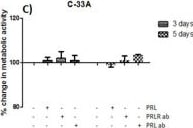
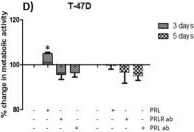
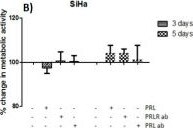
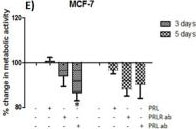
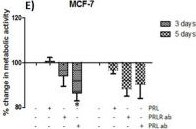
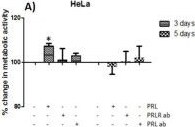
The neuroendocrine pituitary hormone Prolactin (PRL), also known as lactotrophin, mamotrophin, luteotropic hormone (LTH), or luteotropin, is a secreted hormone that affects reproduction and homeostasis in vertebrates. The functions of PRL can be placed in six broad categories: 1) reproduction and lactation; 2) growth and development; 3) endocrinology and metabolism; 4) brain and behavior; 5) immunomodulation; and 6) electrolyte balance (1, 2). PRL is secreted by the anterior pituitary gland, mammary gland, placenta, brain, uterus, decidua, dermal fibroblasts, B cells, T cells, NK cells, and some breast cancer cell lines. Although the major form of PRL is a 23 kDa monomeric protein, splice variants of 14, 16, and 22 kDa have been identified. PRL has also been found to be glycosylated, phosphorylated, dimerized, and polymerized. Glycosylation, phosphorylation, dimerization, or polymerization of PRL result in lower activity (2).
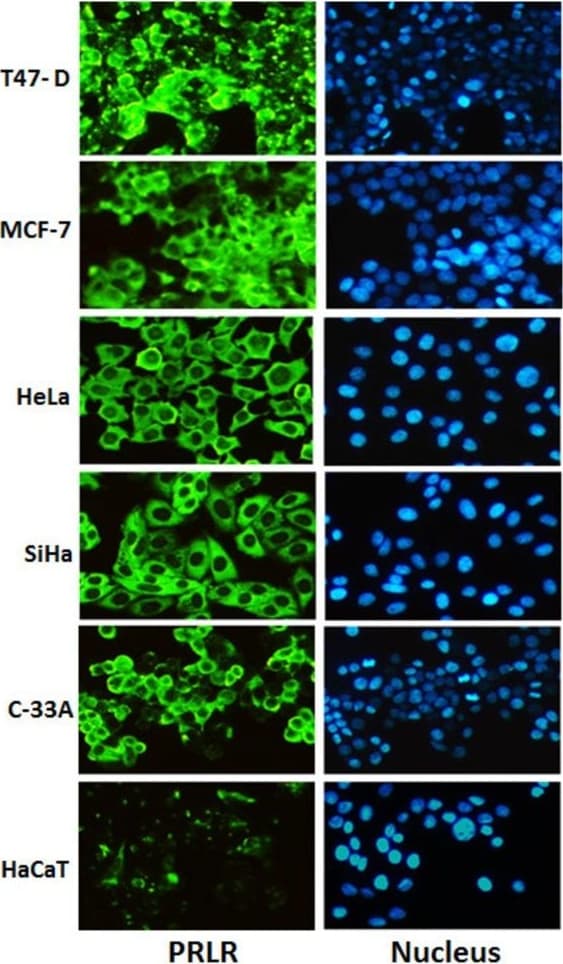
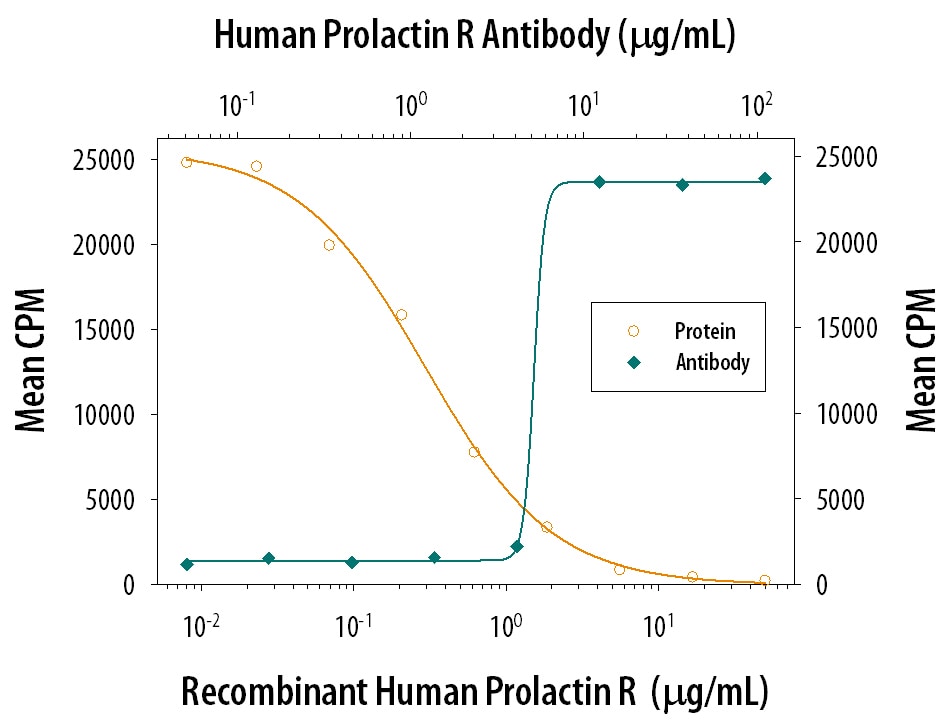
Cell activation by PRL is mediated by a single chain membrane-bound protein belonging to the class 1 cytokine superfamily. The PRL receptor (PRL R) contains an extracellular, transmembrane, and intracellular domain. Transcriptional regulation of the PRL R gene results in several different species-dependent isoforms of PRL R being produced. Although the cytoplasmic domains of the different isoforms vary in length and composition, their extracellular domains are identical. In rats, three major PRL receptor isoforms have been described, a short (291 amino acid), an intermediate (393 amino acid), and a long (591 amino acid) (2). PRL receptors are found in mammary tissue, pituitary gland, brain, heart, lung thymus, spleen, liver, pancreas, kidney, adrenal gland, uterus, skeletal muscle, and skin (3). A soluble form of PRL R containing the 206 NH2-terminal amino acids of the extracellular domain is secreted by mammary epithelial cells and is found in milk. Binding of the transmembrane PRL R results in ligand dimerization followed by binding and phosphorylation of Jak2. Jak2 then phosphorylates STAT and the long form of PRL R. C‑src, fyn, and the Ras/Raf/MAP kinase pathway have also been found to be activated upon PRL R ligand binding (2).