terminal 85 aa V-type Ig domain and two 45-55 aa C2-type Ig domains. The V-domain is believed to mediate Nectin binding to its ligands (6). The short, 60 kDa isoform of Nectin-2 (Nectin-2 alpha ) has the same signal sequence and extracellular domain as nectin-2 delta, but differs in the TM and cytoplasmic region (4, 5). In this case, the cytoplasmic tail is only 94 aa in length. The human extracellular region shows 72% aa sequence identity with the equivalent region in mouse. Nectin-2 is known to bind the pseudorabies virus, and herpes simplex virus-2 (HSV-2), but not HSV-1. It does not bind poliovirus. As a cell adhesion molecule, Nectin-2 will form cis-homodimers (same cell), followed by trans-dimers (across cells). Nectin-2 will not cis-dimerize with other Nectins, but will cis-dimerize with its two splice forms. Notably, a Nectin-2 cis-dimer on one cell will heterodimerize with a Nectin-3 cis-dimer on another cell (1). Nectin-2 is found concentrated in adherens junctions, and exists on neurons, endothelial cells, epithelial cells and fibroblasts.

Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Gln32-Gly360
Accession # Q92692
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human Nectin‑2/CD112 Antibody
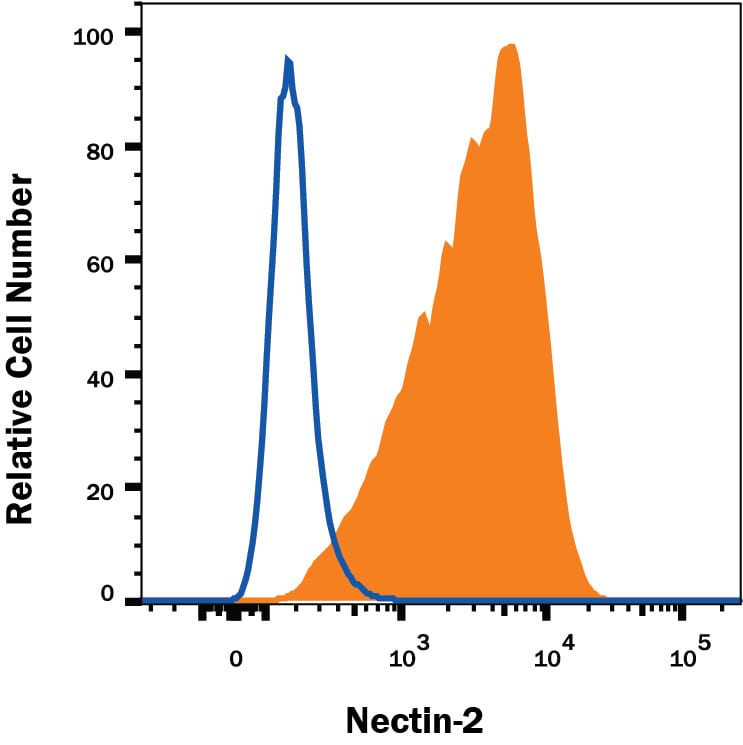
Detection of Nectin‑2/CD112 in K562 Human Cell Line by Flow Cytometry.
K562 human chronic myelogenous leukemia cell line was stained with Mouse Anti-Human Nectin-2/CD112 Monoclonal Antibody (Catalog # MAB2229, filled histogram) or isotype control antibody (Catalog # MAB002, open histogram) followed by PE-conjugated Goat anti-mouse secondary antibody (Catalog # F0102B). View our protocol for Staining Membrane-associated Proteins.Nectin‑2/CD112 in MCF‑7 Human Cell Line.
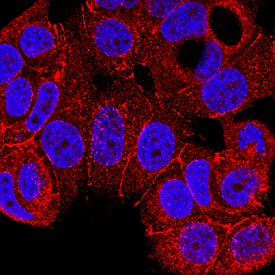
Nectin-2/CD112 was detected in immersion fixed MCF-7 human breast cancer cell line using Mouse Anti-Human Nectin-2/CD112 Monoclonal Antibody (Catalog # MAB2229) at 8 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Mouse IgG Secondary Antibody (red; Catalog # NL007) and counterstained with DAPI (blue). Specific staining was localized to cytoplasm and plasma membrane. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Applications for Human Nectin‑2/CD112 Antibody
Flow Cytometry
Sample: K562 Human cell line
Immunocytochemistry
Sample:
Immersion fixed MCF-7 human breast cancer cell line
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Nectin-2/CD112
References
- Takai, Y. and H. Nakanishi, 2003, J. Cell Sci. 116:17.
- Bottino, C. et al. (2003) J. Exp. Med. 198:557.
- Pende, D. et al. (2005) Mol. Immunol. 42:463.
- Eberle, F. et al. (1995) Gene 159:267.
- Warner, M.S. et al. (1998) Virology 246:179.
- Struyf, F. et al. (2002) J. Virol. 76:12940.
Long Name
Alternate Names
Gene Symbol
UniProt
Additional Nectin-2/CD112 Products
Product Documents for Human Nectin‑2/CD112 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Nectin‑2/CD112 Antibody
For research use only
Related Research Areas
Customer Reviews for Human Nectin‑2/CD112 Antibody
There are currently no reviews for this product. Be the first to review Human Nectin‑2/CD112 Antibody and earn rewards!
Have you used Human Nectin‑2/CD112 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- View all Protocols, Troubleshooting, Illustrated assays and Webinars